Objective: To provide general practitioners with a comparison of major depressive disorder treatments received in primary care and psychiatric clinic settings, a focus on treatment outcomes related to currently prescribed antidepressants, and a review of new and emerging therapeutic strategies.
Data Sources: English-language evidence-based guidelines and peer-reviewed literature published between January 1, 2005, and December 31, 2011, were identified using PubMed, MEDLINE, and EMBASE. All searches contained the terms major depressive disorder and unipolar depression, and excluded the terms bipolar disorder/manic depressive disorder. The following search terms were also included: naturalistic study, antidepressant, relapse, recurrence, residual symptoms, response, remission, sequential medication trials, and treatment-resistant depression.
Study Selection: Meta-analyses, systematic reviews, and practice guidelines were included. Bibliographies were used to identify additional articles of interest.
Data Extraction: Abstracts and articles were screened for relevance to primary care practice. Population-based studies and those involving patients treated in primary care were used whenever possible.
Data Synthesis: Achieving remission from a major depressive episode is important to improve functional outcomes and to reduce relapse and recurrence. Despite the availability of numerous antidepressants, as many as 50% of patients require treatment modifications beyond first-line therapy. Among remitters, 90% report residual symptoms that may interfere with function. Patients treated in primary care often have chronic depression (symptom duration ≥ 24 months at presentation) and medical comorbidities. These are clinical predictors of worse outcomes and require individualized attention when treatment is initiated. Antidepressants differ in efficacy, tolerability, and side effects—factors that may affect adherence to treatment.
Conclusions: Major depressive disorder is highly prevalent in primary care and is among the most common causes of loss of disability-adjusted life-years worldwide. There are few differences in clinical profiles between depressed patients in primary care and those in specialist clinics, although differences in symptoms and comorbid conditions among individual depressed patients present a challenge for the physician providing individualized treatment. The goal of treatment is remission with good functional and psychosocial outcomes. Physicians in primary care should have expertise in working with a number of current antidepressant approaches and an awareness of new and emerging treatments.
Prim Care Companion CNS Disord 2013;15(2):doi:10.4088/PCC.12r01420
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: May 30, 2012; accepted December 10, 2012.
Published online: April 11, 2013.
Corresponding author: Sidney H. Kennedy, MD, FRCPC, University Health Network, EN8-222, 200 Elizabeth St, Toronto, Ontario M5G 2C4, Canada ([email protected]).
A Review of Antidepressant Therapy in Primary Care:
Current Practices and Future Directions
ABSTRACT
Objective: To provide general practitioners with a comparison of major depressive disorder treatments received in primary care and psychiatric clinic settings, a focus on treatment outcomes related to currently prescribed antidepressants, and a review of new and emerging therapeutic strategies.
Data Sources: English-language evidence-based guidelines and peer-reviewed literature published between January 1, 2005, and December 31, 2011, were identified using PubMed, MEDLINE, and EMBASE. All searches contained the terms major depressive disorder and unipolar depression, and excluded the terms bipolar disorder/manic depressive disorder. The following search terms were also included: naturalistic study, antidepressant, relapse, recurrence, residual symptoms, response, remission, sequential medication trials, and treatment-resistant depression.
Study Selection: Meta-analyses, systematic reviews, and practice guidelines were included. Bibliographies were used to identify additional articles of interest.
Data Extraction: Abstracts and articles were screened for relevance to primary care practice. Population-based studies and those involving patients treated in primary care were used whenever possible.
Data Synthesis: Achieving remission from a major depressive episode is important to improve functional outcomes and to reduce relapse and recurrence. Despite the availability of numerous antidepressants, as many as 50% of patients require treatment modifications beyond first-line therapy. Among remitters, 90% report residual symptoms that may interfere with function. Patients treated in primary care often have chronic depression (symptom duration ≥ 24 months at presentation) and medical comorbidities. These are clinical predictors of worse outcomes and require individualized attention when treatment is initiated. Antidepressants differ in efficacy, tolerability, and side effects—factors that may affect adherence to treatment.
