Objective: Positive psychological attributes (eg, optimism) have been associated with a healthier lifestyle and superior medical outcomes in patients with type 2 diabetes; however, there has been minimal study of behavioral interventions that target positive psychological constructs in this population. Accordingly, we developed a novel, telephone-based, 12-week positive psychology intervention and assessed its feasibility and short-term impact in adults with type 2 diabetes and suboptimal health behavior adherence.
Method: This was a pilot and feasibility study in adult inpatients and outpatients at an urban academic medical center recruited between December 2013 and December 2014. Adult patients with (1) type 2 diabetes (meeting American Diabetes Association criteria, eg, glycated hemoglobin A1c [HbA1c] > 6.5% or fasting glucose > 126 mg/dL) and (2) suboptimal adherence (score < 15/18 on the Medical Outcomes Study Specific Adherence Scale items for medication, diet, and exercise) were eligible. Participants received a positive psychology manual, completed exercises (eg, writing a gratitude letter, performing acts of kindness), and reviewed these activities by phone with a study trainer over the 12-week study period. Feasibility and acceptability were assessed via exercise completion rates and postexercise ratings of ease/utility on 0-10 Likert scales. Longer-term efficacy was explored by examining changes in psychological states and health behaviors from baseline to 12 weeks using random-effects regression models and estimates of effect size.
Results: A total of 15 participants enrolled; 12 participants provided complete baseline and follow-up data and were included in the analyses. Over 90% of these participants completed at least 2 exercises, and 75% completed a majority of the exercises. Participants rated the exercises as helpful (mean = 7.8/10) and easy to complete (mean = 7.1/10), and they reported improvements in optimism, gratitude, depression, anxiety, physical function, self-care, and health behaviors (Cohen d = 0.28-1.00).
Conclusion: A positive psychology intervention for suboptimally adherent patients with type 2 diabetes was feasible, acceptable, and associated with broad pre-post psychological and health behavior improvement in a small initial study. Further testing of this promising intervention is warranted.
Trial Registration: ClinicalTrials.gov identifier: NCT02736084
Feasibility and Acceptability of a Positive Psychological Intervention for Patients With Type 2 Diabetes
ABSTRACT
Objective: Positive psychological attributes (eg, optimism) have been associated with a healthier lifestyle and superior medical outcomes in patients with type 2 diabetes; however, there has been minimal study of behavioral interventions that target positive psychological constructs in this population. Accordingly, we developed a novel, telephone-based, 12-week positive psychology intervention and assessed its feasibility and short-term impact in adults with type 2 diabetes and suboptimal health behavior adherence.
Method: This was a pilot and feasibility study in adult inpatients and outpatients at an urban academic medical center recruited between December 2013 and December 2014. Adult patients with (1) type 2 diabetes (meeting American Diabetes Association criteria, eg, glycated hemoglobin A1c [HbA1c] > 6.5% or fasting glucose > 126 mg/dL) and (2) suboptimal adherence (score < 15/18 on the Medical Outcomes Study Specific Adherence Scale items for medication, diet, and exercise) were eligible. Participants received a positive psychology manual, completed exercises (eg, writing a gratitude letter, performing acts of kindness), and reviewed these activities by phone with a study trainer over the 12-week study period. Feasibility and acceptability were assessed via exercise completion rates and postexercise ratings of ease/utility on 0–10 Likert scales. Longer-term efficacy was explored by examining changes in psychological states and health behaviors from baseline to 12 weeks using random-effects regression models and estimates of effect size.
Results: A total of 15 participants enrolled; 12 participants provided complete baseline and follow-up data and were included in the analyses. Over 90% of these participants completed at least 2 exercises, and 75% completed a majority of the exercises. Participants rated the exercises as helpful (mean = 7.8/10) and easy to complete (mean = 7.1/10), and they reported improvements in optimism, gratitude, depression, anxiety, physical function, self-care, and health behaviors (Cohen d = 0.28–1.00).
Conclusion: A positive psychology intervention for suboptimally adherent patients with type 2 diabetes was feasible, acceptable, and associated with broad pre-post psychological and health behavior improvement in a small initial study. Further testing of this promising intervention is warranted.
Trial Registration: ClinicalTrials.gov identifier: NCT02736084
Prim Care Companion CNS Disord 2016;18(3):doi:10.4088/PCC.15m01902
© Copyright 2016 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, Massachusetts General Hospital, Boston
bHarvard Medical School, Boston, Massachusetts
cMGH Diabetes Center, Department of Medicine, Massachusetts General Hospital, Boston
*Corresponding author: Jeff C. Huffman, MD, Massachusetts General Hospital, 55 Fruit St/Blake 11, Boston, MA 02114 ([email protected]).
Type 2 diabetes affects 11% of US adults1 and is associated with impaired functioning, serious medical complications,2 and high rates of mortality.3 Comprehensive evidence-based care for type 2 diabetes, including diabetes education, health coaching, and mental health resources, can significantly improve prognosis and quality of life. However, most patients with type 2 diabetes still struggle to adhere to 1 or more major cornerstones of treatment: physical activity, healthy eating, blood glucose monitoring, and medication.4–6 Nonadherence to these health behaviors is independently associated with poor glycemic control; increased rates of vascular, renal, and other complications; and mortality.4,5,7 Therefore, a continued focus on finding effective and practical approaches to improve health behavior adherence is critical.
Psychological status can play a major role in clinical type 2 diabetes outcomes and health behavior adherence. Depression is associated with nonadherence, low function, and higher rates of complications.8 In contrast, higher baseline levels of positive psychological constructs, such as optimism and positive affect, are associated with greater subsequent improvements in health behaviors (and medical outcomes) in patients with type 2 diabetes and other chronic conditions, controlling for baseline physical activity, medical illness severity, and depression.9–11 These prior studies9–11 suggest a prospective and independent role of positive constructs on health behaviors, above and beyond the effects of medical variables and depression. These constructs may therefore represent an important and novel treatment target for patients with type 2 diabetes who struggle to adhere to recommended health behaviors.
