LESSONS LEARNED AT THE INTERFACE OF MEDICINE AND PSYCHIATRY
The Psychiatric Consultation Service at Massachusetts General Hospital sees medical and surgical inpatients with comorbid psychiatric symptoms and conditions. During their twice-weekly rounds, Dr Stern and other members of the Consultation Service discuss diagnosis and management of hospitalized patients with complex medical or surgical problems who also demonstrate psychiatric symptoms or conditions. These discussions have given rise to rounds reports that will prove useful for clinicians practicing at the interface of medicine and psychiatry.
Prim Care Companion CNS Disord 2023;25(6):23f03570
Author affiliations are listed at the end of this article.
Have you ever wondered whether thyroid disease can precipitate neuropsychiatric symptoms? Have you ever been uncertain about how to evaluate and treat the neuropsychiatric signs and symptoms of thyroid disease? If you have, then the following case vignette and discussion should prove useful.
CASE VIGNETTE
Ms A, a 45-year-old woman with a history of alcohol use disorder (in sustained remission), hypothyroidism (not receiving thyroid supplementation), and an unspecified history of depression (previously treated with duloxetine) was brought to the emergency department (ED) via emergency medical services after being found unresponsive by her husband. On arrival, she was unresponsive and had cool extremities and a delayed relaxation phase of her deep tendon reflexes. Her heart rate was 41 beats/minute, temperature was 88.2°F, blood pressure was within normal range, respiratory rate was 14 breaths/minute, oxygen saturation was 99%, and body mass index was 26.4 kg/m2. Her complete blood count, metabolic panel, urine toxicology, urinalysis, and troponin, lactate, arterial blood gas, and cortisol levels were all within normal limits. However, her thyroid-stimulating hormone (TSH) level was 110 mIU/L (reference range, 0.27–4.20 mIU/L), free thyroxine (FT4) level was 0.5 ng/dL (reference range, 0.93–1.7 ng/dL), creatine phosphokinase was 240 mcg/L (reference range, 10–120 mcg/L), and chest x-ray revealed a left middle lobe opacity. She was intubated for airway protection and admitted to the intensive care unit (ICU) with a diagnosis of myxedema coma and was started on 200 mcg of intravenous (IV) levothyroxine (LT4), 5 mcg of IV liothyronine (LT3), a stress dose of hydrocortisone (100 mg IV), and empiric antibiotics.
The next day, Ms A was disoriented, hallucinating, sleeping poorly, distractible, and noted to have a flat affect. After she implied that she had overdosed, the psychiatric consultation-liaison (C-L) service was asked to assess her suicide risk and to assist with management of her delirium.
On interview, she was oriented (to self, place, time, and situation) and made appropriate eye contact. Her speech was slowed as was her thinking. She denied having hallucinations, thoughts of suicide, or that she made a suicide attempt. She explained that she had not been taking her thyroid supplementation for several years because she had been “feeling better.” Although she denied drinking alcohol in the past 4 years, her husband said that for the past 2 years she had been concerned that people were “out to get her,” that neighbors were peeking at her through her window, and that prescription medications were “mind-controlling” agents. He disclosed that Ms A did not have a source of income, which was putting a strain on their marriage. She had been in the process of applying for disability but did not follow through with the paperwork because of her paranoia. He also reported that Ms A had been dismissed from her primary care provider practice due to unpaid medical bills and that there were no other options for primary care in their rural community.
Over the next 4 days, her mental status improved (ie, she was able to correctly complete cognitive and attentional tasks). Her thyroid function tests (TFTs) also trended toward normal levels. On hospital day 7, her TSH was 20.24 mIU/L (reference, 0.27–4.20 mIU/L) and FT4 was 1.22 ng/dL (reference, 0.93–1.70 ng/dL). However, she now met criteria for catatonia; both the screening and severity scores on the Bush-Francis Catatonia Rating Scale1 (BFCRS) were 5, with staring, immobility, and decreased oral intake. She mistook some staff as being her family members. An electroencephalogram (EEG) showed generalized slowing but no focal abnormalities. She received 2 mg of intravenous (IV) lorazepam; this reduced her BFCRS score to zero. However, shortly after receiving lorazepam, Ms A became tachycardic, her delusions worsened, and she repeatedly tried to leave her hospital room. This prompted the primary team to stop lorazepam and start aripiprazole 5 mg. However, this failed to lessen her agitation, and she was given oral olanzapine 5 mg, which decreased her restlessness.
How Can Hypothyroidism and Hyperthyroidism Be Diagnosed?
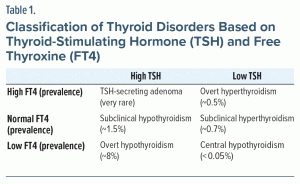
The thyroid axis is driven by the release of thyrotropin-releasing hormone from the hypothalamus, which induces the secretion of TSH from the pituitary gland. TSH, in turn, acts on the thyroid gland to stimulate the synthesis and release of thyroid hormone, mostly in the form of T4. T4 is then converted by the liver and other tissues into the more potent but shorter-lived triiodothyronine (T3). Both T4 and T3 provide central feedback to the hypothalamus and pituitary gland to maintain homeostatic control.2 However, TSH is typically a more sensitive marker of disruptions in the thyroid axis than are T4 and T3.3 Table 1 classifies thyroid disorders based on levels of TSH and FT4.4,5
The prevalence of subclinical hypothyroidism (off treatment) in the United States is estimated at 1.4%, while overt hypothyroidism is estimated at 8.3% based on laboratory results or thyroid hormone replacement.6 In contrast, the prevalence of subclinical hyperthyroidism and overt hyperthyroidism are estimated at 0.7%–2.1% and 0.1%–0.5%, respectively.7,8 Women are approximately 5–8 times more likely to develop thyroid disorders than men.9
Primary hypothyroidism involving thyroid gland dysfunction accounts for > 95% of hypothyroidism. Common causes include autoimmune disease (eg, Hashimoto’s thyroiditis), iatrogenic etiologies (eg, surgery, radiation, medication side effects), inflammation, and iodine imbalance. Central hypothyroidism accounts for < 5% of cases of hypothyroidism. Hypothalamic or pituitary dysfunction results in impaired TSH production, which in turn fails to stimulate the release of thyroid hormone from the thyroid gland. Drugs (eg, glucocorticoids, dopamine, somatostatin analogs) and various pathological processes that impair hypothalamic and pituitary function (eg, tumors, inflammatory disorders, infiltrative diseases, vascular defects) can give rise to central hypothyroidism, which is defined by low TSH and FT4 levels.10
Thyrotoxicosis, or an excess of thyroid hormone, has many etiologies, including Graves’ disease, thyroiditis, thyroid tumors, struma ovarii, exogenous use of thyroid hormone, or side effects of certain medications (eg, amiodarone, iodine/iodinated contrast, LT4, amphetamines).5,11
Some medications can interfere with TFTs in euthyroid individuals and produce results suggestive of thyroid dysfunction. Biotin, a popular dietary supplement that is used to treat neuromuscular disorders, can interfere with the biotinylated commercial assays for TSH and thyroid hormones in a way that mimics the biochemical profile of hyperthyroidism. Other drugs that can cause spurious TFT results in euthyroid individuals include heparin, amiodarone, carbamazepine, phenytoin, and salicylates.2
What Are the Clinical Manifestations of Hypothyroidism and Hyperthyroidism?
