Age at Onset in Trichotillomania: Clinical Variables and Neurocognitive Performance

ABSTRACT
Objective: Trichotillomania (TTM), or compulsive hair pulling, is a common psychiatric disorder characterized by psychosocial impairment and reduced quality of life. The aim of this retrospective study was to characterize the impact of age at TTM onset on clinical variables and neuropsychological function using a variety of clinical and neurocognitive measures.
Method: The study sample included 98 adult treatment-seeking individuals with a DSM-IV diagnosis of TTM. Correlates were explored by grouping participants into childhood-onset (onset at ≤ 11 years old, n = 42) or later-onset (≥ 12 years old, n = 56) TTM and via linear regression. All subjects underwent a semistructured clinical interview with a psychiatrist and completed a variety of paper-pencil tests regarding TTM severity and quality of life. A subset (n = 44) of subjects underwent neurocognitive testing assessing motor inhibition and set-shifting compared to a sample (n = 27) of age- and gender-matched healthy controls. Data were collected from September 2006 through July 2011.
Results: Postpubertal age at onset was significantly associated with greater TTM symptom severity. Clinically, the later-onset group pulled their hair for a significantly greater amount of time daily (P = .008), had higher clinician-rated TTM severity on the Clinical Global Impressions-Severity of Illness scale (P = .042), and had higher patient-rated severity on the Massachusetts General Hospital Hairpulling Scale (P = .022) compared to healthy controls. On the neurocognitive tasks, later-onset TTM was characterized by stop-signal impairments (P = .020) and relatively spared set-shifting, consistent with previous studies in the literature. In contrast, the childhood-onset manifestation was associated with set-shifting deficits in stages of the task completed and total errors adjusted (both P < .001) but relatively spared stop-signal performance compared to healthy controls.
Conclusions: Results indicate that childhood-onset of TTM is common, as confirmed by the fact that 42.9% of our sample met childhood-onset criteria, and may differ neurobiologically from the prototypical later-onset form. Future neurobiological and treatment studies should measure age at onset and explore further these putative differences.
Trial Registration: ClinicalTrials.gov identifiers: NCT00354770 and NCT00775229
Prim Care Companion CNS Disord 2012;14(4):doi:10.4088/PCC.12m01343
© Copyright 2012 Physicians Postgraduate Press, Inc.
Submitted: January 12, 2012; accepted March 9, 2012.
Published online: July 19, 2012.
Corresponding author: Brian L. Odlaug, MPH, Department of Psychiatry, Ambulatory Research Center, University of Minnesota, 2450 Riverside Ave, Minneapolis, MN 55454 ([email protected]).
Trichotillomania (TTM) is a relatively common disorder, with an estimated prevalence of between 1% and 3.4% in the population, and is associated with social disruption, co-occurring depression and anxiety, and reduced quality of life.1 In recent years, there have been increasing efforts to explore the phenomenology and course of TTM and associated morbidity, such as quality of life and psychosocial functioning.2,3 Clinical correlates of age at onset, however, have only started to be studied in TTM.
Age at onset has been considered an important variable across many different disorders such as major depressive disorder,4 schizophrenia,5 and obsessive-compulsive disorder (OCD).6-14 In the case of OCD, which shares clinical, biological, and genetic overlap with TTM, childhood age at onset has been associated with greater severity of illness,7,10,12,14-17 a higher male preponderance,18 and higher prevalence of psychiatric comorbidity.4,6,8,9,11,19,20 In pathological skin picking, a disorder with phenomenologic and likely neurobiological overlap with TTM, childhood age at onset has been associated with lower rates of treatment-seeking behavior, longer duration of illness before seeking treatment, and less conscious awareness surrounding the habit.21 Such findings have clinical implications in that age at onset could be indicative of severity and comorbidity in general and could potentially lead to different therapeutic interventions and expectations for the course of illness.
Despite a traditional body of literature highlighting childhood-onset versus later-onset TTM as candidate subtypes of TTM, survey data have failed thus far to identify clinically significant differences between childhood and later forms.22,23 We sought to expand considerably upon this corpus of literature by investigating potential associations between not only clinical variables and age at onset, but also neuropsychological function and age at onset. On the basis of the above findings, including in OCD, we hypothesized that childhood age at onset of TTM would be associated with higher severity of illness, a lower quality of life, higher rates of comorbid psychiatric disorders, and more profound cognitive dysfunction.
METHOD
Subjects
Data from a total of 98 subjects taking part in pharmacotherapy trials for the treatment of TTM were pooled together for the purposes of this study. Inclusion criteria included male and female outpatients between the ages of 18 and 65 years with a primary DSM-IV diagnosis of TTM. Exclusion criteria included current DSM-IV psychotic disorders, bipolar disorder, or substance use disorders; currently receiving treatment (ie, counseling or pharmacotherapy specifically for TTM); and an inability to understand study procedures and provide written informed consent. All measures of assessment were taken at baseline prior to the implementation of any treatments. Data were collected from September 2006 through July 2011.
There is considerable discrepancy in how childhood age at onset of TTM is determined across studies.7,24 In this study, we chose a cutoff point of under 12 years of age for childhood TTM onset and age 12 years and older for adolescent TTM onset on the basis of data collected from 1930 to 1994 showing that the mean age of puberty for females (the majority of our sample were women) in the United States is 12 to 13 years of age.25 The terminology of childhood and preadolescent is based on the Centers for Disease Control and Prevention’s 26 definitions, in which age 9 to 11 years is classified as childhood and 12 to 14 years is classified as preadolescent. Age at onset was quantified in the current study by asking subjects, “At what age did you start pulling out hair for noncosmetic reasons?”