Positive psychology interventions may represent an innovative and effective approach to improving health behavior adherence in type 2 diabetes. Positive psychology interventions use structured exercises (eg, performing acts of kindness, identifying and using personal strengths) to increase the frequency and intensity of positive emotional states and have reduced distress and improved well-being in > 3,000 study participants.12,13 These interventions are well accepted by patients, require little staff training, and can be delivered remotely.13–15 Positive psychology–based programs have also led to improvements in health behaviors in studies of patients with hypertension or heart disease,15–17 bolstering the case for positive psychology to improve behaviors and clinical outcomes in chronic conditions like type 2 diabetes. Another benefit of positive psychology is that it can offset the common psychological distress and low motivation that impair self-care in type 2 diabetes,18,19 without being limited to use only in the minority of type 2 diabetes patients with clinical depression. Despite these appealing features, there has been minimal use of positive psychology interventions in this population.20
To address this gap, we developed a positive psychology intervention for patients with type 2 diabetes and suboptimal adherence to health behaviors and tested it in a first-step proof-of-concept trial. The primary aims of this initial trial were feasibility (measured by rates of positive psychology exercise completion) and acceptability (measured by participant ratings of exercise ease and utility). We also explored the impact of the intervention by examining pre-post changes in positive psychological constructs, depression, anxiety, diabetes-related distress, diabetes self-care, and self-reported health behavior adherence.
METHOD
Overview
This was a pilot and feasibility study in adult inpatients and outpatients at an urban academic medical center recruited between December 2013 and December 2014 (ClinicalTrials.gov identifier: NCT02736084). Participants had type 2 diabetes and reported suboptimal adherence to health behaviors on the Medical Outcomes Study Specific Adherence Scale (MOS SAS).21 All participants received a 12-week positive psychology intervention and were administered preintervention and postintervention assessments of mood and physical symptoms. Detailed methods and rationale for the study, including the creation of the intervention manual and interventionist training procedures, are described elsewhere.22 The aims of this pilot trial were to examine the feasibility of implementing the intervention; assess its acceptability to patients; explore effects on psychological, medical, and functional outcomes; and adapt it for future studies on the basis of these outcomes and participant feedback. Institutional review board approval from our health care system was obtained prior to any study procedures.
Participants
Study criteria. English-speaking adult patients with (1) type 2 diabetes (meeting American Diabetes Association criteria,23 eg, glycated hemoglobin A1c (HbA1c) > 6.5% or fasting glucose > 126 mg/dL) and (2) suboptimal adherence (score < 15/18 on the MOS SAS items for medication, diet, and exercise) were eligible. We selected this MOS SAS cutoff because it required at least mild nonadherence but allowed inclusion of the majority of patients with type 2 diabetes to ensure a broadly applicable intervention.
Exclusion criteria were (1) cognitive impairment precluding consent or meaningful participation in the positive psychology exercises, assessed using a 6-item screen developed for research,24 and (2) lack of telephone access (given that the intervention was delivered via phone).
Recruitment and baseline assessments. Participants were enrolled from the outpatient diabetes center and inpatient medical units of our urban academic medical center; we included both populations to capture stable outpatients and those with more significant comorbid medical illness. In both locations, research staff introduced the study, assessed for inclusion and exclusion criteria, and obtained written informed consent. Postenrollment, and prior to initiation of the intervention, participants completed baseline self-report measures of clinical outcomes.
Intervention
Written participant and interventionist manuals specific to patients with type 2 diabetes were generated using the team’s positive psychology intervention for patients with other medical illness, existing literature on psychological factors and adherence in type 2 diabetes, and consultation with study team experts in diabetes. The intervention included 7 separate positive psychology exercises13,25–28 (Table 1 provides a description of specific exercises), which were assigned by the interventionist, recorded in the treatment manual by the participant, and then reviewed together at weekly (for the first 4 weeks) or biweekly (for the remainder of the intervention) calls.
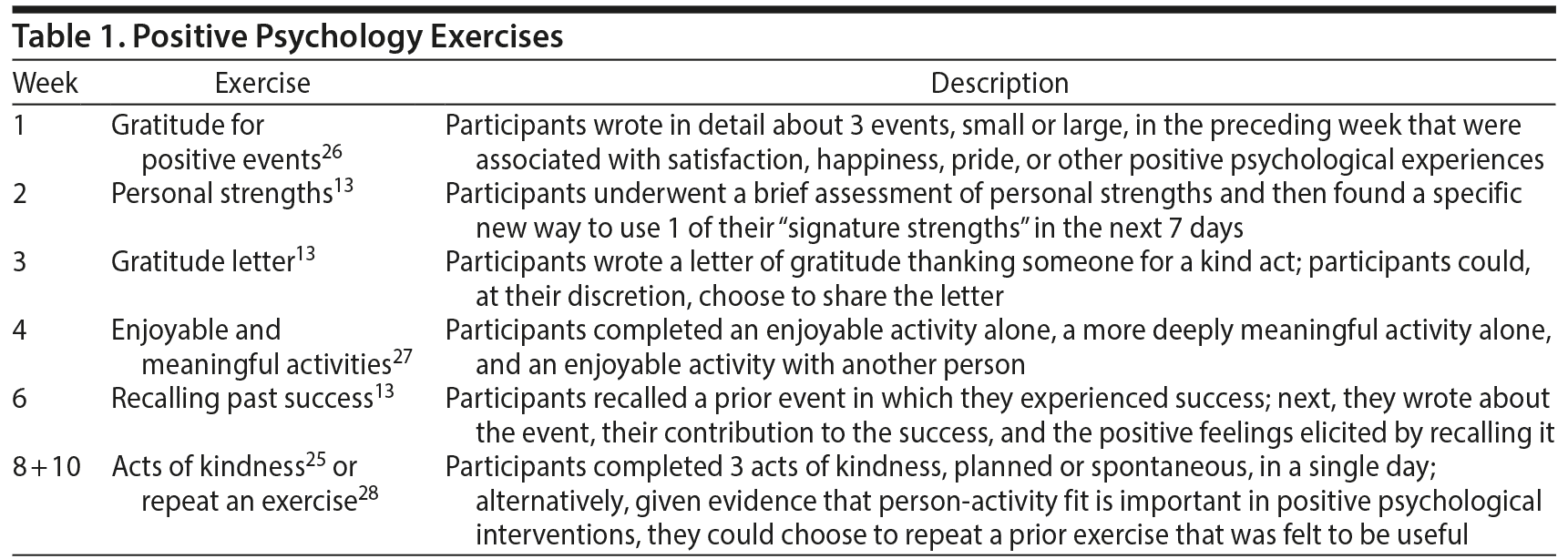
During the initial enrollment visit in the hospital or clinic, participants received a treatment manual. In person or by telephone, the study interventionist reviewed the introductory portion of the manual and discussed the first exercise (gratitude for positive events) with the participant to increase understanding, alliance, and engagement with the intervention. In subsequent weeks, exercises were completed independently by participants and recorded in their treatment manual. In the final week, after exercise review, the interventionist and participant discussed future implementation and ways to incorporate the principles into daily life. Together, they also created a specific, written plan to perform positive psychological activities over the next 4 weeks, with the goal of maintaining use of these skills.
