Prevalence and Treatment of Depression in Children and Adolescents With Sickle Cell Disease: A Retrospective Cohort Study
Objective: To describe the prevalence and treatment of comorbid depressive disorders in children and adolescents diagnosed with sickle cell disease.
Method: A retrospective cohort design evaluating South Carolina Medicaid medical and pharmacy claims between January 1, 1996, and December 31, 2006, was employed to identify 2,194 children and adolescents aged 17 years and younger diagnosed with sickle cell disease. Cohorts diagnosed with and without comorbid unipolar depressive disorders (using DSM-IV-TR criteria) were then compared.
Results: Forty-six percent of the sickle cell disease cohort was diagnosed with a depressive disorder (n = 1,017), either dysthymia (90%) or major depressive disorder (10%). Dysthymia was diagnosed at approximately 9 years of age, whereas major depressive disorder was diagnosed at approximately 14 years of age. Compared with the controls, the sickle cell disease cohort with depression had more acute vaso-occlusive pain and acute chest syndrome visits per year, developed more complications with related organ damage, and incurred significantly higher outpatient, acute (emergency + inpatient), and total sickle cell disease care costs. The depression cohort was primarily treated with selective serotonin reuptake inhibitors (SSRIs; 12%) or serotonin-norepinephrine reuptake inhibitors (SNRIs; 10%) for approximately 9 months. Although alleviating the comorbid depression might positively affect their sickle cell disease pain, over 80% of the patients received no antidepressant medications, and many of the prescribed SSRIs and SNRIs have previously shown no impact on relieving chronic pain.
Conclusions: Comorbid depression in sickle cell disease is associated with adverse course and outcomes. These findings underscore the need for earlier and more aggressive treatment of comorbid depression by primary care or psychiatric providers in order to reduce the chronic, severe pain-depression burden on these patients.
Prim Care Companion CNS Disord 2011;13(2):e1-e7
© Copyright 2011 Physicians Postgraduate Press, Inc.
Submitted: August 2, 2010; accepted September 22, 2010.
Published online: March 10, 2011 (doi:10.4088/PCC.10m01063).
Corresponding author: Jeanette M. Jerrell, PhD, Department of Neuropsychiatry and Behavioral Science, University of South Carolina School of Medicine, 3555 Harden St Ext, CEB 301, Columbia, SC 29203 ([email protected]).
Medical advances in the treatment of sickle cell disease (SCD) have transformed it from a condition associated with very early morbidity and mortality into a life-long, chronic condition.1 Erythrocytes in children with SCD become deoxygenated, dehydrated, and crescent shaped and tend to aggregate or stick to blood vessel walls, thereby blocking blood flow within limbs and organs and causing recurring, acute vaso-occlusive pain episodes. The recurring nature of these acute vaso-occlusive pain episodes is recognized as a chronic pain condition.1,2
Children with chronic, severe pain conditions such as SCD commonly experience difficulties with depression and anxiety, which are associated with adverse course and outcomes for the SCD illness such as increased daily vaso-occlusive pain, increased opioid use, longer hospital lengths of stay or increased use of acute services, increased complications, inappropriate pain coping strategies, poorer physical function, reduced quality of life, adolescent substance abuse, and dependence on opioid analgesics as adults.1,3-10 As a consequence of chronic hemolysis and vaso-occlusion, physical limitations may result from delayed growth and development, eg, lower body weight, shorter stature, cognitive impairment/delays secondary to frequent seizures or stroke and missed school days, frequent fatigue, and delayed puberty.11 By adolescence, these complications and the associated feelings of inadequacy in dealing with daily and frequently acute pain episodes are associated with lower self-esteem, social isolation, poor school performance, and hopelessness.12,13 Rates of successful suicides are relatively high among the chronically ill compared to other populations, but may be reduced with treatment.14
Clinical Points
- Comorbid depression in sickle cell disease is associated with a more severe course and outcomes.
- Earlier and more aggressive treatment of comorbid depression or depressive symptoms is needed to reduce the severe pain-depression burden on these young patients.
- Additional incentives for increasingly effective clinical use of antidepressant treatments in young patients affected by chronic pain and depression conditions need to be provided and investigated.