Clinical Assessments
Psychiatric comorbidity was assessed using the Structured Clinical Interview for DSM-IV.27 We also asked about duration of illness, if the subject had sought past treatment for TTM, and which sites were pulled.

- Clinicians should be aware of the severity associated with trichotillomania at all ages, regardless of age at onset.
- Subjects with a later age at onset spent more time pulling each day and had a more clinically severe form of trichotillomania than the childhood-onset group.
- Consistent with previous studies, neurocognitive testing revealed impaired stop-signal reaction times in the later-onset group versus healthy controls.
Massachusetts General Hospital Hairpulling Scale (MGH-HPS).28,29 The MGH-HPS is a valid and reliable, 7-item, self-report scale that rates urges to pull hair, actual amount of pulling, perceived control over behavior, and distress associated with hair pulling over the preceding 7 days.
Clinical Global Impressions-Severity of Illness scale (CGI-S).30 The CGI-S is a valid and reliable, 7-item scale used to assess symptom severity. The CGI-S uses a Likert-scored scale with 1 = “not ill at all” to 7 = “among the most extremely ill.”
Sheehan Disability Scale.31 The Sheehan Disability Scale is a valid and reliable, 3-item, self-report scale that assesses psychosocial functioning in 3 areas of life: work, social or leisure activities, and home and family life. Scores on the scale range from 0 to 30.
Quality of Life Inventory.32 The Quality of Life Inventory is a valid and reliable 16-item, self-report positive psychology scale that assesses areas of life such as health, love, work, recreation, home, friendships, self-esteem, and standard of living. All subjects, including the healthy controls, completed the scale.
Hamilton Anxiety Rating Scale.33 The Hamilton Anxiety Rating Scale is a valid and reliable clinician-administered, 14-item scale that provides an overall measure of global anxiety.
Hamilton Depression Rating Scale.34 The Hamilton Depression Rating Scale is a valid and reliable 17-item, clinician-administered rating scale that assesses severity of depressive symptoms.
Family history of psychiatric conditions, including substance use/abuse and grooming disorders, was obtained from subjects. Family history data were collected from interviews with the proband.
Cognitive Assessments
Cognitive data from a subsample (n = 44) of TTM participants were available, which were collected as part of the clinical trials concerned. The studies were registered on ClinicalTrials.gov (identifiers: NCT00354770 and NCT00775229). All cognitive data were collected at baseline (clinical trial entry) and comprised a test of motor inhibition (Stop-Signal Task) and of cognitive flexibility (Intradimensional/Extradimensional Set Shift Task).35 We also collected data from age-matched and gender-matched healthy controls (n = 27) with no lifetime or current psychiatric history.
The Stop-Signal Task quantified the participants’ ability to suppress already-initiated motor responses.36,37 The task is dependent on neural circuitry, including the right inferior frontal gyrus and bilateral anterior cingulate cortices, and has previously been reported to be impaired in people with TTM38 and in related disorders of impulse dyscontrol.39
The Intradimensional/Extradimensional Set Shift Task is dependent on neural circuitry including the ventromedial and ventrolateral prefrontal cortices and is based on the Wisconsin Card Sorting Test.40 Intradimensional shifting involves keeping one’s attention on the same previously relevant stimulus dimension, while extradimensional shifting involves cognitive flexibility or shifting attention away from a previously relevant dimension toward a dimension that was previously irrelevant. Set-shifting was reported to be intact in previous TTM research but is often impaired in OCD.38,41
Data Analysis
Between-group differences on demographic and clinical variables were tested using 1-way analysis of variance, t test, or χ2 test, with follow-up post hoc tests as appropriate (protected least significant difference tests or further χ2 test as appropriate). An α level of .05 was used for statistical significance. Effect size using Cohen d and partial-eta squared was reported for significant differences at the .05 α level. Linear regression was used to examine the relationship between severity of symptoms as measured by the MGH-HPS and various demographic and clinical variables at baseline.
In order to examine our primary independent variable, age at onset, in relation to clinical severity, a total of 3 models were computed. The first model examines the relationship between age at onset and scores on the MGH-HPS. Demographics (age, gender, race, marital status, education, and work status) were added to the second model of the regression in order to examine their impact on MGH-HPS scores in relation to age at onset. In the final model, clinical characteristics measured at baseline were added. Nagelkerke r-squared adjusted42 was calculated for each model to determine the variance in MGH-HPS scores accounted for by the various models. We also examined duration of illness as a potential factor of illness severity using linear regression. Analysis was conducted using IBM PASW Statistics, Version 18 software (IBM SPSS, Armonk, New York).
RESULTS
A total of 98 individuals (mean ± SD age of 34.3 ± 11.1 years, n = 85, 86.7% female) meeting current DSM-IV criteria for TTM were included in the sample. The majority of the sample were identified as white (n = 94, 95.9%) and had a college degree or more (n = 67, 68.4%). A total of 47 (47.9%) were single, 44 (44.9%) were married, and 7 (7.1%) were divorced, separated, or widowed. The majority of subjects reported being employed full-time (n = 72, 73.5%), while 26 (26.5%) reported being unemployed, retired, or a full-time student.