Study Outcome Assessments
Feasibility and acceptability (the main aims of this proof-of-concept study) were assessed via several metrics. For feasibility, at each participant phone session, study interventionists recorded rates of exercise completion. To assess acceptability (and immediate impact), participants rated their optimism and positive affect on a 0–10 Likert scale prior to completing the exercise and then immediately following the exercise. After the exercise, participants also rated the ease and overall utility of the exercise on a 0–10 scale. All of these acceptability metrics were reported by participants to the interventionist at each weekly call. In addition to these quantitative measures of feasibility and acceptability, at week 12, open-ended feedback about the overall intervention’s ease, utility, and applicability to type 2 diabetes was elicited from participants.
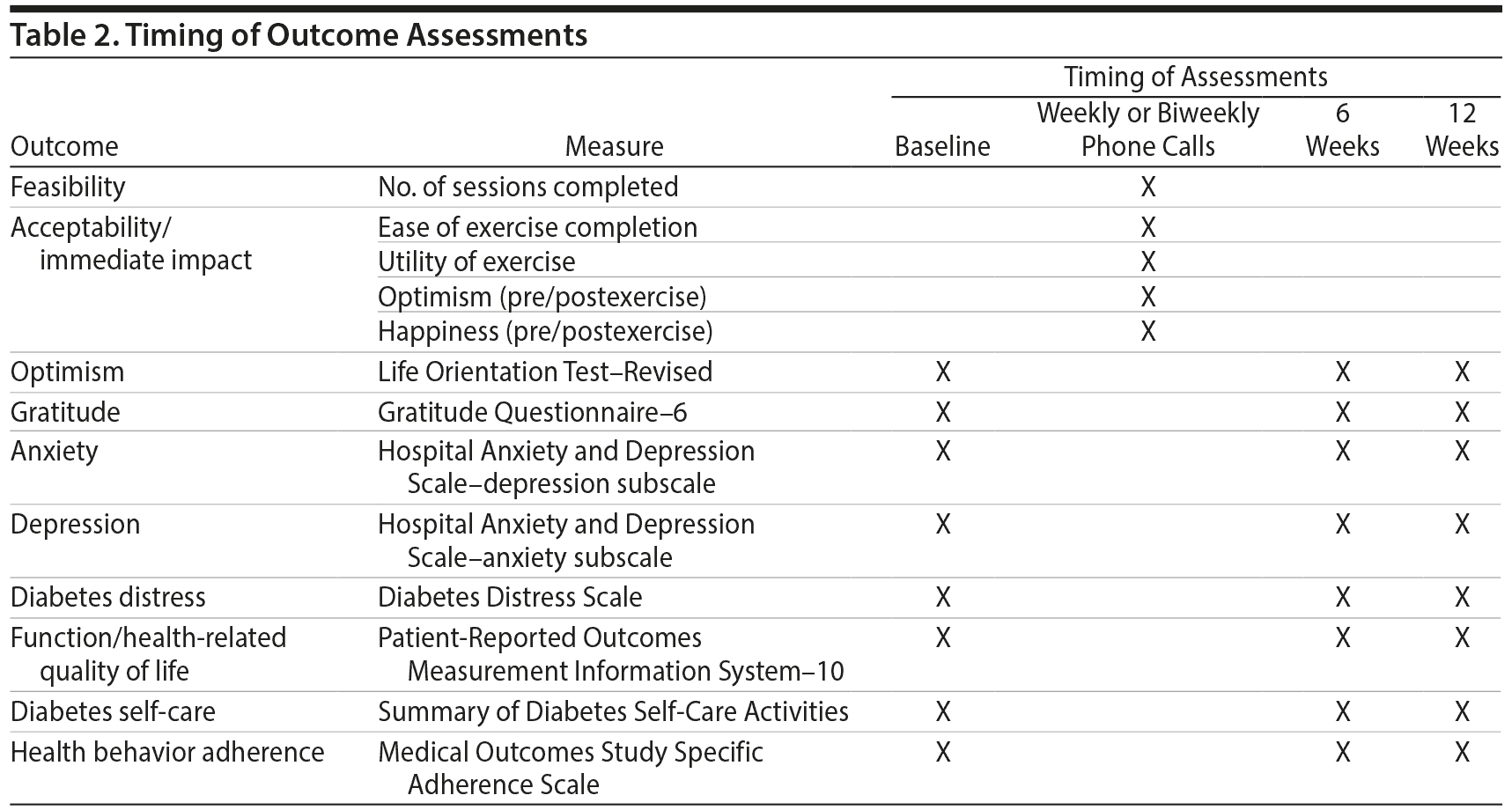
In addition, as a secondary aim, we explored pre-post change in patient-reported clinical outcomes (Table 2). At baseline, 6 weeks, and 12 weeks, noninterventionist study staff obtained the following validated measures of study outcomes:
- Optimism was measured by the 6-item Life Orientation Test–Revised,29 a frequently used measure of dispositional optimism.
- Gratitude was measured by the 6-item Gratitude Questionnaire–6.30
- Anxiety and depression were measured by the 14-item Hospital Anxiety and Depression Scale,31 which was designed for medically ill patients and has few somatic symptom items.
- Diabetes-related distress was measured by the 17-item Diabetes Distress Scale,32 a validated scale for use specifically in patients with diabetes.
- Health-related quality of life and function were measured by the National Institutes of Health–supported 10-item Patient-Reported Outcomes Measurement Information System physical function scale.33
- Diabetes self-care behaviors were measured using 11 items from Summary of Diabetes Self-Care Activities (SDSCA).34 Behaviors measured were diet, physical activity, blood sugar monitoring, and foot care. To reduce the overall number of outcomes and comparisons, we combined these items into a composite scale (Cronbach α = 0.74 in this cohort) for our main analysis and then analyzed behavior-specific subscales and individual items accordingly in supplementary analyses.
- Health behavior adherence was measured by the 3 MOS SAS items assessed at baseline (described previously) and repeated at 6 and 12 weeks; these were measured individually and as a composite score.