Conclusions: Major depressive disorder is highly prevalent in primary care and is among the most common causes of loss of disability-adjusted life-years worldwide. There are few differences in clinical profiles between depressed patients in primary care and those in specialist clinics, although differences in symptoms and comorbid conditions among individual depressed patients present a challenge for the physician providing individualized treatment. The goal of treatment is remission with good functional and psychosocial outcomes. Physicians in primary care should have expertise in working with a number of current antidepressant approaches and an awareness of new and emerging treatments.
Prim Care Companion CNS Disord
2013;15(2):doi:10.4088/PCC.12r01420
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: May 30, 2012; accepted December 10, 2012.
Published online: April 11, 2013.
Corresponding author: Sidney H. Kennedy, MD, FRCPC, University Health Network, EN8-222, 200 Elizabeth St, Toronto, Ontario M5G 2C4, Canada ([email protected]).
Unipolar depression or major depressive disorder (MDD) is one of the leading causes of disability worldwide, ranking fourth among the global causes of disease burden1 and accounting for 65.5 million disability-adjusted life-years worldwide.2 The chronic and episodic nature of MDD, along with poor psychosocial functioning,3 are important drivers of disability and economic burden.4–6
The National Comorbidity Replication Survey reported a 12-month MDD prevalence of 6.7%, with a lifetime prevalence of 16% among adults in the United States.7 Subsequent data from the National Health and Nutrition Examination Survey8 yielded a prevalence of 20% in the previous 12 months on the basis of a score ≥ 5 on the 9-item Patient Health Questionnaire (PHQ-9).9 In the same analysis,8 only 25% of patients with severe symptoms (PHQ-9 score > 20) were receiving evidence-based care, and 37% were not receiving an antidepressant or any form of psychotherapy. Moreover, the majority of adults (60%) under the age of 64 years who received antidepressant therapy had discontinued treatment in the first 6 months.10 This lack of consistent treatment is a likely contributor to depression chronicity and episode recurrence.
The Sequenced Treatment Alternatives to Relieve Depression (STAR*D) study, a large-scale effectiveness trial, demonstrated that as many as 50% of patients required treatment beyond the first-line therapy, and approximately 30% of patients failed to remit even after 4 sequential therapies,11 emphasizing the high prevalence of treatment resistance. In another population-based US study,4 15% of patients failed to remit and 35% had multiple episodes over 23 years of follow-up. Recurrence rates ranged from 40%12 to 85%.13 These results are of particular importance to clinicians, because individuals who are not treated to remission are at higher risk of recurrence.14,15 Further complications in depression treatment arise from the occurrence of residual symptoms in a substantial percentage of patients who do achieve remission.16
The purpose of this article is to review the clinical and demographic characteristics as well as treatment outcomes with current antidepressants in patients treated in primary care and psychiatric settings and to highlight best treatment practices. The relationship between symptom profiles and functional outcomes will also be considered and the potential for individualized diagnostic strategies and treatment selection will be addressed.
METHOD
English-language evidence-based guidelines and peer-reviewed literature published between January 1, 2005, and December 31, 2011, were identified using PubMed, MEDLINE, and EMBASE. All searches contained the terms major depressive disorder and unipolar depression and excluded the terms bipolar disorder/manic depressive disorder. The following search terms were also included: naturalistic study, antidepressant, relapse, recurrence, residual symptoms, response, remission, sequential medication trials, and treatment-resistant depression. Meta-analyses, systematic reviews, and practice guidelines were included. Bibliographies were used to identify additional articles of interest.
Abstracts and articles were screened for relevance to primary care practice. Population-based studies and those involving patients treated in primary care were used whenever possible. Most studies of MDD conducted in primary care practices identified in the searches evaluated process rather than the safety and efficacy of pharmacologic therapies. For that reason, results from a multiple-treatment meta-analysis of 12 new-generation antidepressants17 and the STAR*D “real-world” effectiveness study,18 in which approximately one-third of patients were treated in primary care practices, are emphasized.