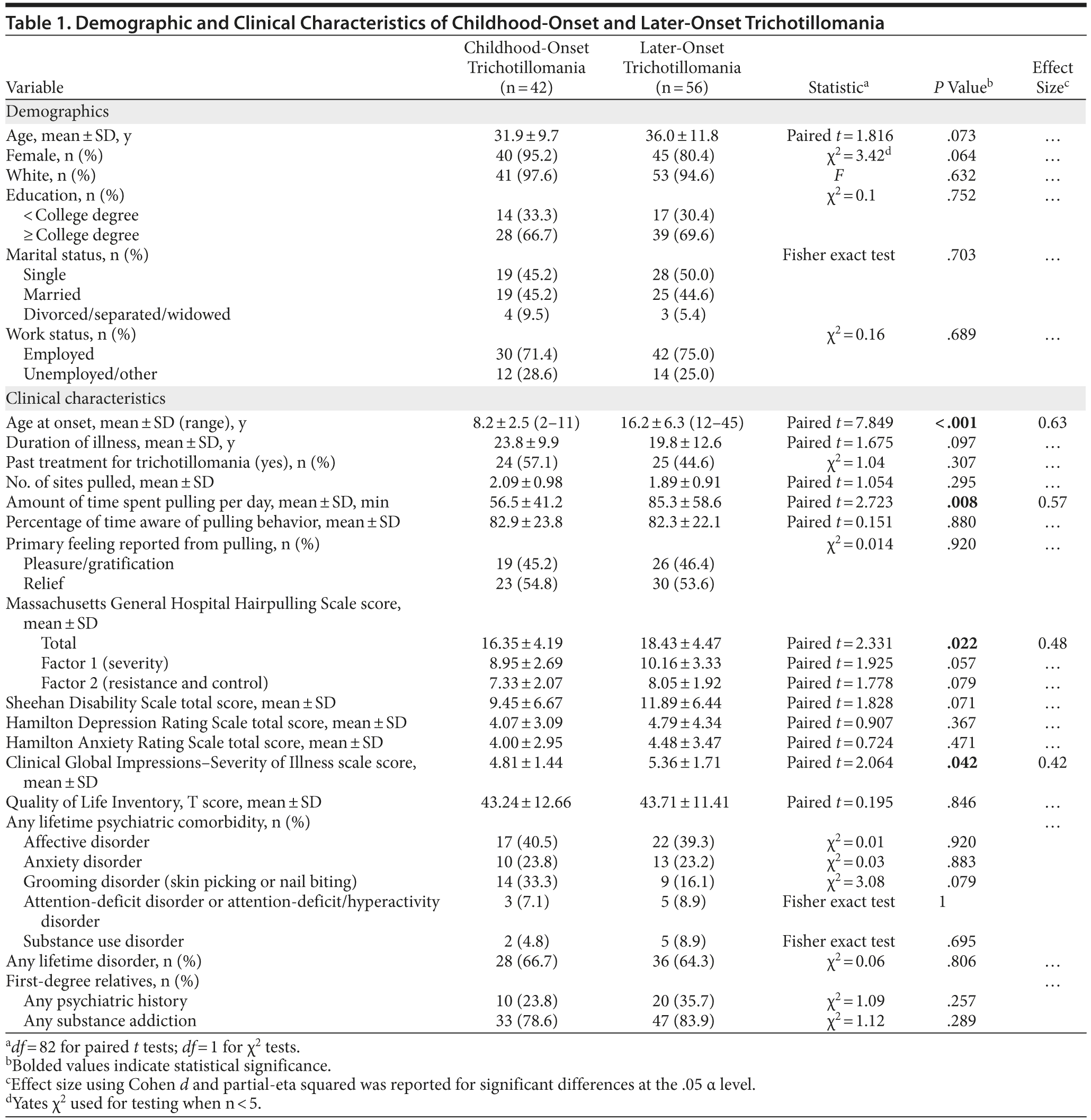
A total of 42 individuals with prepubescent age at onset (mean ± SD age of 31.9 ± 9.7 years, with TTM onset at 8.2 ± 2.5 years; n = 40; 95.2% female) were compared to 56 later-onset (mean ± SD age of 36.0 ± 11.8 years, with TTM onset at 16.2 ± 6.3 years; n = 45; 80.4% female) subjects (Table 1). The childhood-onset group, compared to the later-onset group, spent significantly less time pulling per day (mean ± SD = 56.5 ± 41.2 vs 85.3 ± 58.6 minutes, respectively, P = .008) and had a less severe presentation of illness as illustrated by scores on the MGH-HPS (mean ± SD score of 16.35 ± 4.2 vs 18.43 ± 4.5, respectively, P = .022) and CGI-S (mean ± SD score of 4.81 ± 1.44 vs 5.36 ± 1.71, respectively, P = .042) scales.