Data Analysis
Primary aim: feasibility and acceptability (immediate impact). Descriptive statistics (proportions, means, and standard deviations) were used to assess rates of exercise completion (feasibility) and to summarize the Likert scale ratings of optimism, positive affect, ease, and utility reported immediately following each exercise (acceptability/immediate impact). To assess mean pre-post change in optimism and positive affect across exercises as a measure of acceptability and short-term impact, we utilized random-effects regression models with a random intercept for each participant. Use of random-effects models allowed us to control for within-participant correlations on the serially administered outcome measures.
Our a priori criteria for success on this primary aim were that the positive psychology exercises would be considered feasible if (1) 4 of the 7 positive psychology exercises were completed by a majority of participants and (2) participants had a mean rating of at least 6.5/10 on the rating of ease of completion across exercises. The positive psychology exercises were considered to have adequate initial impact if (1) participants’ mean ratings of exercise utility were 6.5/10 and (2) they rated optimism and positive affect significantly higher (P < .05) postexercise than preexercise.
We expected completion of 60 total positive psychology exercises. Assuming the moderate (Cohen d = 0.5) immediate effect of positive psychology exercises on optimism and positive affect that was seen in our prior positive psychology intervention studies,35 the study was powered at 96% for significantly higher postexercise ratings (compared to preexercise ratings) on optimism and positive affect, using an α = .05 and 2-sided tests.
Secondary aim: changes in clinical outcome measures. We compared pre-post changes in the outcome measures at the 2 timepoints (6 and 12 weeks, with 12 weeks being the primary endpoint for analyses) using random-effects regression models to allow for inclusion of missing data at either timepoint. We also calculated effect size (Cohen d) for each outcome measure by dividing the pre-post change in the measure by the pooled SD for the measure.
This proof-of-concept study was not designed (and was not powered) to detect statistically significant differences between baseline and later follow-up points for this exploratory aim,22 but these effect size calculations would allow an assessment of the potential effect of the positive psychology intervention. All statistical tests were 2-tailed and were performed using Stata 11.2 (StataCorp, College Station, Texas).
RESULTS
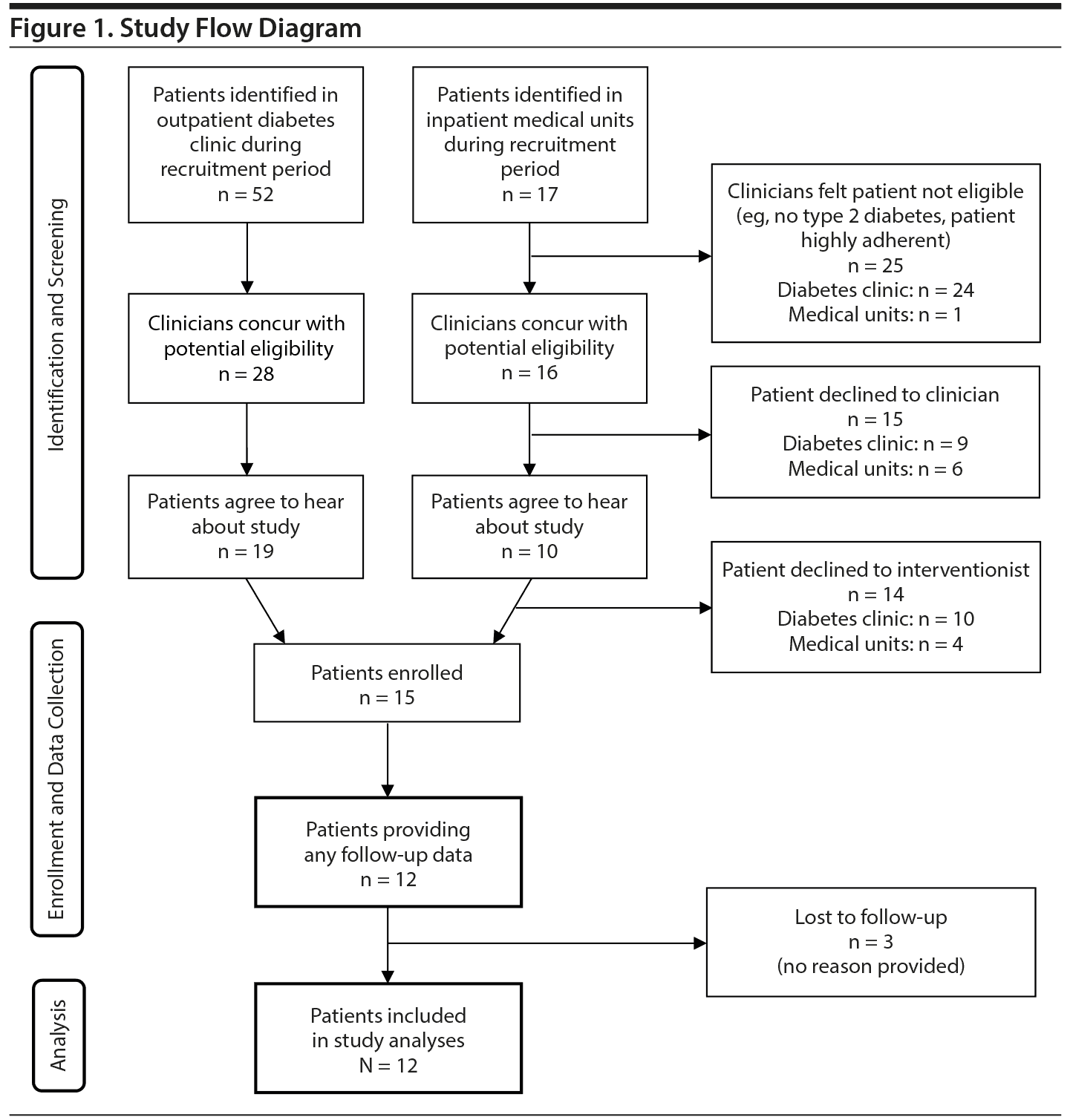
A total of 15 patients were eligible and enrolled; 12 participants provided baseline and follow-up questionnaire data (Figure 1 shows the study flow diagram) and were analyzed for study outcomes. Explicit reasons for dropout of the 3 patients who were lost to follow-up were not given, and there were no consistent characteristics of these patients with respect to demographic data, medical comorbidity, or recruitment location. Table 3 provides participants’ baseline sociodemographic and medical characteristics.