RESULTS
Depression in Primary Care Versus Psychiatric Clinics
It is often assumed that depressed patients in primary care settings are less severely depressed, have a milder course of illness, and are more likely to present with fatigue or other somatic symptoms compared to patients at psychiatry clinics.19–21 However, there were few differences in demographics or symptom profiles between primary care and psychiatric clinic patients as demonstrated in the STAR*D study, wherein 42% of patients were evaluated and treated in a primary care setting.22 Sociodemographic risk factors for poor outcomes were older age, female gender, African American or Hispanic ethnicity, and receiving Medicare or Medicaid. Only 2 core symptoms—psychomotor agitation and decreased concentration—occurred significantly more often in specialty care settings.22
Baseline severity is also predictive of treatment outcome. In a prospective cohort study of over 1,300 consecutive primary care patients treated in Europe and Chile, 17% had a chronic course of depression, and 40% had at least 1 recurrence.23 Depression severity based on PHQ-9 total score was also inversely correlated with the likelihood of achieving a sustained remission.23 Symptom severity at presentation as well as response and remission rates were similar in both primary and specialty settings.18,22 Lower remission rates were associated with chronic depression (symptom duration > 24 months at presentation), as well as medical and psychiatric comorbidities.18 Difficulty stabilizing symptoms and residual symptoms were also predictive of