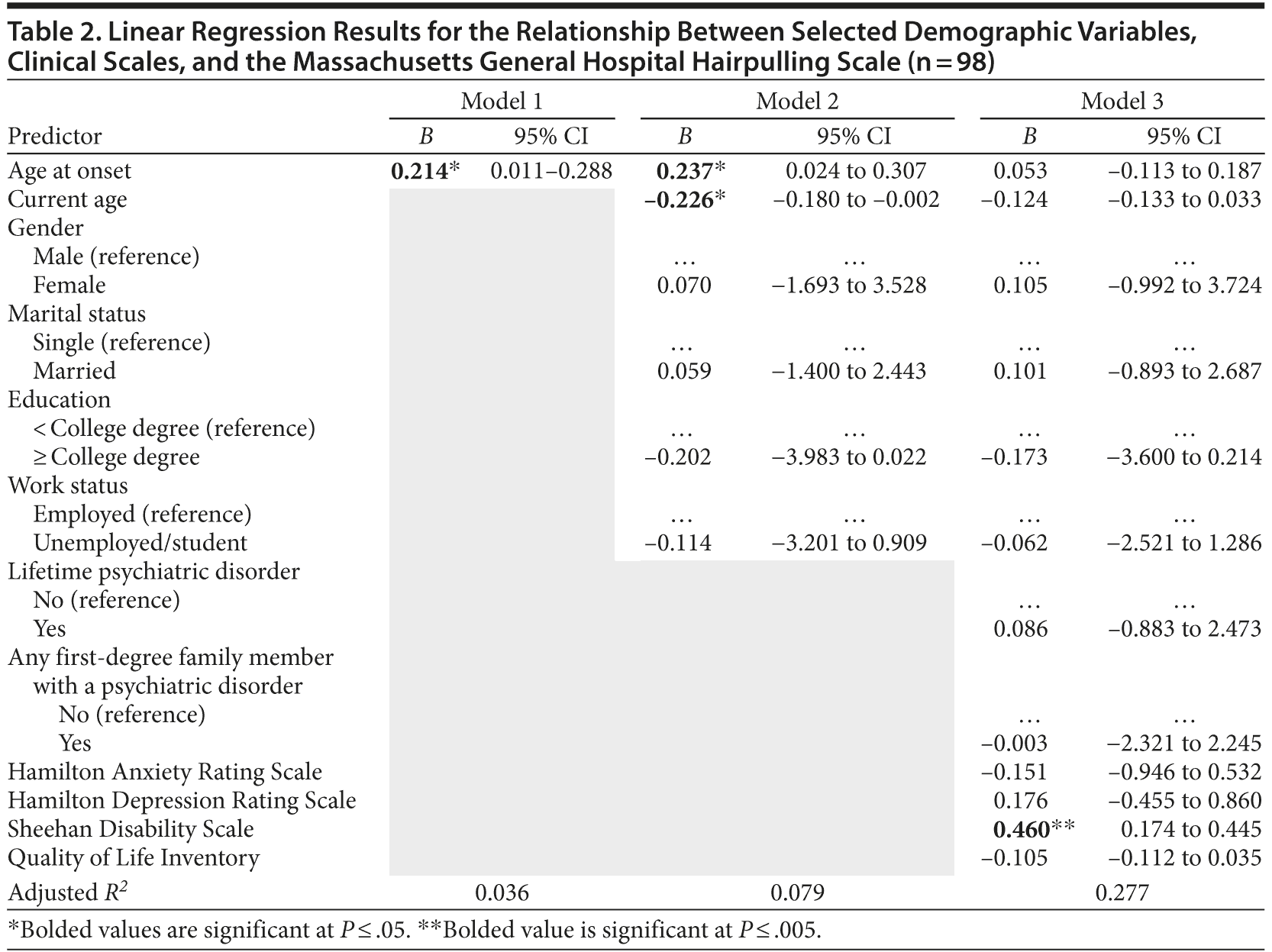
Results of the linear regression between clinical severity as operationalized by the MGH-HPS and various demographic and clinical characteristics can be found in Table 2. Age at onset accounted for only 3.6% of the variance in overall MGH-HPS scores, a nonclinically significant variable. In model 2, current age (B = -0.226, 95% CI = −0.180 to −0.002) and age at onset (B = 0.237, 95% CI = 0.024 to 0.307) were clinically significant and accounted for 7.9% of the variance in MGH-HPS scores. Clinical scales were incorporated into model 3, with the Sheehan Disability Scale significantly correlating to the MGH-HPS scores (B = 0.460, 95% CI = 0.174 to 0.455). Overall, 27.7% of the variance in MGH-HPS scores was accounted for through model 3.
Duration of illness was also examined as a potential factor in clinical severity. Linear regression results indicated that 7.4% of the variance in total scores on the MGH-HPS was accounted for by this variable.
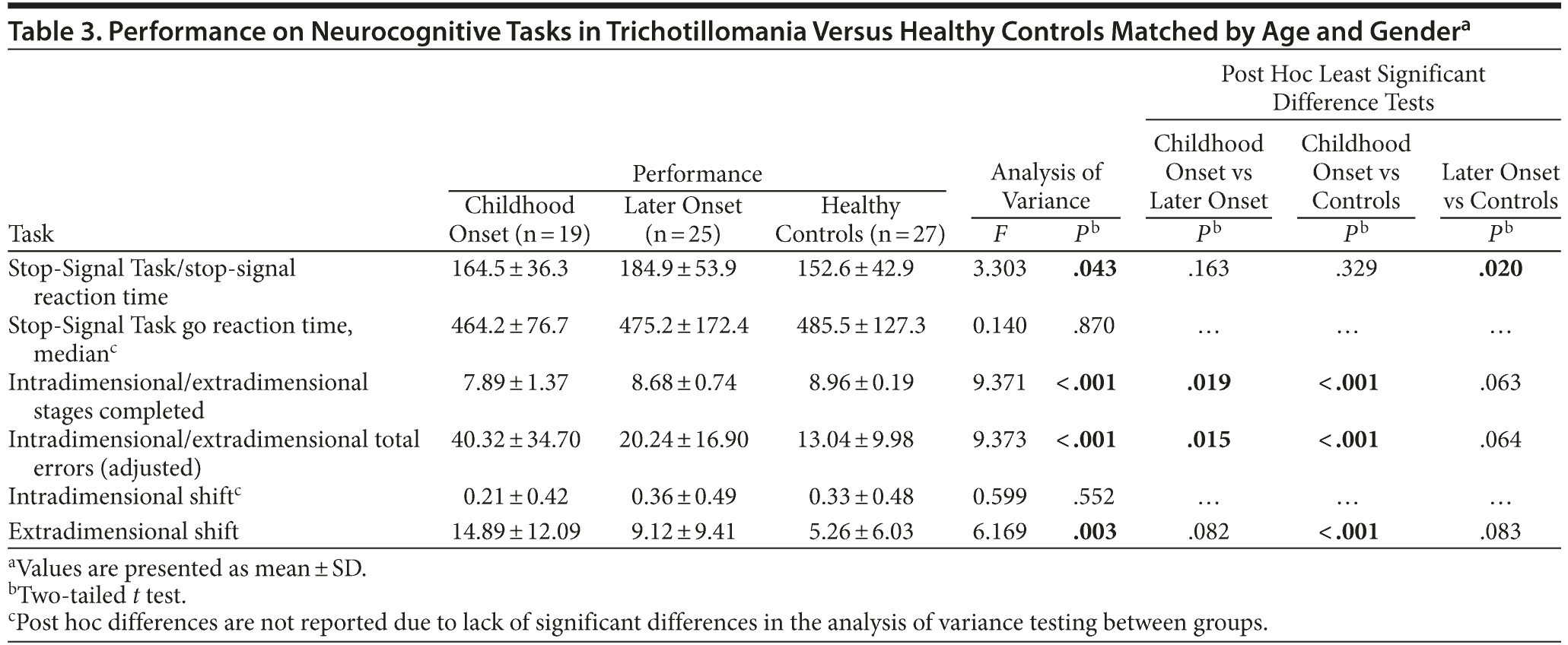
Neurocognitive testing revealed impaired stop-signal reaction times in the older-onset group versus healthy controls (P = .020), while the childhood-onset TTM group showed significantly impaired extradimensional set-shifting versus the controls (P < .001) (Table 3).