Primary Aim (feasibility and acceptability)
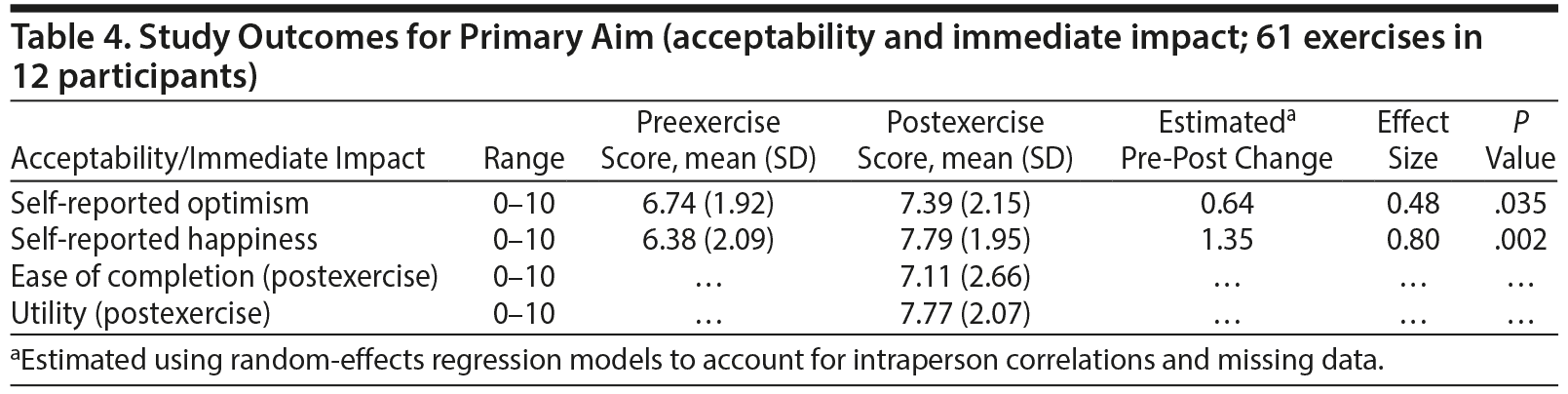
In total, 73% (61/84, mean = 5.1/7) of all possible exercises were completed by participants. In addition, 75% of participants (n = 9) completed a majority of the exercises, and 92% (n = 11) completed at least 2 exercises. By excluding 1 participant who withdrew immediately postdischarge (for medical reasons), these figures rise to 79% (mean = 5.5/7) of exercises completed and 82% of participants completing a majority of the exercises. Mean Likert scale ratings of exercise ease and utility, as well as postexercise ratings of optimism and happiness, were all greater than 7/10, and ratings of optimism (pre-post change in optimism = 0.64 points, d = 0.48, P = .035) and happiness/positive affect (pre-post change = 1.35 points, d = 0.80, P = .002) were significantly higher postexercise than preexercise (Table 4).
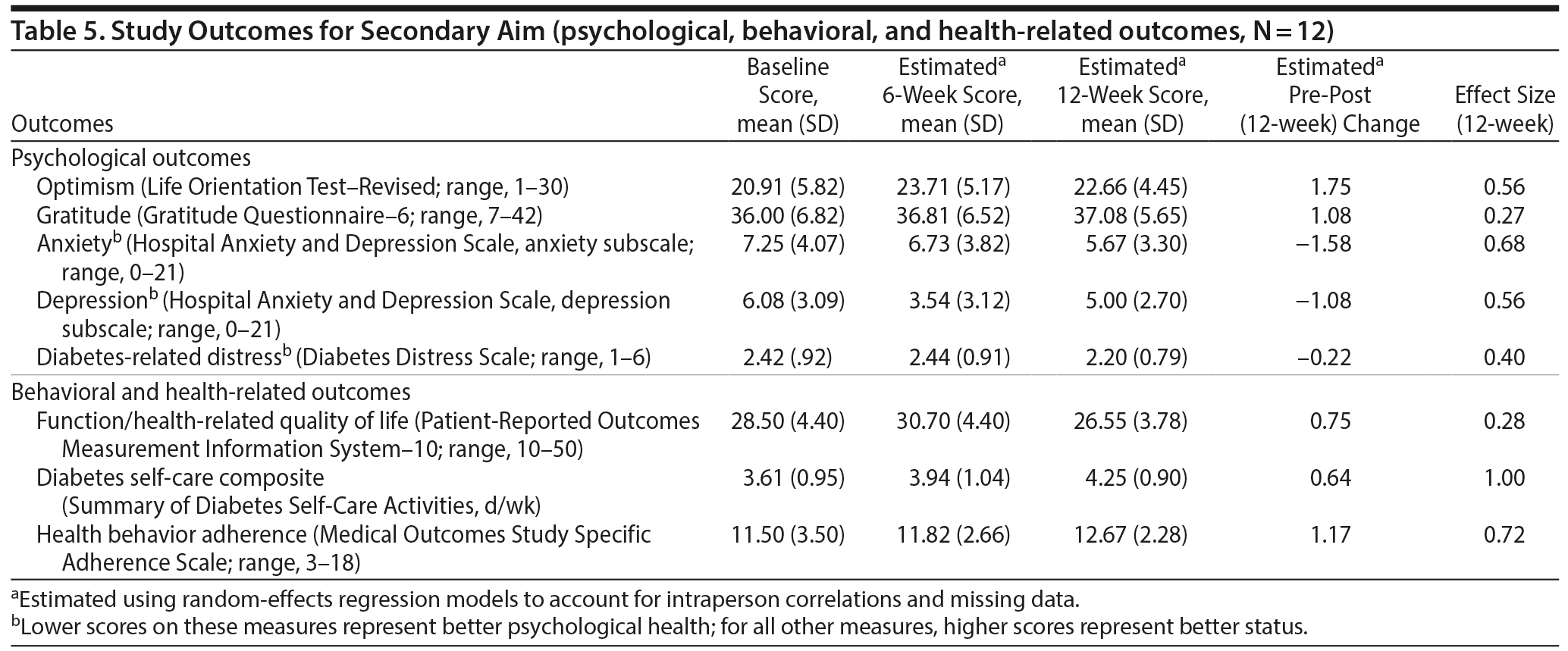
Secondary aim
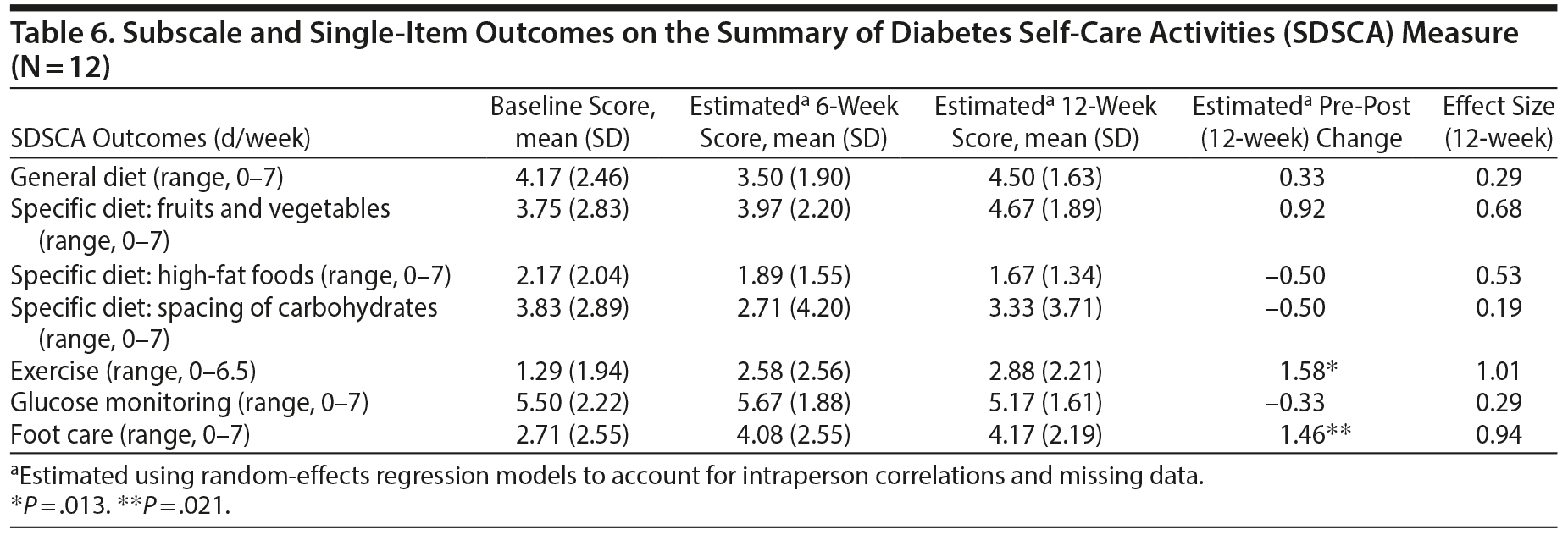
Postintervention scores on psychological measures (optimism, gratitude, depression, anxiety, and distress) were all improved compared to preintervention, with small-moderate effect sizes (d = 0.27–0.68, Table 5). Similarly, participants had improvements on self-reported diabetes self-care, function, and health behavior adherence over the 12-week intervention, with small to large effect sizes (d = 0.28–1.00, Table 5). On individual health behavior items using the MOS SAS, physical activity (mean = 2.25 [baseline] to 3.00 [12 weeks], d = 0.81, P = .047) was the behavior for which there was the greatest pre-post improvement. Similarly, concerning the SDSCA individual items, the greatest changes were seen on the physical activity and foot care items (Table 6).
Participant Feedback
At open-ended assessments at 12 weeks, participants generally reported high satisfaction with the intervention, finding the exercises straightforward, enjoyable, and confidence-boosting. They felt the phone-based intervention combined personal interaction with convenience and would not have preferred in-person or Internet interventions. Challenges associated with the intervention primarily were related to time constraints, with some participants feeing that they did not have time to complete additional activities due to work, caregiving, or medical commitments.
DISCUSSION
In a cohort of patients with type 2 diabetes and suboptimal health behavior adherence, we found that a 12-week, telephone-based intervention combining independent completion of positive psychology exercises with regular telephone sessions for exercise review was feasible and well accepted. A sizable proportion of participants completed a majority of exercises, well above our a priori threshold for feasibility, and participants also reported that the intervention exercises were easy and helpful, with significant improvements in well-being after completion of the exercises. In addition, there were promising pre-post improvements in psychological, functional, and health behavior–related measures.