Comorbid depression in children and adolescents with chronic pain conditions is frequently undiagnosed or misdiagnosed by caregivers and may go untreated until the loss of routine ability and function results.15 Previous studies suggest that black adults with SCD report higher levels of diagnosed depression (26%) or depressive symptoms as measured by symptom scales (32%-38%) than do the US general population (9.5%)16,17 or the black population without SCD.18,19 Small studies in adolescents with SCD have also reported significant problems in social relationships, isolation, and school failure, with 29% meeting the criteria for depression based on symptom scales.1
Treatment of depression with antidepressants and therapeutic interventions is needed to improve the quality of life and the disease course in patients with SCD.18 However, in 1 study only one-third of patients with SCD reported receiving treatment from a mental health professional.14 Antidepressants often form the cornerstone of treatment in affected individuals. As newer, safer, and easier-to-use agents have been developed and marketed, prescribing practices have shifted from tricyclic antidepressants toward selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and heterocyclic second-generation agents.20 Utilization of psychological interventions including patient education, cognitive-behavioral therapy, and special educational support to help improve the quality of life of these patients is recommended but is not always available to patients and their families.5
The high prevalence of and hazards posed by depressive symptoms or disorders in SCD populations provide the impetus for carefully examining the prevalence and treatment of unipolar depressive disorders, ie, major depressive and dysthymic disorders, and exploring their association with vaso-occlusive pain episodes and other complications of SCD in a predominantly black pediatric population covered by Medicaid. Our main hypotheses were that depressive disorders are more common in pediatric SCD than previously recognized and that the high correlation between severe, chronic pain conditions and depression needs to be considered by pediatric and psychiatric providers when prescribing an antidepressant treatment regimen that will effectively relieve both conditions, even in children under the age of 12 years.
METHOD
Claims data for South Carolina’s Medicaid program were obtained through the state’s Office of Research and Statistics. Each Medicaid medical claim identifies a service encounter and gives the date of service and the International Classification of Diseases (ICD), Ninth Revision Clinical Modification diagnosis codes related to that visit (visit file). Pharmacy claims identified the medication dispensed and the date the prescription was filled (pharmacy file). A separate data file regarding eligibility was used to summarize the demographics for each patient (person file). The databases are frequently updated prior to being made available for analysis. This study was approved by the University of South Carolina Institutional Review Board, Columbia, as exempt from human subject research guidelines under 45 Code of Federal Regulations part 46.
Medical and pharmacy claims for the calendar years January 1, 1996, through December 31, 2006, were used to identify a cohort of child and adolescent patients (aged 17 years and under) enrolled in and eligible for Medicaid for a minimum of 9 months in each calendar year included in this analysis who had a service encounter with a diagnosis of SCD (thalassemia and sickle cell trait were not included). This process resulted in the identification of 2,194 SCD patients who were further analyzed to identify those with depression (DSM-IV-TR codes major depressive disorder single and recurring episodes: 296.2 and 296.3 and dysthymic disorder: 300.4; n = 1,017; 46.4%).
Analyses
To address research questions regarding the prevalence and treatment of depression, anxiety, and any other psychiatric conditions (attention-deficit/hyperactivity disorder [ADHD], adjustment disorder, conduct disorder, substance use disorder) among children and adolescents diagnosed with SCD, descriptive statistics were compiled for these diagnoses from the visits file. Primary or secondary diagnosis codes for sickle cell vaso-occlusive pain crises, acute chest syndrome, or pneumonia; percentage of SCD services received from a specialty clinic for SCD; and SCD complications (cardiovascular, hepatic, renal, spleen, pulmonary, cerebrovascular events [ie, stroke and seizures], and bone necrosis) were also compiled for these diagnoses from the visits file. Prescriptions for hydroxyurea and prophylactic penicillin were coded from the pharmacy file. Procedure codes for adenotonsillectomy surgery and blood transfusions were obtained from the visits file. The number of outpatient and acute services (emergency visits + hospital days) associated with depression/anxiety or SCD treatment was aggregated for the depression and control cases.
Descriptive statistics were compiled for prescribed antidepressant and analgesic medications, and these medications were grouped together (categorized) for the multivariate analyses. Antidepressants were categorized as SSRIs for citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, and sertraline or SNRIs/heterocyclics for bupropion, duloxetine, maprotiline, mirtazapine, nefazodone, trazodone, or venlafaxine. Opioid analgesics were coded in the analyses as weak opioids (eg, codeine, hydrocodone) or stronger opioids (eg, morphine, oxycodone).
To summarize the individual risk factors, SCD interventions, and complications that predict being in the depression group or not, a logistic regression was performed using race, gender, vaso-occlusive pain visits per year, acute chest syndrome visits per year, receipt of prophylactic penicillin, adenotonsillectomy, receipt of hydroxyurea, blood transfusions per year, percent of SCD care received from a specialty clinic, and the number of organ system-specific complications involved (categorized as ≤ 1 or ≥ 2 systems) as the independent variables. Odds ratios and 95% confidence intervals are reported for these results.