DISCUSSION
In this study, we examined age at onset of TTM and clinical and cognitive correlates of childhood-onset and later-onset illness in 98 individuals with a current DSM-IV diagnosis of TTM. To our knowledge, this is the first report of more detailed information about age at onset and the first to examine cognitive correlates of age at onset in TTM.
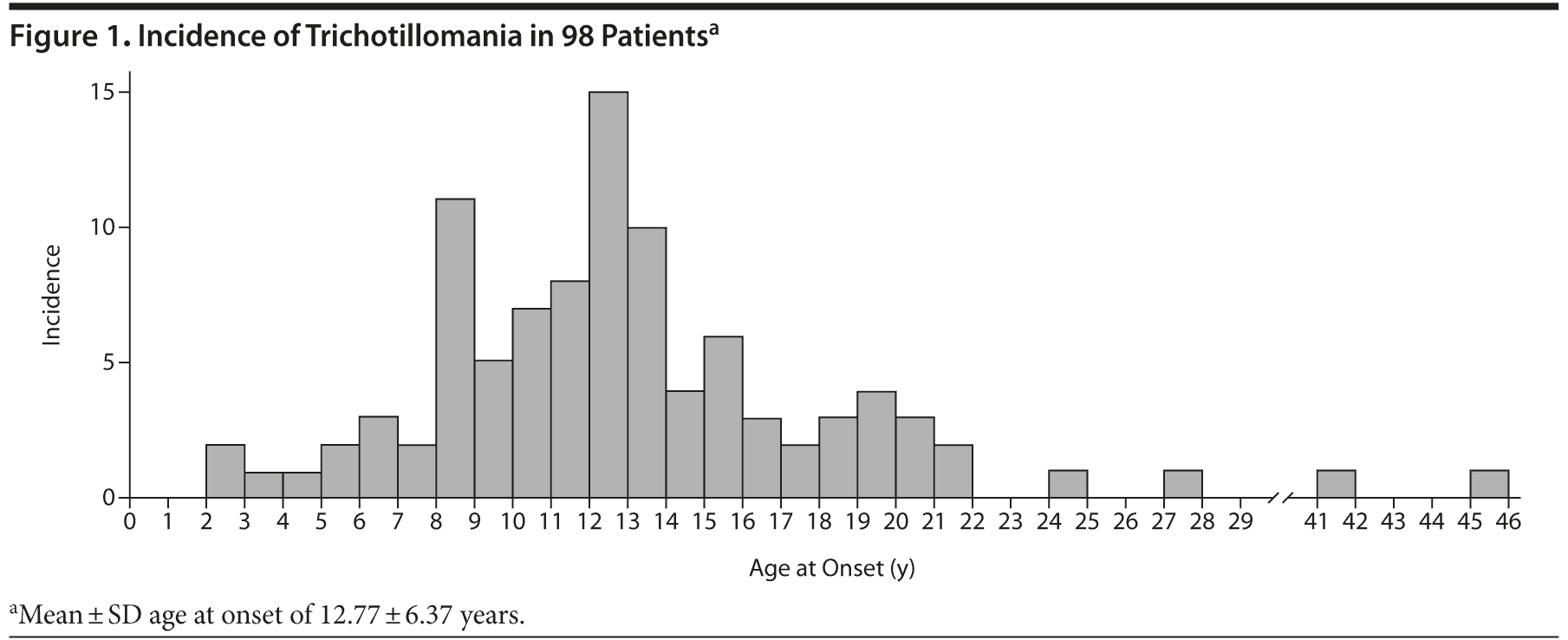
It is notable that over one-third (42.9%) of TTM subjects had illness onset during childhood. Previous studies have reported a mean age at TTM onset of 13.1 years,43 which is consistent with the overall mean age at onset in our sample (Figure 1). The literature suggests, albeit with limited empirical data, that early childhood-onset TTM (“baby trich”) is typically of short duration with spontaneous resolution.44,45 We did identify some individuals with the early childhood-onset form who were still experiencing TTM as adults (Figure 1), suggesting that resolution is not ubiquitous in such individuals.
Contrary to our hypotheses that childhood onset of TTM would be associated with more severe TTM symptoms as well as poorer psychosocial functioning and quality of life, this study found that TTM subjects with childhood-onset and early adolescent-onset illness were generally similar with respect to these aspects and showed significantly lower disease severity scores (MGH-HPS, CGI-S). Age at onset only accounted for approximately 3.6% of the variance in clinical severity scores, and although significant, illustrates the global impact of TTM regardless of when onset occurs. These data also highlight that severity and disability, while related conceptually, can be dissociated to some degree. It may also be that the differences in disease severity scores were not large enough to subtend significant functional differences on day-to-day life. Alternatively, the childhood-onset group, having started pulling at a very young age and having a longer duration of illness, may have developed coping mechanisms for their pulling urges and behaviors and consequently present with a clinically less severe self-reported form of pulling.