Many of the clinical manifestations associated with hypothyroidism are due to a generalized reduction of metabolic processes (eg, fatigue, cold intolerance, bradycardia, weight gain) or accumulation of matrix glycosaminoglycans in the interstitial spaces of tissues (causing voice hoarseness and coarse hair).3 Precipitating events include exposure to cold, heart failure, stroke, trauma, gastrointestinal (GI) bleeding, infections, and drugs (eg, sedatives, analgesics, diuretics).3,9,12
The clinical manifestations of hyperthyroidism are generally characterized by features of increased arousal and adrenergic tone. Common symptoms include palpitations, tremors, insomnia, anxiety, heat intolerance, increased perspiration, and weight loss despite an increased appetite, while some elderly individuals have a paucity of adrenergic symptoms and instead present with depression, lethargy, and weight loss (which has been referred to as “apathetic” thyrotoxicosis).13 At the severe end of the clinical spectrum of hyperthyroidism is thyroid storm, which can be precipitated by surgery, trauma, infection, iodine load, or antithyroid drug withdrawal; it is characterized by delirium, hyperthermia, cardiovascular dysfunction, and GI symptoms.4,5,12
How Are Hypothyroidism and Hyperthyroidism Treated in the Hospital?
Treatment of hypothyroidism consists of thyroid hormone replacement (usually via the oral administration of LT4). The daily replacement dose for young patients with no cardiovascular issues is approximately 1.6 mcg/kg. Because the half-life of LT4 is nearly 1 week, it takes approximately 5 to 6 weeks for levels to equilibrate. The lag between FT4 and TSH normalization depends on both the severity and the duration of the hypothyroidism, and it can range from a few weeks to several months. As LT4 can increase cardiac oxygen demand and induce tachyarrhythmias, the starting LT4 dose is usually lower in older adults and also in those with mild hypothyroidism (eg, the dose may be as low as 25–50 mcg/day, with a slow uptitration every 4–6 weeks).3,9,14
LT4 is normally well absorbed from the GI tract, but it should be taken after fasting and separated from other medications, particularly those containing cations (eg, calcium, magnesium, aluminum, iron, phosphate binders) or bile acid sequestrants. Chronic use of proton pump inhibitors and sucralfate also impair LT4 absorption. Tube feeds should be held for approximately 1 hour before and 1 hour after LT4 administration. In severe cases, or in patients with malabsorption, IV LT4 may be preferred to oral LT4. IV LT4 is commonly dosed at approximately 75% of the oral dose.3,9,14
Myxedema coma is a medical emergency that requires prompt IV administration of LT4 to ensure rapid repletion of thyroid hormone and to bypass impaired GI absorption. The typical starting dose of IV LT4 ranges between 200 and 400 mcg, which is followed by daily IV doses of 50–100 mcg. LT3 is frequently added to IV LT4 for the first few days to accelerate symptomatic reversal. The initial loading dose of LT3 is between 10 and 25 mcg, and it is followed by 2.5–10 mcg every 8 hours, depending on the patient’s age and the presence of cardiovascular risk factors. Because of the condition’s rarity, high-quality trials to determine optimal treatment protocols have not been conducted, but LT3 therapy is typically given for several days until there is clinical improvement. Management of myxedema coma also includes correction of hypocortisolemia, hypoventilation (via intubation and mechanical ventilation), hypothermia, hypovolemia, hyponatremia, hypotension, hypoglycemia, treatment of infection, and identification and elimination of other factors.3,9,12
Thyrotoxicosis from any cause can be treated with β-blockers that reduce the sympathetic hyperactivity that gives rise to symptoms.4 At high doses, most β-blockers can also reduce the peripheral conversion of T4 to T3.15 Additional treatment depends on the cause of the hyperthyroidism.
Initial treatment of Graves’ disease usually involves administration of thionamides, methimazole or propylthiouracil (PTU), to inhibit thyroid hormone synthesis and achieve a euthyroid state. The thyrotoxic phase of thyroiditis is usually transient, and treatment is mostly supportive. Patients with subacute thyroiditis and neck tenderness can be treated with nonsteroidal anti-inflammatory drugs or in more severe cases with prednisone for several weeks to decrease thyroid gland inflammation.4,16
In thyroid storm, high doses of β-blockers and thionamides are administered to control symptoms, block thyroid hormone synthesis, and decrease the conversion of T4 to T3. After thionamide blockade has been initiated, iodine treatment is given to block the release of preformed thyroid hormone from the thyroid gland. Stress dose steroids are also given to further reduce T4 to T3 conversion and treat possible underlying adrenal insufficiency. Bile acid sequestrants can sometimes be added to decrease enterohepatic recycling of thyroid hormone.4,5,12
What Is Myxedema Coma, and How Can It Be Prevented?
The 3 cardinal features of myxedema coma include altered mental status, defective thermoregulation, and a precipitating event. The prototypical patient is an older woman who presents with an altered sensorium during the wintertime, often with a history of hypothyroidism, neck surgery, or radioactive iodine treatment. Somnolence and lethargy have usually been present for several months before the development of stupor/coma. Myxedema coma frequently develops during the winter when body temperature may drop sharply. Precipitating events include exposure to cold, heart failure, stroke, trauma, GI bleeding, infections, and drugs (eg, sedatives, analgesics, diuretics). Avoidance of these precipitating factors and optimization of outpatient management of hypothyroidism are key to preventing future episodes.17 Obtaining objective laboratory findings is essential in the management of myxedma coma; however, the time course for resolution of neuropsychiatric symptoms may fail to correlate with the improvement of TFTs.18
What Medical Risks Are Associated With Treatment of Hyperthyroidism and Hypothyroidism?
Thyroid hormone plays an important role in the regulation of the cardiovascular and skeletal systems and in metabolism. Administration of high-dose thyroid hormone carries the risk of precipitating a fatal tachyarrhythmia, myocardial infarction, or high output heart failure. High doses of LT4 can also give rise to excessive weight loss and symptoms of hyperthyroidism. If the resulting thyrotoxicosis is prolonged, bone loss and osteoporosis may develop.3,14
Thionamides used to treat hyperthyroidism have been associated with rash, agranulocytosis, vasculitis, and hepatotoxicity (especially with PTU). Relative to methimazole, PTU is preferred in the first trimester of pregnancy because of a lower risk of severe birth defects, and methimazole is preferred over PTU in the second and third trimesters because of lower risk of liver failure.4 Radioiodine ablation may aggravate Graves’ orbitopathy (especially in smokers); it is contraindicated in pregnancy due to radiation exposure. Permanent hypothyroidism occurs in most patients, but if the radioactive iodine dose is insufficient, hyperthyroidism may not be cured, and Graves’ disease may recur. Thyroidectomy carries the risks associated with general anesthesia as well as the risk of damaging the recurrent laryngeal nerve. Unilateral damage may lead to hoarseness, and bilateral damage may lead to respiratory distress. Permanent hypoparathyroidism may occur if there is inadvertent damage to, or removal of, the parathyroid glands.19
How Should Myxedema Coma Be Managed After Acute Stabilization, and How Can Recurrence Be Prevented?