To examine the impact of depression and its treatment over time on different facets of the SCD illness (ie, the change in frequency of vaso-occlusive pain or acute chest syndrome/pneumonia visits and in outpatient, acute, and total costs), a series of negative binomial regression models (for non-normally distributed count data; PROC GENMOD facility in SAS version 9.1, SAS Institute Inc, Cary, North Carolina) were employed to calculate a ratio of the log rate of each type of visit or cost per time period that the patient was covered in the Medicaid data set.21,22 Receipt of blood transfusions, prophylactic penicillin, or hydroxyurea (dichotomously coded as yes/no) and the percentage of SCD services received from a specialty clinic for SCD were used as covariates to represent interventions or type of care that might also explain differences in service costs. The resulting model estimates were converted into rate ratios (RRs) and 95% confidence intervals, and P values are used to report statistical significance using 2-tailed tests and α ≤ .05 for statistical significance.
RESULTS
Clinical Characteristics of Patients
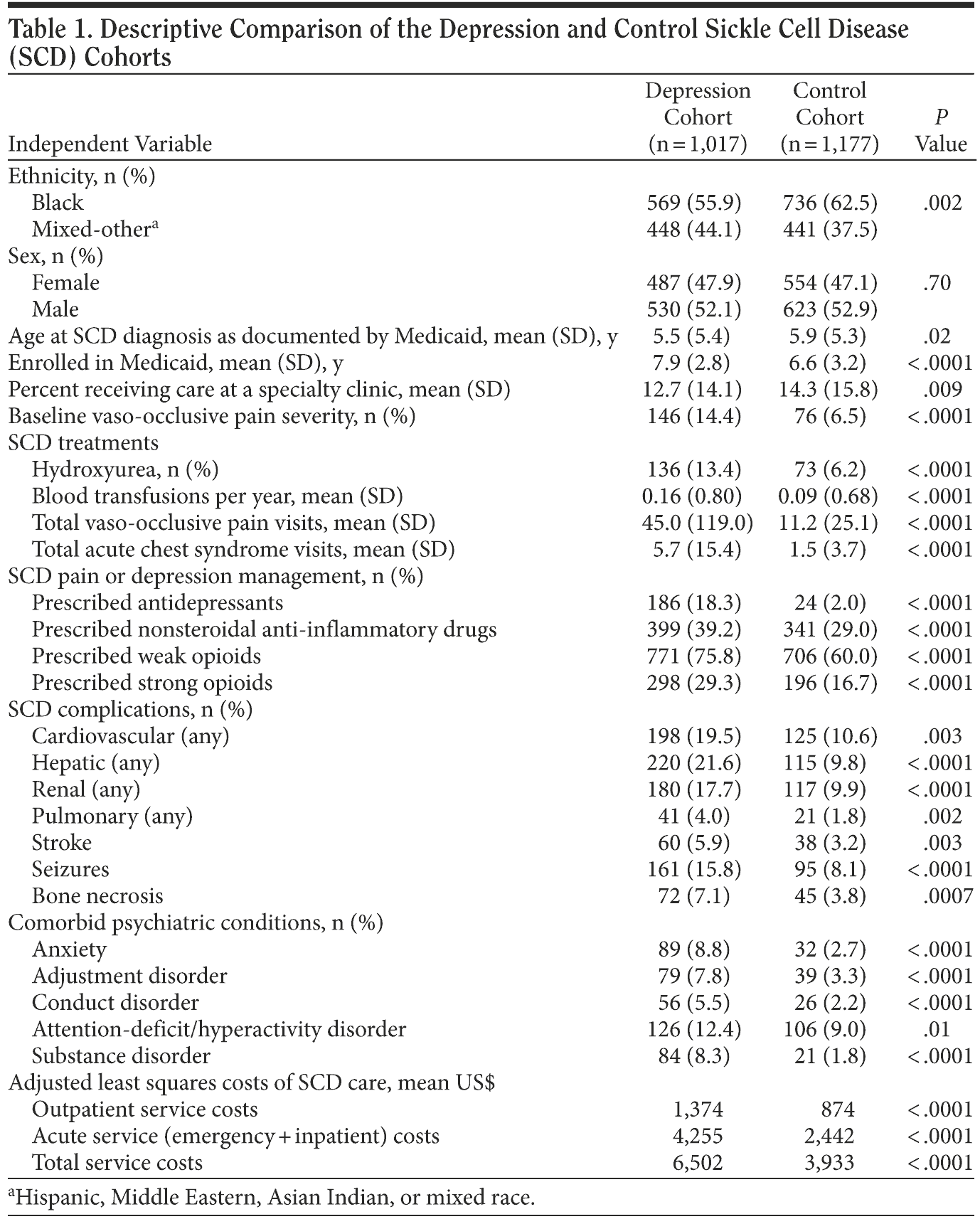
The patients with SCD who were diagnosed with depression and the controls (SCD only) are described in Table 1. Both the depression and control cohorts were predominantly male (52%) and approximately 5 years old when their first SCD diagnosis was documented under Medicaid coverage; 59% were black and 41% were of mixed-other race, which included Hispanic, Middle Eastern, Asian Indian, or mixed ancestry. The cohorts were enrolled in Medicaid a mean of 6-8 years.