These findings are concordant with the literature11 that has found positive psychological characteristics to be associated with greater function and health behavior participation and suggest that positive psychological constructs (eg, optimism) may be dynamic rather than static traits. These findings are also consistent with prior studies14,16,17 of positive psychology interventions in other medical cohorts that have found improvements in psychological and health behavior outcomes. In the 1 prior study20 of a positive psychology–based intervention in patients with type 2 diabetes, the intervention was associated with improvements in psychological outcomes but not health behavior adherence compared to waitlist control. However, that single prior study20 was delivered by Internet and was substantially shorter (5 weeks) than our intervention.
Positive psychology interventions might improve health behaviors and functional outcomes through several mechanisms. Positive psychological states and traits may be associated with greater resilience, a stronger sense of health-related self-efficacy, increased social interactions and social support, and enhanced self-regulation and motivation toward health goals.22,36 These factors, in turn, could lead to easier initiation of physical activity, greater confidence in meeting diet and activity goals, and more vitality and energy to engage in self-management when experiencing positive mood. Furthermore, reductions in negative psychological states (distress, anxiety, and depression) facilitated by positive psychology interventions may avoid the detrimental effects of these states on energy, concentration, motivation, interest, and confidence.8,37 These effects on self-efficacy, motivation, and energy may have the greatest effects on physical activity, consistent with our findings that the greatest improvements in health behaviors associated with the positive psychology intervention were in physical activity.
If the findings of this pilot study persist in larger, controlled studies, they may have substantial public health importance. Nonadherence to critical type 2 diabetes health behaviors is associated with higher rates of complications, greater functional loss, and diabetes-related mortality. Multicomponent health behavior interventions have been effective in research studies, but their complexity and intensity (eg, requiring multiple in-person visits over a long period) have made them difficult to implement in real-world settings.38–40 In contrast, positive psychology exercises are low-burden, and we found that they were feasible and acceptable to patients. In addition, they can be delivered remotely and require minimal training of providers.12,14,41 The focus of positive psychology on wellness and strengths, rather than illness and disease, is also highly patient-centered.42
There were several important limitations to this initial pilot study, including the lack of a control condition, the small sample size, an all-white cohort, and recruitment from a single academic medical center. Next-step studies should include a larger, more diverse sample and compare the intervention to a control condition. In addition, all outcomes in this initial feasibility trial were self-report, and although self-reported function and health behavior outcomes have been linked to adverse medical events, including mortality,43–48 objective measures of behavior may provide more detailed or accurate information.