After the initial treatment of myxedema coma with high doses of IV LT4 with or without LT3 and glucocorticoids, LT4 treatment is continued with the goal of obtaining full recovery. LT3 is usually stopped after 1 to 2 days when there is evidence of clinical improvement. In the absence of malabsorption, IV LT4 can be converted to the oral formulation based on full replacement doses (1.6 mcg/kg) once the patient is awake and able to take oral medications. Lower maintenance doses may be given in the elderly and in those with cardiac risk factors.9,12
Patients can have significant improvement of hypothyroid symptoms within 2 weeks of starting therapy, with possible normalization of TFTs within this period. However, TSH levels take approximately 6 weeks to reach steady state, and full recovery can take several months. Prior to achieving a steady state, both free T4 and TSH values should be assessed to determine the adequacy of thyroid hormone repletion. FT4 and TSH can be measured every 2 to 3 days for the first 2 weeks, then weekly or every other week to ensure continued improvement. Dose adjustments can be made initially based on the FT4 levels, but as the TSH level reaches steady state, the latter becomes a better indicator of thyroid hormone repletion. TSH levels can then be followed every 4–6 weeks after dose adjustment until euthyroidism is achieved. TSH measurements can then be done every 3–6 months to ensure adequate dosing and assess for possible nonadherence.14
It is also important to make LT4 dose adjustments in the setting of conditions that may alter LT4 requirements. These include changes in medications/supplements or the development of medical conditions that alter the absorption, metabolism, and clearance of thyroid hormone.14 TFTs may be more difficult to interpret during combination therapy due to the greater potency and short half-life of LT3, which results in greater biochemical variability. Additional studies are needed to determine if LT4/LT3 combination therapy is beneficial in patients who harbor genetic variants that affect the efficacy of T4 to T3 conversion or alter the expression of thyroid hormone transporters in the brain or pituitary gland.20
Efforts to prevent myxedema coma can be complicated by both patient and health care system barriers that influence treatment adherence and ongoing follow-up. System-level barriers include changes in insurance and provider availability or geography, and these factors should be considered when planning post-hospitalization follow-up. Individually, substance use disorders (SUDs), psychiatric impairment, recent immigration, and other factors can also impede ongoing treatment.21 Additionally, the brain fog that patients may experience as a symptom of hypothyroidism can make it more difficult for patients to stick to a treatment plan.22 From the SUD literature, medication adherence rates can be improved through direct observation of treatment, motivational interviewing, and case management services.21,23,24 Given the long half-life of LT4, dosing under observations 1 to 2 times weekly is feasible and may be useful for patients struggling with medication adherence on their own.25 It is also important to address psychosocial outcomes associated with myxedema coma and its neuropsychiatric symptoms (eg, financial/employment problems, family/relationship problems) through social services or family/couple–based counseling interventions.26
How Does Thyroid Dysregulation Adversely Impact Mental Status?
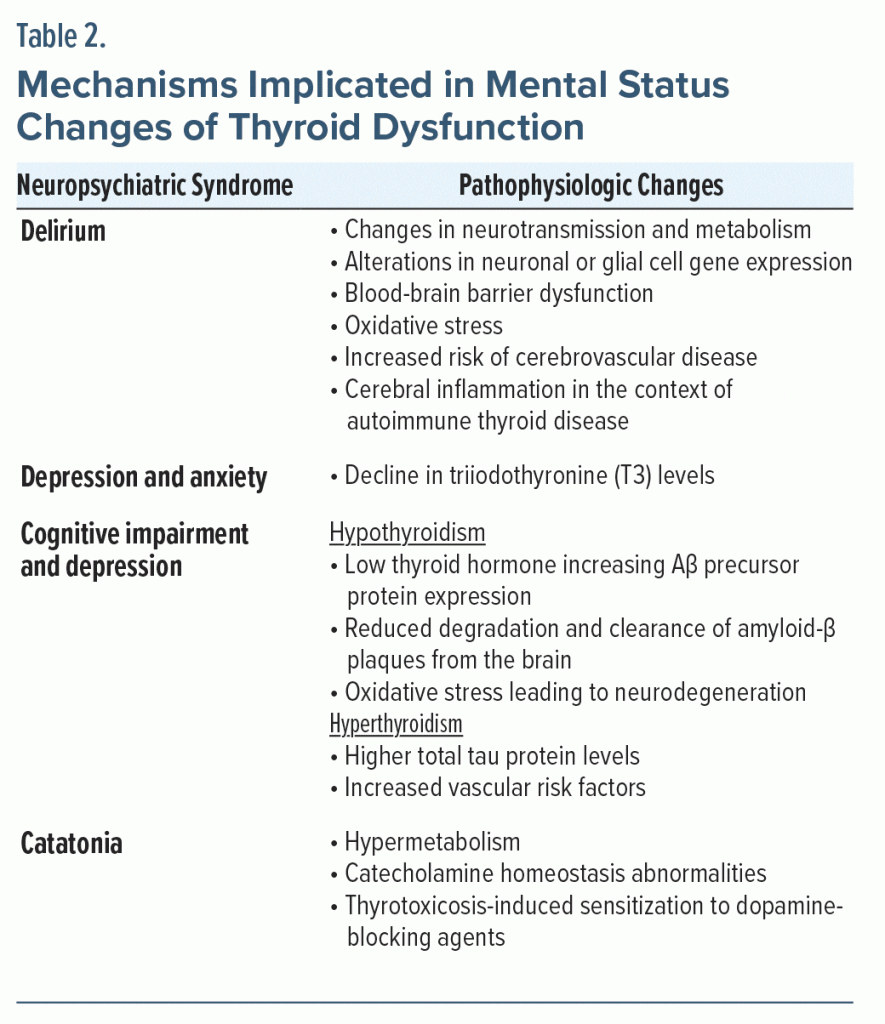
An altered mental status can result from derangements in any organ system. Therefore, utilizing a systems-based approach can aid in the identification of the complexities and comorbidities of potentially life-threatening causes of delirium or catatonia.27,28 The pathophysiology of the thyroid hormone–related changes in the brain is diverse and summarized in Table 2.29
Intracellular T3 controls the level and enhances the effect of serotonin and norepinephrine.30 A decline in T3 levels can cause depression and anxiety through its impact on these neurotransmitters. Augmenting an antidepressant with T3 is a common pharmacologic strategy for treatment-resistant depression.31 Adrenergic hyperactivity, which is seen in patients with hyperthyroidism, also may lead to neuropsychiatric symptoms (eg, agitation).32
Possible mechanisms for cognitive impairment and dementia in thyroid dysfunction include low levels of thyroid hormone that increase amyloid-β (Aβ) precursor protein expression and consequently higher Aβ levels in the central nervous system (CNS). There may also be reduced degradation and clearance of Aβ plaques from the brain. In hyperthyroid patients, oxidative stress from decreased antioxidant metabolites may lead to neurodegeneration and neuronal death. Hyperthyroidism is also associated with increased vascular risk factors that can contribute to cognitive impairment.33
Regarding catatonia, a classic study by Gjessing (1974)34 proposed an association between catatonia and hypermetabolism. This work also implicated endocrine factors such as catecholamine homeostasis abnormalities in catatonia and suggested that thyroxine administration could prevent recurrent catatonic episodes. Additionally, thyrotoxicosis may produce sensitization to medications such as dopamine-blocking agents that can induce a catatonic reaction.35
What Types of Neuropsychiatric Symptoms Can Be Caused by Thyroid Disease?