Of those diagnosed with depression, 90% had dysthymic disorder (n = 914), and the remaining 10% were diagnosed as having major depressive disorder (n = 102). The mean age at onset or documented dysthymia diagnosis was 9.2 years of age (SD = 4.1) compared to the mean age of a major depressive disorder diagnosis of 14.2 years (SD = 5.0). Besides depression, small numbers of these pediatric patients with SCD were also diagnosed with anxiety (n = 121, 5.5%), conduct disorder (n = 82, 3.7%), ADHD (n = 232, 10.6%), or a substance use disorder (n = 105, 4.8%).
The depression and control groups were compared on numerous aspects of their SCD and psychiatric conditions (Table 1). Fourteen percent of the depression cohort and 6.5% of the control cohort had 4 to 6 vaso-occlusive pain episodes during the first year of SCD service (the highest severity indicator) (Table 1). Thirteen percent of those diagnosed with depression were cared for by specialty clinics in South Carolina, whereas 14% of those without depression received this care. A higher percentage of the depression cohort also had diagnosed comorbid anxiety, conduct disorder, ADHD, and substance abuse than did the controls. For vaso-occlusive pain management, significantly more patients with depression were treated with nonsteroidal anti-inflammatory drugs (NSAIDs; 39% vs 29%) and both weak (76% vs 60%) and strong opioids (29% vs 17%) compared to those without depression.
Differences in SCD clinical features between the depression and nondepression cohorts are summarized in Table 2. Those diagnosed with depression were more likely to have vaso-occlusive pain (OR = 1.06; 95% CI, 1.03-1.08) and acute chest syndrome visits in a given year (OR = 1.32; 95% CI, 1.11-1.59), to receive prophylactic penicillin (OR = 2.07; 95% CI, 1.52-2.81) and adenotonsillectomy (OR = 1.43; 95% CI, 1.09-1.88), and to develop 2 or more SCD complications with related organ damage (OR = 3.62; 95% CI, 1.70-7.74) than those who were not depressed.
Pharmacologic treatment for their diagnosed depression consisted mainly of SSRIs (n = 122, 12%), SNRI/heterocyclics (n = 111, 10.9%), and tricyclic antidepressants (n = 2, 0.2%); 41 patients were treated sequentially with both SSRI and SNRI/heterocyclic agents. Of those diagnosed with depression, 81% (n = 825) received no antidepressant treatment during the mean of 8 years they were covered by Medicaid. The mean number of days covered by prescription fills/refills in the pharmacy file for each type of antidepressant was 291 days (SD = 473) for SSRIs, 300 days (SD = 423) for SNRI/heterocyclic agents, and 165 days (SD = 106) for tricyclic antidepressants. Within these broader categories, the majority of patients treated with SSRIs were taking citalopram 20 mg, escitalopram 10 mg, paroxetine 10 or 20 mg, fluoxetine 20 mg, or sertraline 50 mg. Those prescribed SNRI/heterocyclic agents were mainly taking venlafaxine 75 mg, mirtazapine 15 mg, bupropion 150 mg, or duloxetine 60 mg. The percentage of patients prescribed antidepressant medication did not differ significantly for those receiving any SCD specialty clinic services (n = 145, 15.6%) or receiving only primary care services (n = 165, 13.1%). The Medicaid pharmacy file does not allow for the calculation of the prescribed daily dose, nor can we distinguish whether the antidepressant was prescribed by a psychiatrist or a pediatrician/primary care provider. No data are available on nonmedication treatments for depression in the visits file.