Our results indicating that 7.4% of the variance in MGH-HPS scores was accounted for by duration of illness illustrates the need for future research examining TTM along a continuum. Furthermore, in our second regression model, which included various demographic characteristics, we found that age was the only significant demographic factor influencing MGH-HPS scores. The findings here are somewhat discordant with OCD research, which has found childhood age at onset to be associated with greater OCD symptom severity rather than vice versa.7,10,12,14-16 It may be that a greater proportion of childhood-onset OCD remits as compared to childhood-onset TTM, leading to a predominance of more “extreme/severe” forms in adult studies of the former. Another possible explanation for the discrepancies between this study and the OCD literature may be due to the variability in defining age at onset and childhood onset. Age at onset has been variously defined as the age when symptoms begin,7,46 the age at which the person displays significant distress or impairment,16 or the age when the person first meets DSM-IV criteria.9 In our study, we used when symptoms began to classify our subjects. Without consistent definitions regarding age at onset phenotypes, comparisons between research studies of childhood and later onset of illness are hindered.
The issue of cognitive dysfunction in TTM is important since knowledge of this area may help to model neurobiological dysfunction and differences between putative subtypes by using endophenotypic markers. In the current study, we found that later-onset TTM was associated with significantly impaired stop-signal inhibitory control in comparison to healthy controls but intact cognitive flexibility on the set-shift task. These findings are consistent with the existing literature using these tasks in TTM.38 It was interesting to note that, in contrast, childhood-onset TTM was associated with significantly elevated errors on the set-shifting task compared to both the healthy controls and the later-onset group. Further exploration of individual task stages suggested that these flexibility deficits in childhood-onset TTM were particularly pronounced at the extradimensional shift stage, a finding commonly reported in OCD.38 While caution is warranted when making inferences regarding neurobiological dysfunction based solely on cognitive tests, these data may implicate dorsolateral prefrontal dysfunction in early-onset TTM, with more inferiofrontal dysfunction in later-onset TTM.47 These novel cognitive findings suggest that the application of structural and functional magnetic resonance imaging in childhood-onset versus later-onset TTM would be valuable. They also may have treatment implications, in that response inhibition and extradimensional set-shifting are thought to be subserved by distinct neural and neurochemical mediators.47,48
This study has several limitations. First, the small sample size may limit generalizability to non-treatment seeking TTM patients in the population. Second, some research indicates that early childhood age at onset (ie, ≤ 5 years old) is rare and often resolves without sequelae.44 While we identified several such individuals with persisting symptoms as adults, further research should explore the possible heterogeneity of TTM as a function of age at onset. Furthermore, onset of TTM was retrospectively assigned in this study on the basis of subjects’ recollections. Although the interviewers took great care to ascertain age at onset as accurately as possible, it can be difficult to remember the details of an event that occurred, on average, decades earlier, and some of our subjects may not have been good historians. Thus, the reported ages at onset found in this study may not accurately reflect the true age at onset of symptoms. Finally, first-degree family history of psychiatric conditions was obtained via a semistructured interview with the proband only. There is currently a paucity of information on family history in the development of TTM. Given the shame and embarrassment often seen in mental illness, however, it is possible that subjects in this study were unable to provide accurate accounts of psychiatric family history. Future studies should conduct formal interviews with family members to examine clinical correlates of familial psychiatric illness. Strengths of the study include examination of a range of cognitive and clinically important variables and use of both self-report and interviewer-administered measures with strong psychometric properties and established norms.
There are several clinical implications of this study. The primary implication is that clinicians should be aware that TTM often begins during childhood. Childhood screening and intervention efforts targeting children with TTM are needed. For clinicians who work with adults, it is important to be aware that this disorder has likely been present since childhood or adolescence and also that possible negative effects on attainment of developmental transitions and tasks may be an important part of the case conceptualization and treatment plan.
Although childhood age at onset was not associated with severity of TTM symptoms on most measures or with significantly poorer psychosocial functioning or quality of life, the disorder may have a critical impact on other areas of development that were not assessed in this study. Developmental transitions and tasks during childhood and adolescence include affiliation transitions (greater autonomy from the family and peer affiliations, development of romantic affiliations), achievement transitions (school and work), and identity transitions (changes in self-definition). Severe psychopathology may adversely affect key developmental transitions. Such failures may have adverse repercussions throughout life. Future studies of age at onset in TTM, an often severe disorder, should examine these important constructs. We also wish to underscore the importance of pediatric screening for TTM since the prevalence of childhood-onset TTM was relatively high (42.9%) in our sample and given the fact that these individuals experienced a chronic course of illness. Finally, we strongly recommend that future studies in TTM record age at onset, since the novel cognitive data reported here hint at differential neural dysfunction between childhood-onset and later-onset forms of the condition.
Author affiliations: Department of Psychiatry, Ambulatory Research Center, University of Minnesota, Minneapolis (Dr Grant and Messrs Odlaug and Harvanko); and Department of Psychiatry, University of Cambridge, Cambridge, United Kingdom (Dr Chamberlain).
Potential conflicts of interest: Dr Grant has received grant/research support from Forest, Psyadon, and the University of South Florida and serves as the Editor in Chief of the Journal of Gambling Studies. Mr Odlaug has received honoraria from Oxford University Press and Current Medicine Group, LLC. Dr Chamberlain has served as a consultant for Cambridge Cognition, Eli Lilly, P1Vital, and Shire. Mr Harvanko reports no conflicts of interest related to the subject of this study.