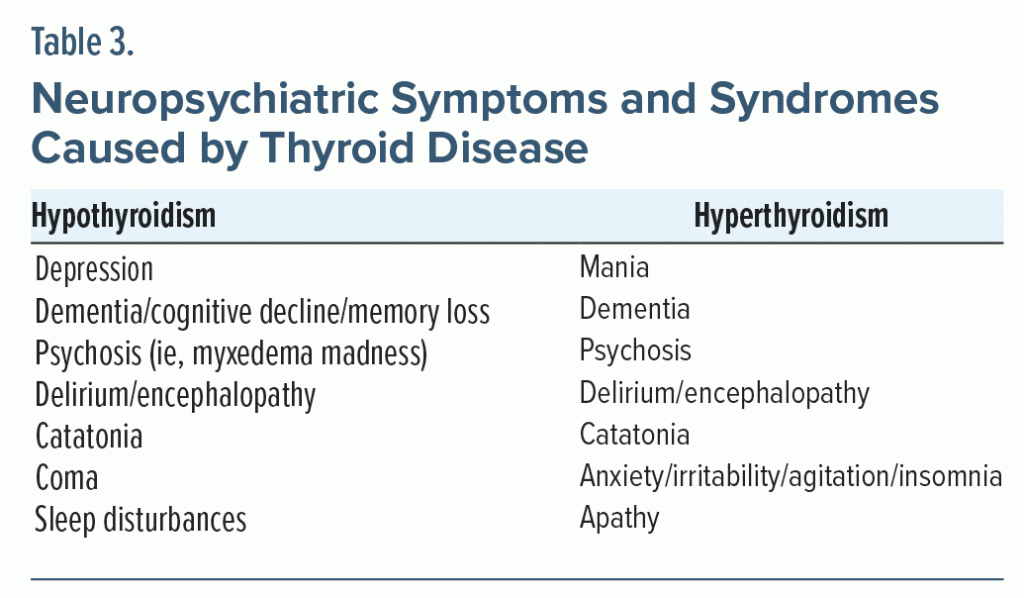
Thyroid disease can give rise to myriad psychiatric sequelae (Table 3).29 Although these manifestations can be reversed by normalizing thyroid function, neuropsychiatric symptoms may linger after the normalization of abnormal TFTs.11,18
For decades, hypothyroidism has been known to mimic depression.36 Symptoms common to both conditions include depressed mood, anhedonia, decreased libido, appetite loss, fatigue, impaired concentration, and sleep disturbance.37,38 Various theories attempt to explain this association, including modulation of serotonin receptor activity in the brain by T3 or T4, changes in circadian TSH release in depressed patients, or centrally-acting effects of antithyroid peroxidase (TPO) antibodies on astrocytes or serum cytokine concentrations.38,39 Although the data are mixed, a recent meta-analysis40 demonstrated a moderate association between hypothyroidism and depressive disorders, which was found to be greater in females than in males. Furthermore, Tang et al39 found that the prevalence of depressive disorders in patients with subclinical hypothyroidism was higher than in the general population; however, when stratified by age, the association was found only in those aged ≥ 50 years. The above associations suggest that individuals with subclinical hypothyroidism and overt hypothyroidism should be screened for depression, and individuals with depression should be screened for thyroid disorders.41,42 There is some evidence to suggest an association between elevated anti-TPO antibodies and depressive/anxiety disorders43; however, this finding was not supported by a recent meta-analysis.40
Depression induced by hypothyroidism often improves with LT4 treatment, although this may not always lead to complete remission of depressive symptoms.11,18,41,42 The literature indicates that bringing TSH into a normal range is typically sufficient to treat depressive symptoms caused by overt hypothyroidism.44 However, in subclinical hypothyroidism, LT4 did not show improvement in psychological distress scores when compared to placebo.42 In patients whose depressive symptoms persist after normalization of their TFTs, additional workup can include a complete blood count, a comprehensive metabolic panel, an erythrocyte sedimentation rate, tissue transglutaminase antibody, a sleep study, vitamin D levels, and cortisol levels.45 If this testing is unrevealing, lifestyle factor optimization is appropriate (eg, improving diet, exercise, sleep habits, and stress reduction), as well as the assessment and treatment of any contributing primary psychiatric disorder.45
Psychotic symptoms are another possible neuropsychiatric presentation of hypothyroidism, appearing in roughly 5%–15% of affected patients.46,47 This association was first reported in 1888 by the Committee on Myxedema of the Clinical Society of London, and the descriptor “myxedema madness” was coined by Asher in 1949 to describe psychosis induced by hypothyroidism.47–50 Physical symptoms of hypothyroidism usually occur months to years before the onset of thyroid-related psychosis, although this presentation can be variable.11,51,52 In cases of psychosis induced by hypothyroidism, thyroid replacement is the first-line therapy. While antipsychotics can supplement replacement therapy, recent data suggest that antipsychotics may not hasten recovery.53,54 In addition, antipsychotics may reduce levels of free T3/T4 and increase TSH, thereby worsening hypothyroidism. Moreover, decreases in free T4 levels are negatively correlated with improvement in psychotic symptoms.55,56 However, antipsychotics are indicated in certain conditions (eg, acute agitation or concern for harm to self or hospital staff).
Another possible neuropsychiatric sequela of hypothyroidism is Hashimoto’s encephalopathy, also known as steroid-responsive encephalopathy associated with autoimmune thyroiditis (SREAT).57 SREAT is characterized by serum thyroid antibodies and a variety of symptoms that improve with steroid therapy. Symptoms of SREAT include cognitive impairment, delirium, paranoia, hallucinations, tremor, myoclonus, seizures, gait difficulties, and sleep abnormalities.58,59 In addition to steroids, immunomodulators (eg, azathioprine, cyclophosphamide, methotrexate, chloroquine, immunoglobulins, and rituximab) may be required to treat this condition.59 SREAT and other autoimmune encephalitides often go undiagnosed and untreated if they are presumed to be manifestations of a primary psychotic disorder.