relapse.24,25 Response and remission rates were similar in the 2 treatment settings.22 These results emphasize that effective treatment for moderate to severe depression can be provided in the primary care setting with the use of evidence-based care.
Guidelines for Evidence-Based Care
The primary goal of acute treatment is to achieve symptom remission, which is typically defined in clinical trials as a score ≤ 7 on the 17-item Hamilton Depression Rating Scale26 or ≤ 10 on the Montgomery-Asberg Depression Rating Scale.27 Beyond remission, there is robust evidence that antidepressants prevent relapse28,29 and should be prescribed for at least 1 year after a patient achieves remission30 (Table 1 and Figure 1).
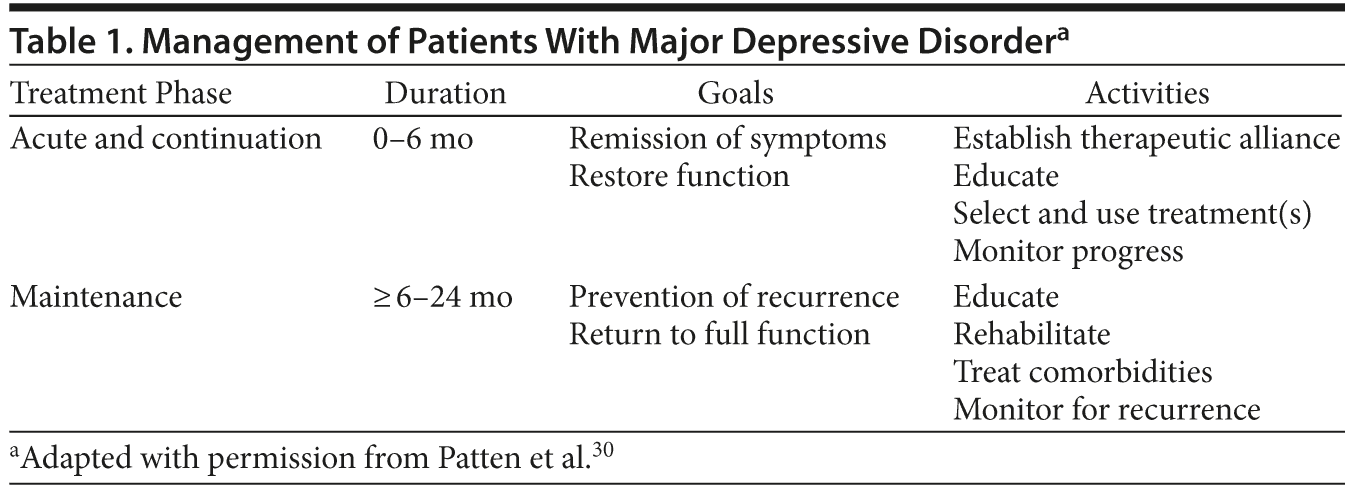
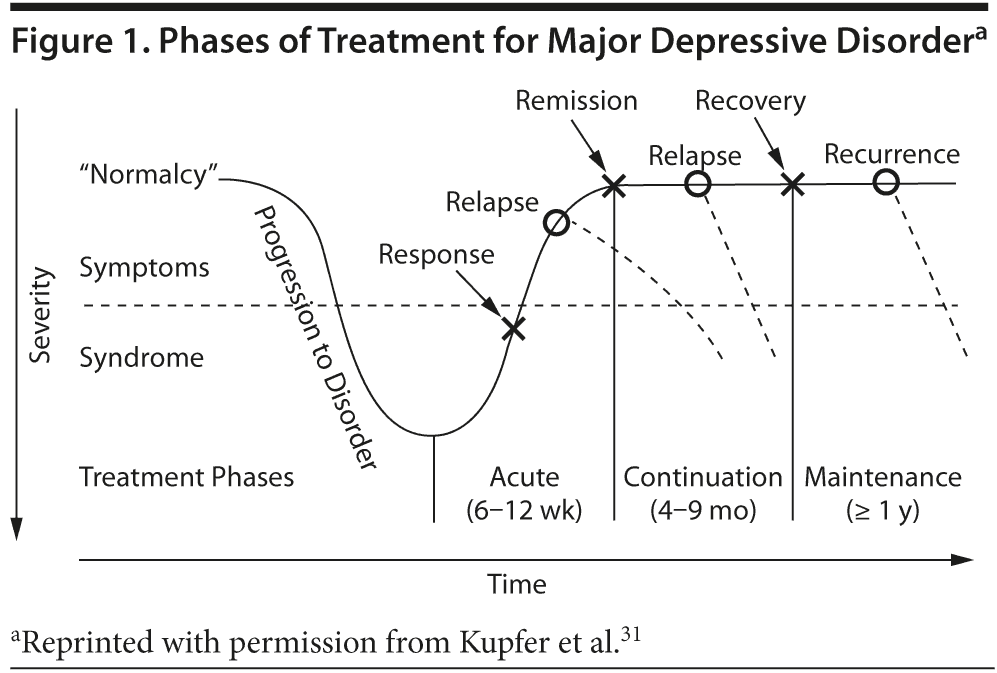
A number of international guidelines such as those of the National Institute for Health and Clinical Excellence,32 American Psychiatric Association,33 Canadian Network for Mood and Anxiety Treatments,34 and British Association for Psychopharmacology35 address the management of MDD across the treatment phases (Table 2). Although there are differences in recommendations based on depression severity, there is considerable agreement on treatment of an acute major depressive episode. All guidelines recommend psychotherapy (cognitive-behavioral therapy [CBT] or interpersonal therapy) as an option in mild-to-moderate depression. However, lack of immediate access tends to result in pharmacotherapy being the first choice of treatment for a major depressive episode. Selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), mirtazapine, and bupropion are all first-line agents for treating patients with an acute major depressive episode.