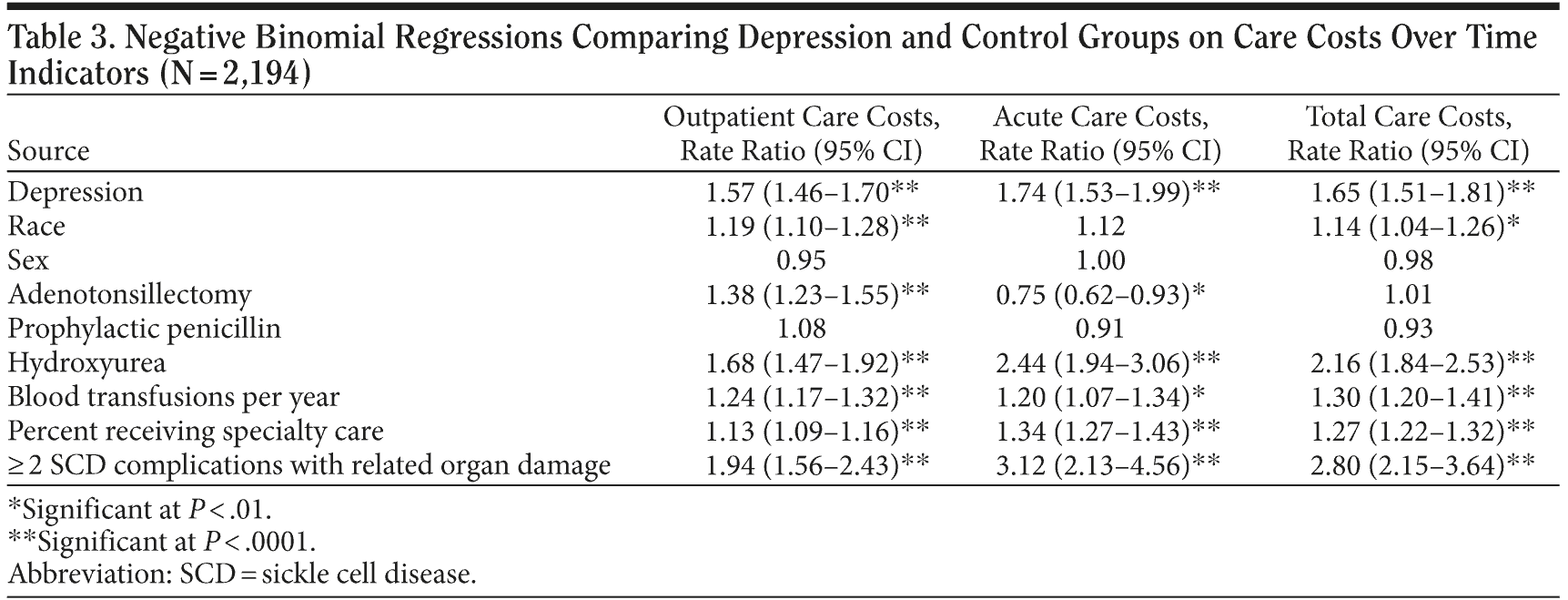
Service costs for SCD care were also associated with the presence of comorbid depression, with significantly higher outpatient, acute (emergency + inpatient), and total SCD service costs for those in the depression group (Table 1). Receipt of hydroxyurea, SCD specialty care, or blood transfusions and having 2 or more complications indicating organ damage were associated with higher acute, outpatient, and total SCD care costs but were controlled for in estimating the independent effect of a depression diagnosis on service utilization/care costs (Table 3).
DISCUSSION
The prevalence of diagnosed depression in this pediatric cohort (46%) was higher than previously reported in either adult or adolescent samples using depression rating scales (26% and 29%, respectively).16,17 The sociodemographic strata served by Medicaid, ie, those living at the poverty level in predominantly rural or small urban areas, could be a contributing factor in the higher rates of depression observed in our cohort compared to previous studies, which normally serve urban, clinic-based patients. Not only were a higher percentage of children diagnosed with a depressive disorder than might be expected from previous reports, but also the age at onset was younger among these patients with SCD. Dysthymic disorder was diagnosed in 914 younger patients (mean age of 9 years), whereas major depressive disorder was diagnosed in 102 adolescents (mean age of 14 years).
Furthermore, every clinical severity indicator employed in this study underscores the fact that when compared to individuals without depression, pediatric patients with SCD with depression experienced more severe SCD symptoms (ie, more vaso-occlusive pain visits, acute chest syndrome/pneumonia visits), were prescribed more hydroxyurea and blood transfusions, and had more cardiovascular, hepatic, renal, and pulmonary complications. By definition, these clinical “severity” and “disability” levels are higher than pediatric cases of depression that are not complicated by severe pain, regardless of whether a major depressive episode is diagnosed. The extent of SCD and depression severity and disability in this young cohort deserves further study as does the development of new approaches to treating both of these comorbid conditions.
Over 80% of those children and adolescents diagnosed with a unipolar depressive disorder were prescribed no antidepressant medication, but were significantly more likely to be prescribed opioid analgesics. Patients prescribed SSRIs or SNRIs were treated for approximately 9 months. We were unable to determine from this administrative data set whether the low rate of treatment of these depressive disorders is due to a lack of approved clinical guidelines for using antidepressants in children and adolescents, a lingering concern about safety issues associated with antidepressant treatment (eg, suicidality), underdiagnosis of psychiatric conditions, or a reluctance of the patient/family to accept treatment with “psychiatric” medications. However, considering the cumulative burden that severe chronic pain and comorbid depression place on these children and their families, it seems prudent to reassess long-term treatment and management protocols in order to reduce this burden.12,13
Many patients with chronic pain are depressed, and alleviating the depression has a salutary effect on the pain. Since we have previously demonstrated that the combination of antidepressants and stronger opioids is significantly associated with fewer acute vaso-occlusive pain visits,23 further exploration of the use of antidepressants in conjunction with opioid treatments for management of pain and depression seems warranted. Several antidepressants have been proven to be efficacious in the management of disparate pain disorders, including the tricyclic antidepressants (particularly amitriptyline, doxepin, and imipramine) as well as venlafaxine, bupropion, and duloxetine.24-30 SSRIs appear to be much less effective in managing chronic pain conditions.24 Few of the antidepressants found to be effective in previous adult studies for pain and approved for use in children 6-18 years of age are being used with these children.