Neuropsychiatric symptoms can be seen in hyperthyroidism of any etiology, including the possibility of a mania-like state.30 It may be difficult to differentiate such a state from bipolar mania, but in cases that are of a purely thyroid etiology, one can expect to see the presence of other physical symptoms of the thyroid derangement as discussed previously. Additionally, obtaining collateral information about similar prior episodes, diagnosis, and treatment history may help with differentiation. It is also relevant to consider that the peak age of first symptoms of bipolar mania is 15–19 years old, whereas symptoms due to hypothyroidism or hyperthyroidism would peak according to the timeline of the underlying thyroid dysfunction.60 However, there is a complex interplay between the treatment of mood disorders and thyroid function. For example, hypothyroidism can be a complication of treatment with lithium, and thyroid hormones have been used to treat bipolar mood states. This complexity highlights the importance of obtaining TFTs in patients presenting with any type of mood episode.61–63
Other symptoms specific to thyrotoxicosis include anxiety, dysphoria, and emotional lability.64 Depressive disorders have been found in 31%–69% of patients with thyrotoxicosis, while anxiety disorders have developed in 60%.65,66 In older adults, neuropsychiatric presentations of thyrotoxicosis include depression, lethargy, cognitive impairment, and apathy.11,67,68 Additionally, hyperthyroidism-induced psychosis has been described in case reports, although it appears to develop less frequently than in hypothyroidism.69,70 Thyroid storm can include symptoms such as hyperirritability, anxiety, confusion, apathy, and coma.11 Meanwhile, in subclinical hyperthyroidism, nervousness and irritability are the predominant symptoms.11 One study67 showed that patients > 55 years of age with subclinical hyperthyroidism have a 3-fold increased risk of dementia and Alzheimer’s disease; however, this finding was not replicated in a follow-up study.71
Like hypothyroidism, the treatment of psychiatric symptoms secondary to hyperthyroidism is predicated on the correction of the underlying thyroid dysfunction.72 In cases of hyperthyroidism-induced psychosis, correction of the hyperthyroid state with antithyroid medications, radioactive iodine ablation, or surgery is the “gold standard” treatment.69,70
Nonetheless, it is important to note that several studies73–76 have shown that neuropsychiatric symptoms remain even after correction of thyroid levels and resolution of other symptoms of the thyroid condition. In one study, 43% of patients who previously had hyperthyroidism but who were treated and obtained a euthyroid state had elevated scores on a neuropsychological rating scale of depression, mania, hostility, anxiety, and sleep disturbance versus only 10% of controls without hyperthyroidism.74 This study also found that neuropsychological symptoms differed based on the length of time after normalization of TFTs: patients < 6 months after normalization scored higher on anxiety, mania, and hostility, while patients > 6 months after normalization scored higher in symptoms of depression and sleep disturbance.74 Given the frequency of neuropsychiatric symptoms, thyroid dysfunction can be easily mistaken for several primary psychiatric disorders, and studies have shown that patients are often misdiagnosed or delay presenting for care due to the stigma associated with psychiatric conditions.11,73 When assessing a patient with such psychiatric symptoms of an unclear etiology, consideration of their past history of psychiatric disorders, past history of thyroid disorders, and level of medication adherence for both types of conditions can help with making an accurate diagnosis.
What Is Catatonia and How Is It Treated?
In the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, Text Revision, catatonia is defined as the presence of 3 or more of 12 possible motor and behavioral symptoms.77–79 Such motor symptoms include catalepsy, waxy flexibility, grimacing, maintenance of posture, and lack of psychomotor activity.80 Behavioral symptoms include mutism, oppositionality, stereotypies, agitation, echo phenomena, and caricatures of normal actions.79 Historically, catatonia has been mistaken as a syndrome occurring only as a subtype of schizophrenia; however, in the last half century it has been recognized to also occur in the context of mood disorders and neurologic/medical conditions, and identification and treatment of the underlying cause is critical in the management of this condition.78 The term malignant catatonia has been used to describe some cases involving hyperthermia or autonomic instability, both of which are indicators of a higher mortality risk and require urgent intervention.81
In clinical practice, the BFCRS is often used to measure catatonia severity and response to treatment, which typically starts with use of IV lorazepam. However, electroconvulsive therapy (ECT) is often highly effective for treating catatonia, and it should be used in cases that are refractory to medication management and in cases of malignant catatonia.77,80,81 Discontinuation of dopamine-blocking medications is important, as is restarting dopamine agonists that have recently been withdrawn.77,81 Additional supportive care includes maintaining adequate hydration and nutrition status, mobilization, anticoagulation, aspiration precautions, and cooling, if necessary.77,81 In catatonia secondary to thyroid dysfunction, it may be even more important to avoid dopamine antagonists. Through disruption in CNS metabolism, hypothyroid states can result in disordered neurotransmission and even structural changes in the brain.82 Additionally, there is evidence in animal models that the blood-brain barrier is disrupted in chronic hypothyroidism.83 This could mean that patients with severe, untreated hypothyroidism are more susceptible to adverse effects of medications that are active in the CNS.
What Happened to Ms A?
Over the next 2 days, during which she received no additional psychotropics, Ms A’s tachycardia resolved, but the catatonic symptoms worsened. At this point, her BFCRS increased to a screening score of 8 and a severity score of 10, with immobility, mutism, staring, posturing, negativism, decreased oral intake, perseveration, and autonomic instability. Lorazepam was resumed (3 times daily). Although she intermittently refused several doses, her adherence improved, and within 72 hours her BFCRS was zero. The endocrinology team adjusted her LT4 dose, with a final dose of 125 mcg/d. After being medically cleared by the primary team, she was transferred to the inpatient psychiatry service for further management of catatonia.
Her length of stay on the psychiatric unit was 6 days. Due to her alcohol use disorder history, she was uncomfortable with using long-term benzodiazepines, and she requested a scheduled taper before discharge; therefore, her lorazepam was weaned and discontinued without recurrence of catatonia. The endocrinology team determined that her TFTs were stable, with TSH around 15 mIU/L (reference, 0.27–4.20 mIU/L), and they made no further dose changes. She was discharged on LT4 and vitamin B12. Follow-up was arranged with an outpatient psychiatrist as well as with a primary care provider through the hospital-based practice. This was not ideal, as Ms A lived 2 hours away; however, there were no primary care providers in her region with openings in their practice who were accepting new patients. At the time of discharge, Ms A was also looking into participating in a vocational rehabilitation program to improve her financial situation and her relationship with her husband. Since her hospital discharge (with 2 years of follow-up care), there have been no changes to her medication regimen and no documented recurrences of catatonia or psychosis.