In sum, a positive psychology intervention in patients with type 2 diabetes was feasible and exhibited a range of promising effects on clinically relevant outcome measures. Future studies of this intervention—alone or in combination with existing behavioral treatments—can determine whether this simple intervention can have substantial benefits in the large, vulnerable, and growing population of people with type 2 diabetes.
Submitted: October 30, 2015; accepted
February 3, 2016.
Published online: May 5, 2016.
Potential conflicts of interest: None.
Funding/support: This work was supported in part by National Institutes of Health (NIH) grant R01HL113272 to Dr Huffman and NIH grant K23HL123607 to Dr Celano. There were no other funding sources.
Role of the sponsor: The funding agencies did not have a role in the design or conduct of the study; the collection, management, analysis, or interpretation of data; or the preparation, review, or approval of the manuscript.
REFERENCES
1. Fast facts: data and statistics about diabetes. American Diabetes Association Web site. http://professional.diabetes.org/admin/UserFiles/0%20-%20Sean/FastFacts%20March%202013.pdf. Updated March 2013. Accessed November 20, 2015.
2. Chilton R, Wyatt J, Nandish S, et al. Cardiovascular comorbidities of type 2 diabetes mellitus: defining the potential of glucagonlike peptide-1-based therapies. Am J Med. 2011;124(suppl):S35–S53. doi:10.1016/j.amjmed.2010.11.004 PubMed
3. Lespérance F, Frasure-Smith N, Laliberté MA, et al. An open-label study of nefazodone treatment of major depression in patients with congestive heart failure. Can J Psychiatry. 2003;48(10):695–701. PubMed
4. Ho PM, Rumsfeld JS, Masoudi FA, et al. Effect of medication nonadherence on hospitalization and mortality among patients with diabetes mellitus. Arch Intern Med. 2006;166(17):1836–1841. doi:10.1001/archinte.166.17.1836 PubMed
5. Dunkler D, Dehghan M, Teo KK, et al; ONTARGET Investigators. Diet and kidney disease in high-risk individuals with type 2 diabetes mellitus. JAMA Intern Med. 2013;173(18):1682–1692. PubMed
6. Ford ES, Herman WH. Leisure-time physical activity patterns in the US diabetic population: findings from the 1990 National Health Interview Survey—Health Promotion and Disease Prevention Supplement. Diabetes Care. 1995;18(1):27–33. doi:10.2337/diacare.18.1.27 PubMed
7. Sloan FA, Bethel MA, Lee PP, et al. Adherence to guidelines and its effects on hospitalizations with complications of type 2 diabetes. Rev Diabet Stud. 2004;1(1):29–38. doi:10.1900/RDS.2004.1.29 PubMed
8. Lin EH, Katon W, Von Korff M, et al. Relationship of depression and diabetes self-care, medication adherence, and preventive care. Diabetes Care. 2004;27(9):2154–2160. doi:10.2337/diacare.27.9.2154 PubMed
9. Scheier MF, Matthews KA, Owens JF, et al. Dispositional optimism and recovery from coronary artery bypass surgery: the beneficial effects on physical and psychological well-being. J Pers Soc Psychol. 1989;57(6):1024–1040. doi:10.1037/0022-3514.57.6.1024 PubMed
10. Steptoe A, Dockray S, Wardle J. Positive affect and psychobiological processes relevant to health. J Pers. 2009;77(6):1747–1776. doi:10.1111/j.1467-6494.2009.00599.x PubMed
11. Celano CM, Beale EE, Moore SV, et al. Positive psychological characteristics in diabetes: a review. Curr Diab Rep. 2013;13(6):917–929. doi:10.1007/s11892-013-0430-8 PubMed
12. Bolier L, Haverman M, Westerhof GJ, et al. Positive psychology interventions: a meta-analysis of randomized controlled studies. BMC Public Health. 2013;13(1):119. doi:10.1186/1471-2458-13-119 PubMed
13. Seligman ME, Steen TA, Park N, et al. Positive psychology progress: empirical validation of interventions. Am Psychol. 2005;60(5):410–421. doi:10.1037/0003-066X.60.5.410 PubMed
14. Huffman JC, Mastromauro CA, Boehm JK, et al. Development of a positive psychology intervention for patients with acute cardiovascular disease. Heart Int. 2011;6(2):e14. doi:10.4081/hi.2011.e14 PubMed
15. Huffman JC, Millstein RA, Mastromauro CA, et al. A positive psychology intervention for patients with an acute coronary syndrome: treatment development and proof-of-concept trial [published online ahead of print October 19, 2015]. J Happiness Stud. doi:10.1007/s10902-015-9681-1
16. Ogedegbe GO, Boutin-Foster C, Wells MT, et al. A randomized controlled trial of positive-affect intervention and medication adherence in hypertensive African Americans. Arch Intern Med. 2012;172(4):322–326. doi:10.1001/archinternmed.2011.1307 PubMed
17. Peterson JC, Charlson ME, Hoffman Z, et al. A randomized controlled trial of positive-affect induction to promote physical activity after percutaneous coronary intervention. Arch Intern Med. 2012;172(4):329–336. doi:10.1001/archinternmed.2011.1311 PubMed
18. Aikens JE. Prospective associations between emotional distress and poor outcomes in type 2 diabetes. Diabetes Care. 2012;35(12):2472–2478. doi:10.2337/dc12-0181 PubMed
19. Gonzalez JS, Shreck E, Psaros C, et al. Distress and type 2 diabetes-treatment adherence: a mediating role for perceived control. Health Psychol. 2015;34(5):505–513. doi:10.1037/hea0000131 PubMed
20. Cohn MA, Pietrucha ME, Saslow LR, et al. An online positive affect skills intervention reduces depression in adults with type 2 diabetes. J Posit Psychol. 2014;9(6):523–534. doi:10.1080/17439760.2014.920410 PubMed
21. DiMatteo MR, Hays RD, Sherbourne CD. Adherence to cancer regimens: implications for treating the older patient. Oncology (Williston Park). 1992;6(suppl 2):50–57. PubMed
22. Huffman JC, DuBois CM, Millstein RA, et al. Positive psychological interventions for patients with type 2 diabetes: rationale, theoretical model, and intervention development. J Diabetes Res. 2015;2015:428349. doi:10.1155/2015/428349 PubMed