Funding/support: This research is supported by a Center for Excellence in Gambling Research grant from the National Center for Responsible Gaming and an American Recovery and Reinvestment Act grant from the National Institute on Drug Abuse (1RC1DA028279-01) to Dr Grant.
REFERENCES
1. Christenson GA, Mackenzie TB, Mitchell JE. Characteristics of 60 adult chronic hair pullers. Am J Psychiatry. 1991;148(3):365-370. PubMed
2. Woods DW, Flessner CA, Franklin ME, et al; Trichotillomania Learning Center-Scientific Advisory Board. The Trichotillomania Impact Project (TIP): exploring phenomenology, functional impairment, and treatment utilization. J Clin Psychiatry. 2006;67(12):1877-1888. PubMed doi:10.4088/JCP.v67n1207
3. Odlaug BL, Kim SW, Grant JE. Quality of life and clinical severity in pathological skin picking and trichotillomania. J Anxiety Disord. 2010;24(8):823-829. PubMed doi:10.1016/j.janxdis.2010.06.004
4. Zisook S, Lesser I, Stewart JW, et al. Effect of age at onset on the course of major depressive disorder. Am J Psychiatry. 2007;164(10):1539-1546. PubMed doi:10.1176/appi.ajp.2007.06101757
5. Rajji TK, Ismail Z, Mulsant BH. Age at onset and cognition in schizophrenia: meta-analysis. Br J Psychiatry. 2009;195(4):286-293. PubMed doi:10.1192/bjp.bp.108.060723
6. Geller DA, Biederman J, Faraone SV, et al. Disentangling chronological age from age of onset in children and adolescents with obsessive-compulsive disorder. Int J Neuropsychopharmacol. 2001;4(2):169-178. PubMed doi:10.1017/S1461145701002395
7. Rosario-Campos MC, Leckman JF, Mercadante MT, et al. Adults with early-onset obsessive-compulsive disorder. Am J Psychiatry. 2001;158(11):1899-1903. PubMed doi:10.1176/appi.ajp.158.11.1899
8. Hemmings SM, Kinnear CJ, Lochner C, et al. Early- versus late-onset obsessive-compulsive disorder: investigating genetic and clinical correlates. Psychiatry Res. 2004;128(2):175-182. PubMed doi:10.1016/j.psychres.2004.05.007
9. Chabane N, Delorme R, Millet B, et al. Early-onset obsessive-compulsive disorder: a subgroup with a specific clinical and familial pattern? J Child Psychol Psychiatry. 2005;46(8):881-887. PubMed doi:10.1111/j.1469-7610.2004.00382.x
10. T×¼kel R, Ertekin E, Batmaz S, et al. Influence of age of onset on clinical features in obsessive-compulsive disorder. Depress Anxiety. 2005;21(3):112-117. PubMed doi:10.1002/da.20065
11. Pinto A, Mancebo MC, Eisen JL, et al. The Brown Longitudinal Obsessive Compulsive Study: clinical features and symptoms of the sample at intake. J Clin Psychiatry. 2006;67(5):703-711. PubMed doi:10.4088/JCP.v67n0503
12. Grant JE, Mancebo MC, Pinto A, et al. Late-onset obsessive compulsive disorder: clinical characteristics and psychiatric comorbidity. Psychiatry Res. 2007;152(1):21-27. PubMed doi:10.1016/j.psychres.2006.09.015
13. Garcia AM, Freeman JB, Himle MB, et al. Phenomenology of early childhood onset obsessive compulsive disorder. J Psychopathol Behav Assess. 2009;31(2):104-111. PubMed doi:10.1007/s10862-008-9094-0
14. Lomax CL, Oldfield VB, Salkovskis PM. Clinical and treatment comparisons between adults with early- and late-onset obsessive-compulsive disorder. Behav Res Ther. 2009;47(2):99-104. PubMed doi:10.1016/j.brat.2008.10.015
15. Sobin C, Blundell ML, Karayiorgou M. Phenotypic differences in early- and late-onset obsessive-compulsive disorder. Compr Psychiatry. 2000;41(5):373-379. PubMed doi:10.1053/comp.2000.9009
16. Fontenelle LF, do Rosסrio-Campos MC, Mendlowicz MV, et al. Treatment-response by age at onset in obsessive-compulsive disorder. J Affect Disord. 2004;83(2-3):283-284. PubMed doi:10.1016/j.jad.2004.07.007
17. Millet B, Kochman F, Gallarda T, et al. Phenomenological and comorbid features associated in obsessive-compulsive disorder: influence of age of onset. J Affect Disord. 2004;79(1-3):241-246. PubMed doi:10.1016/S0165-0327(02)00351-8
18. Swedo SE, Rapoport JL, Leonard H, et al. Obsessive-compulsive disorder in children and adolescents: clinical phenomenology of 70 consecutive cases. Arch Gen Psychiatry. 1989;46(4):335-341. PubMed doi:10.1001/archpsyc.1989.01810040041007
19. Segu× J, Mסrquez M, Garc×a L, et al. Differential clinical features of early-onset panic disorder. J Affect Disord. 1999;54(1-2):109-117. PubMed doi:10.1016/S0165-0327(98)00148-7
20. de Mathis MA, do Rosario MC, Diniz JB, et al. Obsessive-compulsive disorder: influence of age at onset on comorbidity patterns. Eur Psychiatry. 2008;23(3):187-194. PubMed doi:10.1016/j.eurpsy.2008.01.002
21. Odlaug BL, Grant JE. Childhood-onset pathologic skin picking: clinical characteristics and psychiatric comorbidity. Compr Psychiatry. 2007;48(4):388-393. PubMed doi:10.1016/j.comppsych.2007.03.007
22. Flessner CA, Lochner C, Stein DJ, et al. Age of onset of trichotillomania symptoms: investigating clinical correlates. J Nerv Ment Dis. 2010;198(12):896-900. PubMed doi:10.1097/NMD.0b013e3181fe7423
23. Lochner C, Seedat S, Stein DJ. Chronic hair-pulling: phenomenology-based subtypes. J Anxiety Disord. 2010;24(2):196-202. PubMed doi:10.1016/j.janxdis.2009.10.008
24. Cohen LJ, Stein DJ, Simeon D, et al. Clinical profile, comorbidity, and treatment history in 123 hair pullers: a survey study. J Clin Psychiatry. 1995;56(7):319-326. PubMed
25. Walvoord EC. The timing of puberty: is it changing? does it matter? J Adolesc Health. 2010;47(5):433-439. PubMed doi:10.1016/j.jadohealth.2010.05.018
26. Centers for Disease Control and Prevention. Child development: positive parenting tips. http://www.cdc.gov/ncbddd/childdevelopment/positiveparenting/index.html. Accessed April 27, 2012.