CONCLUSION
The case of Ms A illustrates the complexities of diagnosis and treatment of neuropsychiatric manifestations of medical (including endocrinologic) disorders. In general, patients with hypothyroidism, such as Ms A, are at risk of developing neuropsychiatric symptoms (such as depression, psychosis, and encephalopathy).36,46,47,57 Treatment of these symptoms involves normalizing the underlying thyroid dysfunction with thyroid replacement and administration of steroids in the case of SREAT.11,59 Antipsychotics should not be the primary treatment, but they can be used in the setting of behavioral emergencies. During her hospital course, Ms A also developed catatonia, a syndrome with motor signs as well as changes in mood, behavior, and thought.77 Although catatonia has historically been attributed to underlying mood or psychotic disorders, this phenomenon is also caused by neurologic/medical conditions such as hypothyroidism.78 Of note, catatonia secondary to hypothyroidism may be very responsive to ECT), while patients diagnosed with catatonia due to hyperthyroidism should not be treated with ECT due to risk of thyrotoxicosis.84 Ms A’s catatonia was treated successfully with lorazepam.77,81
Article Information
Published Online: December 26, 2023. https://doi.org/10.4088/PCC.23f03570
© 2023 Physicians Postgraduate Press, Inc.
Submitted: May 20, 2023; accepted August 30, 2023.
To Cite: Rasmussen MJ, Robbins H, Fipps DC, et al. Neuropsychiatric sequelae of thyroid dysfunction: evaluation and management. Prim Care Companion CNS Disord. 2023;25(6):23f03570.
Author Affiliations: Dartmouth Hitchcock Medical Center, Lebanon, New Hampshire (Rasmussen, Robbins); Mayo Clinic Alix School of Medicine, Rochester, Minnesota (Fipps); Transplant Center, Mayo Clinic, Rochester, Minnesota (Fipps); Psychiatry and Psychology, Mayo Clinic, Rochester, Minnesota (Fipps); Addiction Services, Mayo Clinic, Rochester, Minnesota (Fipps); Department of Psychiatry, Geisel School of Medicine at Dartmouth, Lebanon, New Hampshire (Rustad); Department of Psychiatry, Larner College of Medicine at the University of Vermont, Burlington (Rustad); Department of Mental Health and Behavioral Sciences, White River Junction VA Medical Center, White River Junction, Vermont (Rustad); Center for Endocrine Genetics, Brigham and Women’s Hospital, Boston, Massachusetts (Pallais); Brigham and Women’s Hospital Internal Medicine Residency Program, Boston, Massachusetts (Pallais); Department of Psychiatry, Massachusetts General Hospital, Boston, Massachusetts (Stern); Harvard Medical School, Boston, Massachusetts (Stern).
Corresponding Author: Matthew J. Rasmussen, MD, Dartmouth Hitchcock Medical Center, Department of Psychiatry, 1 Medical Center Dr, Lebanon, NH 03766 ([email protected]).
Relevant Financial Relationships: None.
Funding/Support: None.
Previous Presentation: Presented in part at Academy of Consult Liaison Psychiatry, C-L Psychiatry Case Conference; October 13, 2021; virtual (Rasmussen MJ, Rustad JK [discussant]. “I need to be more decisive”: a case conference”).
CLINICAL POINTS
- The mainstay of hypothyroidism treatment is thyroid hormone replacement with levothyroxine; in cases of myxedema coma, this should be administered intravenously, and coadministration of liothyronine should be considered for more rapid symptom reversal.
- Thyroid disorders can cause a variety of neuropsychiatric symptoms including depression, psychosis, encephalopathy, anxiety, and mania.
- Treatment of psychosis that is secondary to hypothyroidism should focus on normalization of thyroid levels; antipsychotic agents can be used adjunctively to manage behavioral emergencies.
- Catatonia may occur in the context of both hypothyroidism and hyperthyroidism; treatment should involve both correction of the underlying thyroid disorder, as well as treatment of catatonia, typically with benzodiazepines or electroconvulsive therapy.
References (84)

- Bush G, Fink M, Petrides G, et al. Catatonia. I. Rating scale and standardized examination. Acta Psychiatr Scand. 1996;93(2):129–136. PubMed CrossRef
- Burch HB. Drug effects on the thyroid. N Engl J Med. 2019;381(8):749–761.
- Chaker L, Razvi S, Bensenor IM, et al. Hypothyroidism. Nat Rev Dis Primers. 2022;8(1):30.
- Ross DS, Burch HB, Cooper DS, et al. 2016 American thyroid association guidelines for diagnosis and management of hyperthyroidism and other causes of thyrotoxicosis. Thyroid. 2016;26(10):1343–1421. PubMed CrossRef
- De Leo S, Lee SY, Braverman LE. Hyperthyroidism. Lancet. 2016;388(10047):906–918. PubMed CrossRef
- Wyne KL, Nair L, Schneiderman CP, et al. Hypothyroidism prevalence in the United States: a retrospective study combining national health and nutrition examination survey and claims data, 2009–2019. J Endocr Soc. 2022;7(1):bvac172. PubMed CrossRef
- Hollowell JG, Staehling NW, Flanders WD, et al. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab. 2002;87(2):489–499. PubMed CrossRef
- Canaris GJ, Manowitz NR, Mayor G, et al. The Colorado thyroid disease prevalence study. Arch Intern Med. 2000;160(4):526–534. PubMed CrossRef
- Jonklaas J, Bianco AC, Bauer AJ, et al; American Thyroid Association Task Force on Thyroid Hormone Replacement. Guidelines for the treatment of hypothyroidism: prepared by the american thyroid association task force on thyroid hormone replacement. Thyroid. 2014;24(12):1670–1751. PubMed CrossRef
- Beck-Peccoz P, Rodari G, Giavoli C, et al. Central hypothyroidism: a neglected thyroid disorder. Nat Rev Endocrinol. 2017;13(10):588–598. PubMed CrossRef
- Feldman AZ, Shrestha RT, Hennessey JV. Neuropsychiatric manifestations of thyroid disease. Endocrinol Metab Clin North Am. 2013;42(3):453–476. PubMed CrossRef
- Ylli D, Klubo-Gwiezdzinska J, Wartofsky L. Thyroid emergencies. Pol Arch Intern Med. 2019;129(7-8):526–534. PubMed
- Papaleontiou M, Haymart MR. Approach to and treatment of thyroid disorders in the elderly. Med Clin North Am. 2012;96(2):297–310. PubMed CrossRef
- Duntas LH, Jonklaas J. Levothyroxine dose adjustment to optimise therapy throughout a patient’s lifetime. Adv Ther. 2019;36(suppl 2):30–46. PubMed CrossRef
- Perrild H, Hansen JM, Skovsted L, et al. Different effects of propranolol, alprenolol, sotalol, atenolol and metoprolol on serum T3 and serum rT3 in hyperthyroidism. Clin Endocrinol (Oxf). 1983;18(2):139–142. PubMed CrossRef
- Ray I, D’Souza B, Sarker P, et al. Management of subacute thyroiditis: a systematic review of current treatment protocols. Int J Gen Med. 2022;15:6425–6439. PubMed CrossRef
- Wiersinga WM. Myxedema and coma (severe hypothyroidism). In: Feingold KR, Anawalt B, Boyce A, et al, eds. Endotext [Internet]. South Dartmouth, Massachusetts: MDText.com, Inc. 2000.