23. American Diabetes Association. Standards of medical care in diabetes—2013. Diabetes Care. 2013;36(suppl 1):S11–S66. PubMed
24. Callahan CM, Unverzagt FW, Hui SL, et al. Six-item screener to identify cognitive impairment among potential subjects for clinical research. Med Care. 2002;40(9):771–781. doi:10.1097/00005650-200209000-00007 PubMed
25. Otake K, Shimai S, Tanaka-Matsumi J, et al. Happy people become happier through kindness: a counting kindnesses intervention. J Happiness Stud. 2006;7(3):361–375. doi:10.1007/s10902-005-3650-z PubMed
26. Emmons RA, McCullough ME. Counting blessings versus burdens: an experimental investigation of gratitude and subjective well-being in daily life. J Pers Soc Psychol. 2003;84(2):377–389. doi:10.1037/0022-3514.84.2.377 PubMed
27. Peterson C, Park N, Seligman ME. Orientations to happiness and life satisfaction: the full life versus the empty life. J Happiness Stud. 2005;6(1):25–41. doi:10.1007/s10902-004-1278-z
28. Lyubomirsky S, Sheldon KM, Schkade D. Pursuing happiness: the architecture of sustainable change. Rev Gen Psychol. 2005;9(2):111–131. doi:10.1037/1089-2680.9.2.111
29. Scheier MF, Carver CS, Bridges MW. Distinguishing optimism from neuroticism (and trait anxiety, self-mastery, and self-esteem): a reevaluation of the Life Orientation Test. J Pers Soc Psychol. 1994;67(6):1063–1078. doi:10.1037/0022-3514.67.6.1063 PubMed
30. Mccullough ME, Emmons RA, Tsang JA. The grateful disposition: a conceptual and empirical topography. J Pers Soc Psychol. 2002;82(1):112–127. doi:10.1037/0022-3514.82.1.112 PubMed
31. Zigmond AS, Snaith RP. The Hospital Anxiety and Depression Scale. Acta Psychiatr Scand. 1983;67(6):361–370. doi:10.1111/j.1600-0447.1983.tb09716.x PubMed
32. Polonsky WH, Fisher L, Earles J, et al. Assessing psychosocial distress in diabetes: development of the Diabetes Distress Scale. Diabetes Care. 2005;28(3):626–631. doi:10.2337/diacare.28.3.626 PubMed
33. Cella D, Yount S, Rothrock N, et al; PROMIS Cooperative Group. The Patient-Reported Outcomes Measurement Information System (PROMIS): progress of an NIH Roadmap cooperative group during its first two years. Med Care. 2007;45(suppl 1):S3–S11. doi:10.1097/01.mlr.0000258615.42478.55 PubMed
34. Toobert DJ, Hampson SE, Glasgow RE. The Summary of Diabetes Self-Care Activities Measure: results from 7 studies and a revised scale. Diabetes Care. 2000;23(7):943–950. doi:10.2337/diacare.23.7.943 PubMed
35. Huffman JC, DuBois CM, Healy BC, et al. Feasibility and utility of positive psychology exercises for suicidal inpatients. Gen Hosp Psychiatry. 2014;36(1):88–94. doi:10.1016/j.genhosppsych.2013.10.006 PubMed
36. Pressman SD, Cohen S. Does positive affect influence health? Psychol Bull. 2005;131(6):925–971. doi:10.1037/0033-2909.131.6.925 PubMed
37. Katon W, Lin EH, Kroenke K. The association of depression and anxiety with medical symptom burden in patients with chronic medical illness. Gen Hosp Psychiatry. 2007;29(2):147–155. doi:10.1016/j.genhosppsych.2006.11.005 PubMed
38. Knowler WC, Barrett-Connor E, Fowler SE, et al; Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346(6):393–403. doi:10.1056/NEJMoa012512 PubMed
39. Stevens VJ, Obarzanek E, Cook NR, et al; Trials for the Hypertension Prevention Research Group. Long-term weight loss and changes in blood pressure: results of the Trials of Hypertension Prevention, phase II. Ann Intern Med. 2001;134(1):1–11. doi:10.7326/0003-4819-134-1-200101020-00007 PubMed
40. Svetkey LP, Stevens VJ, Brantley PJ, et al; Weight Loss Maintenance Collaborative Research Group. Comparison of strategies for sustaining weight loss: the weight loss maintenance randomized controlled trial. JAMA. 2008;299(10):1139–1148. doi:10.1001/jama.299.10.1139 PubMed
41. Schueller SM, Parks AC. Disseminating self-help: positive psychology exercises in an online trial. J Med Internet Res. 2012;14(3):e63. doi:10.2196/jmir.1850 PubMed
42. Lauver DR, Ward SE, Heidrich SM, et al. Patient-centered interventions. Res Nurs Health. 2002;25(4):246–255. doi:10.1002/nur.10044 PubMed
43. Grodin JL, Hammadah M, Fan Y, et al. Prognostic value of estimating functional capacity with the use of the Duke Activity Status Index in stable patients with chronic heart failure. J Card Fail. 2015;21(1):44–50. doi:10.1016/j.cardfail.2014.08.013 PubMed
44. Myint PK, Luben RN, Surtees PG, et al. Physical functional health predicts the incidence of coronary heart disease in the European Prospective Investigation into Cancer-Norfolk prospective population-based study. Int J Epidemiol. 2010;39(4):996–1003. doi:10.1093/ije/dyq061 PubMed
45. Parakh K, Bhat U, Thombs BD, et al. Self-assessed physical health predicts 10-year mortality after myocardial infarction. J Cardiopulm Rehabil Prev. 2010;30(1):35–39. doi:10.1097/HCR.0b013e3181c85a11 PubMed
46. Myint PK, Luben RN, Surtees PG, et al. Relation between self-reported physical functional health and chronic disease mortality in men and women in the European Prospective Investigation into Cancer (EPIC-Norfolk): a prospective population study. Ann Epidemiol. 2006;16(6):492–500. doi:10.1016/j.annepidem.2005.04.005 PubMed
47. Kassab Y, Hassan Y, Abd Aziz N, et al. Patients’ adherence to secondary prevention pharmacotherapy after acute coronary syndromes. Int J Clin Pharmacol. 2013;35(2):275–280. doi:10.1007/s11096-012-9735-y PubMed
48. Katon W, Russo J, Lin EH, et al. Diabetes and poor disease control: is comorbid depression associated with poor medication adherence or lack of treatment intensification? Psychosom Med. 2009;71(9):965–972. doi:10.1097/PSY.0b013e3181bd8f55 PubMed
Enjoy this premium PDF as part of your membership benefits!