Most of the commonly used SSRI and SNRI/heterocyclic antidepressant medications have uncommon, but potentially important, hematologic side effects. These include increased risk of a bleeding complication or bruising, especially with the concomitant use of aspirin or NSAIDs, leukocytosis, leucopenia, thrombocytopenia, or anemia.3 Moreover, tricyclic antidepressants may potentiate the adverse effects of opioids,1 and patients prescribed hydroxyurea are also at increased risk of developing thrombocytopenia.31 SSRI and SNRI/heterocyclic antidepressant medications must be labeled for potential suicidality and other safety issues in patients less than 18 years of age. However, our previous investigations of the adverse events associated with the prescription of these antidepressants for a mean of 6 months of active treatment in children and adolescents served through Medicaid pointed to an elevated risk of developing only obesity/excessive weight gain, type 2 diabetes mellitus, and dyslipidemia, with no increase in the death rates.32 As therapeutic doses of antidepressants used for the treatment of pain are frequently lower than for treatment of major depressive disorder, the adverse event profiles for children and adolescents with SCD treated with antidepressant agents for comorbid pain and depression may be more manageable. When evaluating the overall benefit-risk ratio of antidepressants in children and adolescents with SCD with severe pain and comorbid depression, the primary care practitioner should carefully consider both the effectiveness and relative safety of these agents, while realizing that none of these agents is US Food and Drug Administration-approved for use in children and adolescents.
This study has some major strengths. First, the SCD cohort represents a large, heterogeneous group of children and adolescents. Second, the long-term observational study design provides additional information regarding important clinical correlates and their impact on pediatric SCD. Third, there is sufficient power in the SCD cohort to detect low-incidence complications and treatments. Finally, previous studies have found that although Medicaid databases provide much less detailed information on individuals than a structured research interview, the physician diagnoses and utilization data are more reliable than client or family self-reports, and the administrative data correspond to clinical medical record reviews in 75%-95% of the cases examined.33-36
Notwithstanding the strength of our analyses, these results also need to be interpreted with several limitations in mind. First, the data were not gathered using a prospective, controlled design. Second, structured clinical research interviews were not employed to confirm any of the assigned medical disorders. Third, these results primarily report associations and, as a result, directions of causality cannot be inferred. Fourth, there is no way to estimate how representative this Medicaid cohort is in relation to those in other states and service systems. Fifth, children and adolescents with SCD who dropped out of treatment or were periodically ineligible for Medicaid coverage are not represented in this data set, and their outcomes may differ from those patients who remained in Medicaid over time. Finally, no clinical data were available on these children prior to their Medicaid coverage.
In conclusion, comorbid depressive disorders appear to be undertreated with antidepressants by primary and specialty care providers serving patients with SCD, both in terms of the number of patients prescribed antidepressants and the use of antidepressants, which have demonstrated efficacy for adjuvant pain relief. Undertreatment of comorbid depression and SCD pain may be a “perpetuating factor” for recurring, severe vaso-occlusive pain episodes, worse illness course, higher medical costs, poor coping/adjustment to SCD symptoms, substance abuse to self-medicate vaso-occlusive pain symptoms, or addiction-like (ie, “drug-seeking”) behavior/inappropriate use of opioids in adolescence and adulthood.37,38 These results buttress recommendations for increased assessment and surveillance of depressive symptoms and episodes in individuals with SCD and provide additional incentives for exploring increasingly effective clinical use of antidepressant treatment strategies in young patients affected by chronic pain and depression conditions. Optimal treatment of depression in SCD populations by multidisciplinary providers also holds promise for improving medical outcomes and reducing care costs to Medicaid.
Drug names: bupropion (Wellbutrin, Aplenzin, and others), citalopram (Celexa and others), doxepin (Zonalon, Silenor, and others), duloxetine (Cymbalta), escitalopram (Lexapro and others), fluoxetine (Prozac and others), fluvoxamine (Luvox and others), hydrocodone (Vicodin, Zydone, and others), imipramine (Tofranil and others), mirtazapine (Remeron and others), morphine (Apokyn, Kadian, and others), oxycodone (OxyContin, Roxicodone, and others), paroxetine (Paxil, Pexeva, and others), sertraline (Zoloft and others), trazodone (Oleptro and others), venlafaxine (Effexor and others).