- Davis JD, Tremont G. Neuropsychiatric aspects of hypothyroidism and treatment reversibility. Minerva Endocrinol. 2007;32(1):49–65. PubMed
- Kravets I. Hyperthyroidism: Diagnosis and Treatment. Am Fam Physician. 2016;93(5):363–370. PubMed
- Jonklaas J, Bianco AC, Cappola AR, et al. Evidence-based use of levothyroxine/liothyronine combinations in treating hypothyroidism: a consensus document. Thyroid. 2021;31(2):156–182. PubMed CrossRef
- Clements KM, Hydery T, Tesell MA, et al. A systematic review of community-based interventions to improve oral chronic disease medication regimen adherence among individuals with substance use disorder. Drug Alcohol Depend. 2018;188:141–152. PubMed CrossRef
- Haskard-Zolnierek K, Wilson C, Pruin J, et al. The relationship between brain fog and medication adherence for individuals with hypothyroidism. Clin Nurs Res. 2022;31(3):445–452. PubMed CrossRef
- Durvasula R, Miller TR. Substance abuse treatment in persons with HIV/AIDS: challenges in managing triple diagnosis. Behav Med. 2014;40(2):43–52. PubMed CrossRef
- Penzenstadler L, Machado A, Thorens G, et al. Effect of case management interventions for patients with substance use disorders: a systematic review. Front Psychiatry. 2017;8:51. PubMed CrossRef
- Lipp HP. Administration and pharmacokinetics of levothyroxine. In: Kahaly GJ, ed. 70 Years of Levothyroxine [Internet]. Cham, Switzerland: Springer; 2021.
- Berry E, Davies M, Dempster M. Exploring the effectiveness of couples interventions for adults living with a chronic physical illness: a systematic review. Patient Educ Couns. 2017;100(7):1287–1303. PubMed CrossRef
- Fricchione GL, Nejad SH, Esses JA, et al. Postoperative delirium. Am J Psychiatry. 2008;165(7):803–812. PubMed CrossRef
- Oldham MA. The probability that catatonia in the hospital has a medical cause and the relative proportions of Its causes: a systematic review. Psychosomatics. 2018;59(4):333–340. PubMed CrossRef
- Jurado-Flores M, Warda F, Mooradian A. Pathophysiology and clinical features of neuropsychiatric manifestations of thyroid disease. J Endocr Soc. 2022;6(2):bvab194. PubMed CrossRef
- Fukao A, Takamatsu J, Arishima T, et al. Graves’ disease and mental disorders. J Clin Transl Endocrinol. 2019;19:100207. PubMed
- Aronson R, Offman HJ, Joffe RT, et al. Triiodothyronine augmentation in the treatment of refractory depression. a meta-analysis. Arch Gen Psychiatry. 1996;53(9):842–848. PubMed CrossRef
- Urias-Uribe L, Valdez-Solis E, González-Milán C, et al. Psychosis crisis associated with thyrotoxicosis due to Graves’ disease. Case Rep Psychiatry. 2017;2017:6803682. PubMed CrossRef
- Joy Mathew C, Jose MT, Elshaikh AO, et al. Is hyperthyroidism a possible etiology of early onset dementia? Cureus. 2020;12(9):e10603. PubMed CrossRef
- Gjessing LR. A review of periodic catatonia. Biol Psychiatry. 1974;8(1):23–45. PubMed
- Iskandar M, Stepanova E, Francis A. Two cases of catatonia with thyroid dysfunction. Psychosomatics. 2014;55(6):703–707. PubMed CrossRef
- Whybrow PC, Prange AJ Jr, Treadway CR. Mental changes accompanying thyroid gland dysfunction. a reappraisal using objective psychological measurement. Arch Gen Psychiatry. 1969;20(1):48–63. PubMed CrossRef
- Boonen E, Van den Berghe G. Endocrine responses to critical illness: novel insights and therapeutic implications. J Clin Endocrinol Metab. 2014;99(5):1569–1582. PubMed CrossRef
- Kotkowska Z, Strzelecki D. Depression and autoimmune hypothyroidism-their relationship and the effects of treating psychiatric and thyroid disorders on changes in clinical and biochemical parameters including BDNF and other cytokines: a systematic review. Pharmaceuticals (Basel). 2022;15(4):391. PubMed CrossRef
- Tang R, Wang J, Yang L, et al. Subclinical hypothyroidism and depression: a systematic review and meta-analysis. Front Endocrinol (Lausanne). 2019;10:340. PubMed CrossRef
- Bode H, Ivens B, Bschor T, et al. Association of hypothyroidism and clinical depression: a systematic review and meta-analysis. JAMA Psychiatry. 2021;78(12):1375–1383. PubMed CrossRef
- Dayan CM, Panicker V. Hypothyroidism and depression. Eur Thyroid J. 2013;2(3):168–179. PubMed CrossRef
- Samuels MH. Psychiatric and cognitive manifestations of hypothyroidism. Curr Opin Endocrinol Diabetes Obes. 2014;21(5):377–383. PubMed CrossRef
- Carta MG, Loviselli A, Hardoy MC, et al. The link between thyroid autoimmunity (antithyroid peroxidase autoantibodies) with anxiety and mood disorders in the community: a field of interest for public health in the future. BMC Psychiatry. 2004;4(1):25. PubMed CrossRef
- Walsh JP, Ward LC, Burke V, et al. Small changes in thyroxine dosage do not produce measurable changes in hypothyroid symptoms, well-being, or quality of life: results of a double-blind, randomized clinical trial. J Clin Endocrinol Metab. 2006;91(7):2624–2630. PubMed CrossRef
- McDermott MT. Does combination T4 and T3 therapy make sense? Endocr Pract. 2012;18(5):750–757. PubMed CrossRef
- Hall RC. Psychiatric effects of thyroid hormone disturbance: psychosomatic illness review: no 5 in a series. Psychosomatics. 1983;24(1):7–11, 15–18. PubMed CrossRef
- Heinrich TW, Grahm G. Hypothyroidism presenting as psychosis: myxedema madness revisited. Prim Care Companion J Clin Psychiatry. 2003;5(6):260–266. PubMed CrossRef
- Kaplan JL, Castro-Revoredo I. Severe hypothyroidism manifested as acute mania with psychotic features: a case report and review of the literature. J Psychiatr Pract. 2020;26(5):417–422. PubMed CrossRef
- Asher R. Myxoedematous madness. BMJ. 1949;2(4627):555–562. PubMed CrossRef
- Clinical Society of London. Report of a Committee of the Clinical Society of London, Nominated December 14, 1883, to Investigate the Subject of Myxoedema. Supplement to Volume 21. Longmans, Green, and Company; 1888.