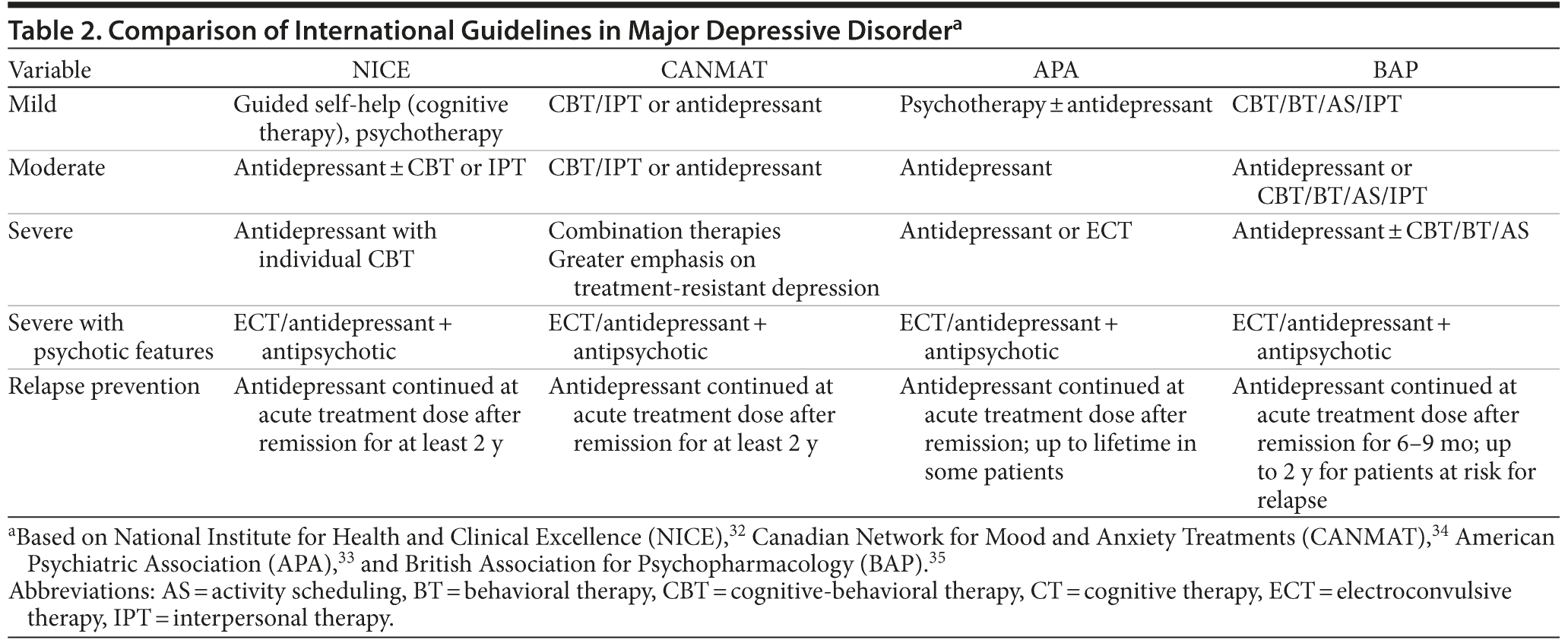
In the absence of at least a modest improvement in symptom severity, defined as a reduction of ≥ 20% in total Hamilton Depression Rating Scale26 score, optimizing acute treatment by increasing the dose of the first-line medication is usually the first strategy. Switching to another drug within the same class or to a different class or adopting an “add-on strategy,” typically with an augmentation agent (eg, lithium or an atypical antipsychotic) or with a second antidepressant, are also recommended.
Once remission is achieved, treatment should be maintained at the same dose for at least 1 year. Maintenance therapy with drug treatment and/or evidence-based psychotherapies is recommended for a longer duration (24–48 months) for individuals who have ≥ 3 risk factors for recurrence (Table 3).34 During follow-up visits, physicians should ask about residual symptoms, drug-related side effects, and treatment adherence as well as functional and psychosocial outcomes.

Efficacy and Effectiveness Across Antidepressants
Evidence from efficacy trials. There is considerable controversy as to whether current first-line antidepressants have similar efficacy and safety/tolerability profiles. In a large meta-analysis of almost 26,000 patients in 117 antidepressant trials, antidepressants differed in both efficacy (measured as response rates) and tolerability (measured as discontinuation rates)17 (Figure 2). Response