Author affiliations: Department of Neuropsychiatry and Behavioral Science, University of South Carolina School of Medicine, Columbia (Dr Jerrell); Department of Epidemiology and Biostatistics, University of South Carolina Arnold School of Public Health, Columbia (Dr Tripathi); and Departments of Psychiatry and Pharmacology, University of Toronto, Ontario, Canada (Dr McIntyre).
Potential conflicts of interest: Dr McIntyre has received research funding from AstraZeneca, GlaxoSmithKline, Merck, Servier, and Wyeth and has served as a consultant to and on the speaker’s boards for AstraZeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Organon, Ortho-McNeil-Janssen, Oryx, Pfizer, Prestwick, and Wyeth. Drs Jerrell and Tripathi report no conflicts of interest related to the subject of this article.
Funding/support: Data analysis was supported by the Departments of Neuropsychiatry, Pediatrics, and Internal Medicine, University of South Carolina School of Medicine, Columbia.
Disclaimer: The views expressed do not necessarily represent those of the funding agency or official findings of the South Carolina Department of Health and Human Services (Medicaid).
REFERENCES
1. Benton TD, Ifeagwu JA, Smith-Whitley K. Anxiety and depression in children and adolescents with sickle cell disease. Curr Psychiatry Rep. 2007;9(2):114-121. PubMed doi:10.1007/s11920-007-0080-0
2. Bodhise PB, Dejoie M, Brandon Z, et al. Non-pharmacologic management of sickle cell pain. Hematology. 2004;9(3):235-237. PubMed doi:10.1080/10245330410001701495
3. Becker M, Axelrod DJ, Oyesanmi O, et al. Hematologic problems in psychosomatic medicine. Psychiatr Clin North Am. 2007;30(4):739-759. PubMed doi:10.1016/j.psc.2007.07.006
4. Levenson JL, McClish DK, Dahman BA, et al. Depression and anxiety in adults with sickle cell disease: the PiSCES project. Psychosom Med. 2008;70(2):192-196. PubMed doi:10.1097/PSY.0b013e31815ff5c5
5. Simon K, Barakat LP, Patterson CA, et al. Symptoms of depression and anxiety in adolescents with sickle cell disease: the role of intrapersonal characteristics and stress processing variables. Child Psychiatry Hum Dev. 2009;40(2):317-330. PubMed doi:10.1007/s10578-009-0129-x
6. Anie KA. Psychological complications in sickle cell disease. Br J Haematol. 2005;129(6):723-729. PubMed doi:10.1111/j.1365-2141.2005.05500.x
7. Artz N, Zhang J, Meltzer D. Physical and mental health in adults hospitalized with sickle cell disease: impact on resource use. J Natl Med Assoc. 2009;101(2):139-144. PubMed
8. Barakat LP, Patterson CA, Daniel LC, et al. Quality of life among adolescents with sickle cell disease: mediation of pain by internalizing symptoms and parenting stress. Health Qual Life Outcomes. 2008;6(1):60. PubMed doi:10.1186/1477-7525-6-60
9. Cepeda ML, Allen FH, Price C, et al. Depression in children and adolescents with sickle cell disease. J Natl Med Assoc. 2000;92:10-14. PubMed
10. Morgan SA, Jackson J. Psychological and social concomitants of sickle cell anemia in adolescents. J Pediatr Psychol. 1986;11(3):429-440. PubMed doi:10.1093/jpepsy/11.3.429
11. Hurtig AL, Park KB. Adjustment and coping in adolescents with sickle cell disease. Ann N Y Acad Sci. 1989;565(1 Sickle Cell D):172-182. PubMed doi:10.1111/j.1749-6632.1989.tb24164.x
12. Edwards CL, Green M, Wellington CC, et al. Depression, suicidal ideation, and attempts in black patients with sickle cell disease. J Natl Med Assoc. 2009;101(11):1090-1095. PubMed
13. Palermo TM, Riley CA, Mitchell BA. Daily functioning and quality of life in children with sickle cell disease pain: relationship with family and neighborhood socioeconomic distress. J Pain. 2008;9(9):833-840. PubMed doi:10.1016/j.jpain.2008.04.002
14. Barakat LP, Schwartz LA, Simon K, et al. Negative thinking as a coping strategy mediator of pain and internalizing symptoms in adolescents with sickle cell disease. J Behav Med. 2007;30(3):199-208. PubMed doi:10.1007/s10865-007-9103-x
15. Chambliss CR, Heggen J, Copelan DN, et al. The assessment and management of chronic pain in children. Paediatr Drugs. 2002;4(11):737-746. PubMed