- Logothetis J. Psychotic behaviors as the initial indicator of adult myxedema. J Nerv Ment Dis. 1963;136(6):561–568. CrossRef
- Azzopardi L, Murfin C, Sharda A, et al. Myxoedema madness. BMJ Case Rep. 2010:bcr0320102841. PubMed CrossRef
- Schofield A, Bracken P. Thyroid-induced psychosis in myxoedema. Ir Med J. 1983;76(12):495–496. PubMed
- Sanchez DL, Fusick AJ. Psychosis due to hypothyroidism: are antipsychotics indicated? Int J Case Rep. 2019;3:103.
- Bunevicius R, Steibliene V, Prange AJ Jr. Thyroid axis function after in-patient treatment of acute psychosis with antipsychotics: a naturalistic study. BMC Psychiatry. 2014;14(1):279. PubMed CrossRef
- Steibliene V, Bunevicius A, Savickas A, et al. Triiodothyronine accelerates and enhances the antipsychotic effect of risperidone in acute schizophrenia. J Psychiatr Res. 2016;73:9–16. PubMed CrossRef
- Lee SW, Donlon S, Caplan JP. Steroid responsive encephalopathy associated with autoimmune thyroiditis (SREAT) or Hashimoto’s encephalopathy: a case and review. Psychosomatics. 2011;52(2):99–108. PubMed CrossRef
- Castillo P, Woodruff B, Caselli R, et al. Steroid-responsive encephalopathy associated with autoimmune thyroiditis. Arch Neurol. 2006;63(2):197–202. PubMed CrossRef
- Laurent C, Capron J, Quillerou B, et al. Steroid-responsive encephalopathy associated with autoimmune thyroiditis (SREAT): Characteristics, treatment and outcome in 251 cases from the literature. Autoimmun Rev. 2016;15(12):1129–1133. PubMed CrossRef
- Bauer M, Pfennig A. Epidemiology of bipolar disorders. Epilepsia. 2005;46(suppl 4):8–13. PubMed CrossRef
- Bennett B, Mansingh A, Fenton C, et al. Graves’ disease presenting with hypomania and paranoia to the acute psychiatry service. BMJ Case Rep. 2021;14(2):e236089. PubMed CrossRef
- Sathya A, Radhika R, Mahadevan S, et al. Mania as a presentation of primary hypothyroidism. Singapore Med J. 2009;50(2):e65–e67. PubMed
- Walshaw PD, Gyulai L, Bauer M, et al. Adjunctive thyroid hormone treatment in rapid cycling bipolar disorder: a double-blind placebo-controlled trial of levothyroxine (L-T4) and triiodothyronine (T3). Bipolar Disord. 2018;20(7):594–603. PubMed CrossRef
- Marqusee E, Haden ST, Utiger RD. Subclinical thyrotoxicosis. Endocrinol Metab Clin North Am. 1998;27(1):37–49. PubMed CrossRef
- Kathol RG, Delahunt JW. The relationship of anxiety and depression to symptoms of hyperthyroidism using operational criteria. Gen Hosp Psychiatry. 1986;8(1):23–28. PubMed CrossRef
- Trzepacz PT, McCue M, Klein I, et al. A psychiatric and neuropsychological study of patients with untreated Graves’ disease. Gen Hosp Psychiatry. 1988;10(1):49–55. PubMed CrossRef
- Kalmijn S, Mehta KM, Pols HA, et al. Subclinical hyperthyroidism and the risk of dementia: the Rotterdam study. Clin Endocrinol (Oxf). 2000;53(6):733–737. PubMed CrossRef
- Taylor JW. Depression in thyrotoxicosis. Am J Psychiatry. 1975;132(5):552–553. PubMed CrossRef
- Lazarus A, Jaffe R. Resolution of thyroid-induced schizophreniform disorder following subtotal thyroidectomy: case report. Gen Hosp Psychiatry. 1986;8(1):29–31. PubMed CrossRef
- Brownlie BE, Rae AM, Walshe JW, et al. Psychoses associated with thyrotoxicosis - ‘thyrotoxic psychosis: a report of 18 cases, with statistical analysis of incidence. Eur J Endocrinol. 2000;142(5):438–444. PubMed CrossRef
- Van Der Cammen TJ, Mattace-Raso F, van Harskamp F, et al. Lack of association between thyroid disorders and Alzheimer’s disease in older persons: a cross-sectional observational study in a geriatric outpatient population. J Am Geriatr Soc. 2003;51(6):884. PubMed CrossRef
- Reid JR, Wheeler SF. Hyperthyroidism: diagnosis and treatment. Am Fam Physician. 2005;72(4):623–630. PubMed
- Stern RA, Robinson B, Thorner AR, et al. A survey study of neuropsychiatric complaints in patients with Graves’ disease. J Neuropsychiatry Clin Neurosci. 1996;8(2):181–185. PubMed CrossRef
- Bommer M, Eversmann T, Pickardt R, et al. Psychopathological and neuropsychological symptoms in patients with subclinical and remitted hyperthyroidism. Klin Wochenschr. 1990;68(11):552–558. PubMed CrossRef
- Bunevicius R, Velickiene D, Prange AJ Jr. Mood and anxiety disorders in women with treated hyperthyroidism and ophthalmopathy caused by Graves’ disease. Gen Hosp Psychiatry. 2005;27(2):133–139. PubMed CrossRef
- Fahrenfort JJ, Wilterdink AM, van der Veen EA. Long-term residual complaints and psychosocial sequelae after remission of hyperthyroidism. Psychoneuroendocrinology. 2000;25(2):201–211. PubMed CrossRef
- Francis A. Catatonia: diagnosis, classification, and treatment. Curr Psychiatry Rep. 2010;12(3):180–185. PubMed CrossRef
- Tandon R, Heckers S, Bustillo J, et al. Catatonia in DSM-5. Schizophr Res. 2013;150(1):26–30. PubMed CrossRef
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorder, DSM-V-TR. Fifth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2022.
- Philbrick KL, Rummans TA. Malignant catatonia. J Neuropsychiatry Clin Neurosci. 1994;6(1):1–13. PubMed CrossRef
- Rustad JK, Landsman HS, Ivkovic A, et al. Catatonia: an approach to diagnosis and treatment. Prim Care Companion CNS Disord. 2018;20(1):17. PubMed CrossRef
- Sawicka-Gutaj N, Zawalna N, Gut P, et al. Relationship between thyroid hormones and central nervous system metabolism in physiological and pathological conditions. Pharmacol Rep. 2022;74(5):847–858. PubMed CrossRef
- Pancotto T, Rossmeisl JH Jr, Panciera DL, et al. Blood-brain-barrier disruption in chronic canine hypothyroidism. Vet Clin Pathol. 2010;39(4):485–493. PubMed CrossRef
- Asnis GM. Catatonia secondary to hypothyroidism may be very responsive to electroconvulsive therapy: a case study. J ECT. 2020;36(4):e46–e47. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!