27. First MB, Spitzer RL, Gibbon M, et al. Structured Clinical Interview for DSM-IV-Patient Edition (SCID-I/P, Version 2.0). New York, NY: Biometrics Research Department, New York State Psychiatric Institute; 1995.
28. Keuthen NJ, O’ Sullivan RL, Ricciardi JN, et al. The Massachusetts General Hospital (MGH) Hairpulling Scale, 1: development and factor analyses. Psychother Psychosom. 1995;64(3-4):141-145. PubMed doi:10.1159/000289003
29. Keuthen NJ, Flessner CA, Woods DW, et al; Trichotillomania Learning Center Scientific Advisory Board. Factor analysis of the Massachusetts General Hospital Hairpulling Scale. J Psychosom Res. 2007;62(6):707-709. PubMed doi:10.1016/j.jpsychores.2006.12.003
30. Guy W. ECDEU Assessment Manual for Psychopharmacology. US Dept Health, Education and Welfare publication (ADM) 76-338. Rockville, MD: National Institute of Mental Health; 1976:218-222.
31. Sheehan DV. The Anxiety Disease. New York, NY: Scribner’s ; 1983.
32. Frisch MB. The Quality of Life Inventory: a cognitive-behavioral tool for complete problem assessment, treatment planning, and outcome evaluation. Behav Ther. 1993;16:42-44.
33. Hamilton M. The assessment of anxiety states by rating. Br J Med Psychol. 1959;32(1):50-55. PubMed doi:10.1111/j.2044-8341.1959.tb00467.x
34. Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23(1):56-62. PubMed doi:10.1136/jnnp.23.1.56
35. Cambridge Cognition Limited. CANTABeclipse, version 3. Cambridge, UK: Cambridge Cognition Limited; 2007.
36. Logan GD, Cowan WB, Davis KA. On the ability to inhibit simple and choice reaction time responses: a model and a method. J Exp Psychol Hum Percept Perform. 1984;10(2):276-291. PubMed doi:10.1037/0096-1523.10.2.276
37. Aron AR, Robbins TW, Poldrack RA. Inhibition and the right inferior frontal cortex. Trends Cogn Sci. 2004;8(4):170-177. PubMed doi:10.1016/j.tics.2004.02.010
38. Chamberlain SR, Fineberg NA, Blackwell AD, et al. Motor inhibition and cognitive flexibility in obsessive-compulsive disorder and trichotillomania. Am J Psychiatry. 2006;163(7):1282-1284. PubMed doi:10.1176/appi.ajp.163.7.1282
39. Odlaug BL, Chamberlain SR, Grant JE. Motor inhibition and cognitive flexibility in pathologic skin picking. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34(1):208-211. PubMed doi:10.1016/j.pnpbp.2009.11.008
40. Lezak MD, Howieson DB, Loring DW. Neuropsychological Assessment. New York, NY: Oxford University Press; 2004.
41. Grant JE, Odlaug BL, Chamberlain SR. A cognitive comparison of pathological skin picking and trichotillomania. J Psychiatr Res. 2011;45(12):1634-1638. PubMed doi:10.1016/j.jpsychires.2011.07.012
42. Nagelkerke NJD. A note on a general definition of the coefficient of determination. Biometrika. 1991;78(3):691-692. doi:10.1093/biomet/78.3.691
43. Christenson GA, Mansueto CS. Trichotillomania: descriptive characteristics and phenomenology. In: Stein DJ, Christenson G, Hollander E, eds. Trichotillomania. Washington, DC: American Psychiatric Publishing; 1999.
44. Sah DE, Koo J, Price VH. Trichotillomania. Dermatol Ther. 2008;21(1):13-21. PubMed doi:10.1111/j.1529-8019.2008.00165.x
45. Swedo SE, Leonard HL. Trichotillomania: an obsessive compulsive spectrum disorder? Psychiatr Clin North Am. 1992;15(4):777-790. PubMed
46. Delorme R, Golmard JL, Chabane N, et al. Admixture analysis of age at onset in obsessive-compulsive disorder. Psychol Med. 2005;35(2):237-243. PubMed doi:10.1017/S0033291704003253
47. Hampshire A, Chamberlain SR, Monti MM, et al. The role of the right inferior frontal gyrus: inhibition and attentional control. Neuroimage. 2010;50(3):1313-1319. PubMed doi:10.1016/j.neuroimage.2009.12.109
48. Chamberlain SR, Sahakian BJ. The neuropsychiatry of impulsivity. Curr Opin Psychiatry. 2007;20(3):255-261. PubMed