16. Jenerette C, Funk M, Murdaugh C. Sickle cell disease: a stigmatizing condition that may lead to depression. Issues Ment Health Nurs. 2005;26(10):1081-1101. PubMed doi:10.1080/01612840500280745
17. Burlew K, Telfair J, Colangelo L, et al. Factors that influence adolescent adaptation to sickle cell disease. J Pediatr Psychol. 2000;25(5):287-299. PubMed doi:10.1093/jpepsy/25.5.287
18. Hasan SP, Hashmi S, Alhassen M, et al. Depression in sickle cell disease. J Natl Med Assoc. 2003;95(7):533-537. PubMed
19. Laurence B, George D, Woods D. Association between elevated depressive symptoms and clinical disease severity in African-American adults with sickle cell disease. J Natl Med Assoc. 2006;98(3):365-369. PubMed
20. McIntyre RS, Jerrell JM. Polypharmacy in children and adolescents with major depressive disorder. J Clin Psychiatry. 2009;70(2):240-246. PubMed doi:10.4088/JCP.08m04212
21. Dodd S, Bassi A, Bodger K, et al. A comparison of multivariable regression models to analyse cost data. J Eval Clin Pract. 2006;12(1):76-86. PubMed doi:10.1111/j.1365-2753.2006.00610.x
22. Lipscomb HJ, Dement JM, Silverstein B, et al. Who is paying the bills? health care costs for musculoskeletal back disorders, Washington State Union Carpenters, 1989-2003. J Occup Environ Med. 2009;51(10):1185-1192. PubMed doi:10.1097/JOM.0b013e3181b68d0a
23. Jerrell JM, Tripathi A, Stallworth JR. Pain management in children and adolescents with sickle cell disease. Am J Hematol. 2011;86(1):82-84. doi:10.1002/ajh.21873 PubMed
24. Jackson KC 2nd, St Onge EL. Antidepressant pharmacotherapy: considerations for the pain clinician. Pain Pract. 2003;3(2):135-143. PubMed doi:10.1046/j.1533-2500.2003.03020.x
25. Sharp J, Keefe B. Psychiatry in chronic pain: a review and update. Curr Psychiatry Rep. 2005;7(3):213-219. PubMed doi:10.1007/s11920-005-0056-x
26. Gallagher RM. Management of neuropathic pain: translating mechanistic advances and evidence-based research into clinical practice. Clin J Pain. 2006;22(1 suppl):S2-S8. PubMed
27. Wolfe GI, Trivedi JR. Painful peripheral neuropathy and its nonsurgical treatment. Muscle Nerve. 2004;30(1):3-19. PubMed doi:10.1002/mus.20057
28. Sullivan MD, Robinson JP. Antidepressant and anticonvulsant medication for chronic pain. Phys Med Rehabil Clin N Am. 2006;17(2):381-400, vi-vii. PubMed
29. Jann MW, Slade JH. Antidepressant agents for the treatment of chronic pain and depression. Pharmacotherapy. 2007;27(11):1571-1587. PubMed doi:10.1592/phco.27.11.1571
30. Chong MS, Brandner B. Neuropathic agents and pain: new strategies. Biomed Pharmacother. 2006;60(7):318-322. PubMed doi:10.1016/j.biopha.2006.06.016
31. Stallworth JR, Jerrell JM, Tripathi A. Cost-effectiveness of hydroxyurea in reducing the frequency of pain episodes and hospitalization in pediatric sickle cell disease. Am J Hematol. 2010;85(10):795-797. PubMed doi:10.1002/ajh.21772
32. Jerrell JM. Adverse events associated with psychotropic treatment in African American children and adolescents. J Natl Med Assoc. 2010;102(5):375-383. PubMed
33. Shankar SM, Arbogast PG, Mitchel E, et al. Medical care utilization and mortality in sickle cell disease: a population-based study. Am J Hematol. 2005;80(4):262-270. PubMed doi:10.1002/ajh.20485
34. Grosse SD, Boulet SL, Amendah DD, et al. Administrative data sets and health services research on hemoglobinopathies: a review of the literature. Am J Prev Med. 2010;38(suppl):S557-S567. PubMed doi:10.1016/j.amepre.2009.12.015
35. Hennessy S, Leonard CE, Freeman CP, et al. Validation of diagnostic codes for outpatient-originating sudden cardiac death and ventricular arrhythmia in Medicaid and Medicare claims data. Pharmacoepidemiol Drug Saf. 2010;19(6):555-562. PubMed doi:10.1002/pds.1869
36. Chibnik LB, Massarotti EM, Costenbader KH. Identification and validation of lupus nephritis cases using administrative data. Lupus. 2010;19(6):741-743. PubMed doi:10.1177/0961203309356289
37. Labbé E, Herbert D, Haynes J. Physicians’ attitude and practices in sickle cell disease pain management. J Palliat Care. 2005;21(4):246-251. PubMed
38. Weissman DE, Haddox JD. Opioid pseudoaddiction: an iatrogenic syndrome. Pain. 1989;36(3):363-366. PubMed doi:10.1016/0304-3959(89)90097-3