rates after acute treatment for 8 weeks were 25% to 40% higher with mirtazapine, escitalopram, venlafaxine, or sertraline compared with duloxetine, fluoxetine, fluvoxamine, paroxetine, or reboxetine.17 Escitalopram and sertraline, both SSRIs, had significantly lower discontinuation rates than duloxetine, fluvoxamine, paroxetine, reboxetine (not available in the United States or Canada), and venlafaxine.17 On the basis of these findings, escitalopram and sertraline provided the best balance between efficacy and tolerability. In addition, the authors of a recently updated Agency for Health Care Research and Quality report concluded that there were no clinically significant differences among antidepressants in acute, continuation, or maintenance phases of MDD treatment but identified individual differences in head-to-head comparisons, including superiority of escitalopram over citalopram and sertraline over fluoxetine.36
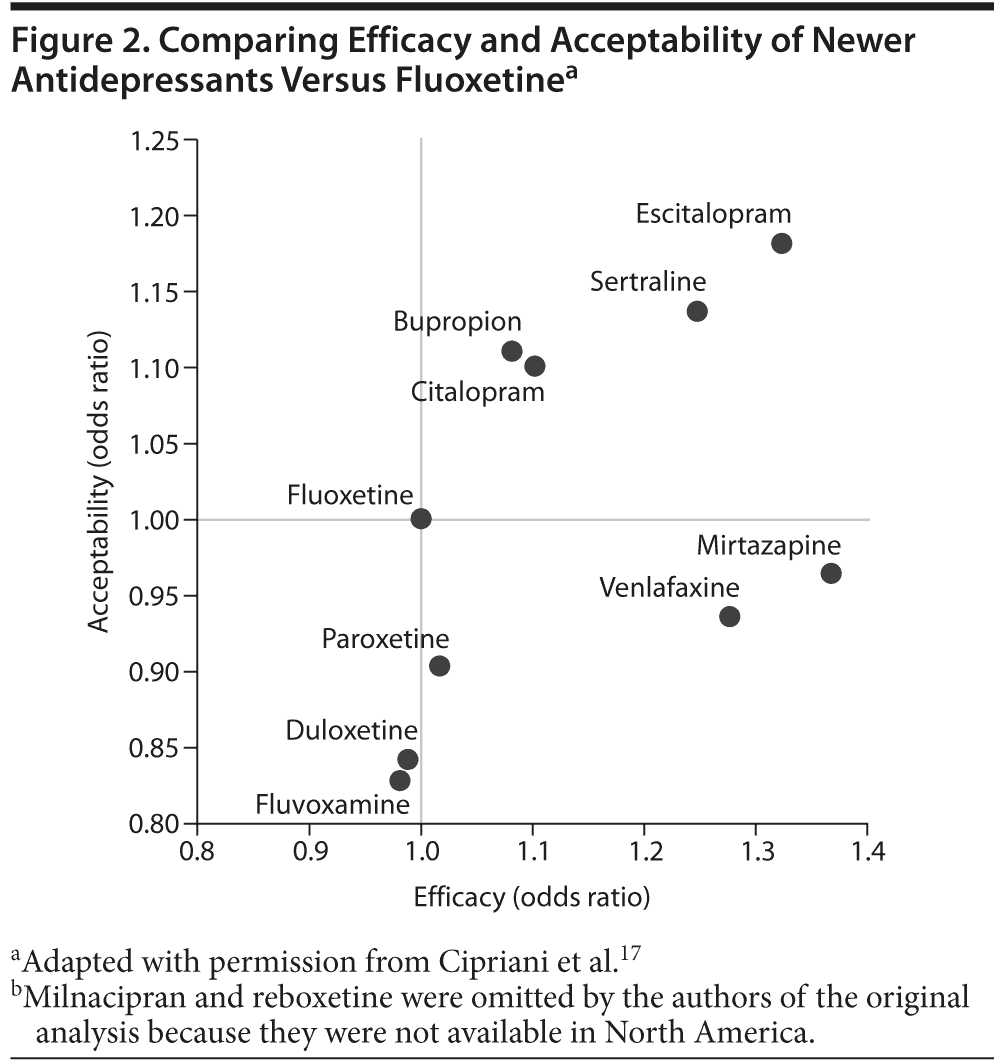
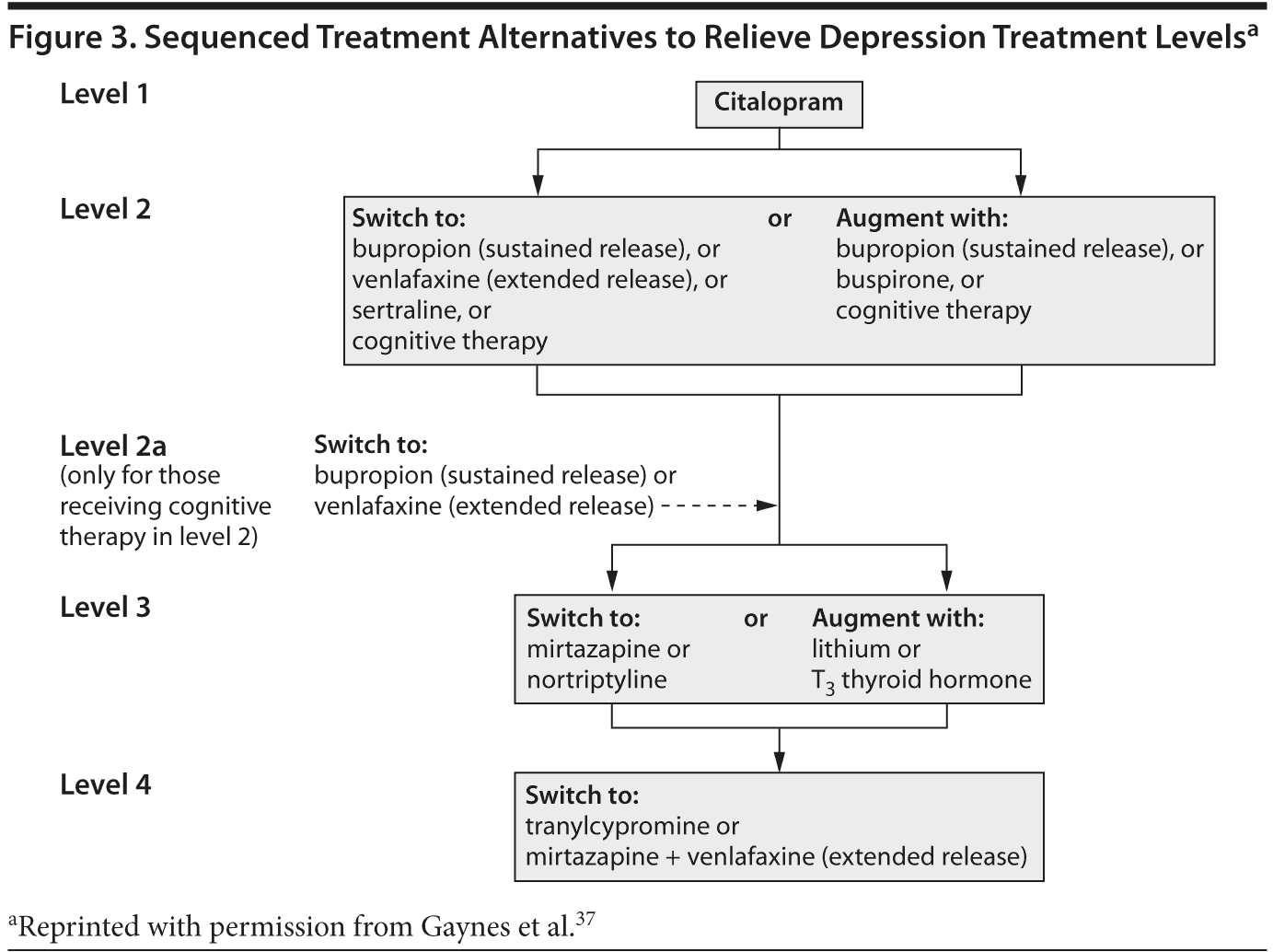
Evidence from effectiveness trials. The STAR*D trial evaluated effectiveness in a 4-phase sequential approach (Figure 3) to treating patients in both psychiatric outpatient and primary care clinics. During the first phase of treatment with citalopram,18,25,38 the response rate was 47% and the remission rate was 33% based on the Quick Inventory of Depressive Symptomatology. Options for nonremitters in step 2 were switching to another SSRI (sertraline), an SNRI (venlafaxine), or bupropion. Regardless of the option selected, the remission and response rates were similar.38,39 Patients could also switch to CBT monotherapy or augment the citalopram regimen with CBT. Switching to CBT achieved a similar remission rate as switching to a different drug therapy, although it took a longer time to reach remission with CBT augmentation compared to drug augmentation.40 In summary, switching within the SSRI class (citalopram to sertraline) was no less effective than switching to a different class of antidepressant (citalopram to venlafaxine or bupropion), and there were no significant differences in response or remission rates for patients who progressed to step 3, which involved augmenting step 2 therapy with either bupropion sustained release or buspirone. Overall, the treatments were well tolerated, although approximately 20% of patients discontinued treatment at each step. There were no significant differences in rates of adverse events across the pharmacologic treatment options except when buspirone add-on therapy was associated with a higher incidence of intolerance than bupropion (29% vs 13%, respectively).39 This trial reaffirmed the importance of treating to remission in the acute phase, as nonremitters and partial remitters (regardless of treatment) were more likely to relapse, and relapses occurred sooner in nonremitters than in remitters.
Concurrent Combination Therapies
In contrast to the STAR*D approach involving sequential combination strategies, several investigators have evaluated the benefits of coinitiating treatment with 2 antidepressants from the outset rather than waiting to add a second agent if the first treatment fails. In a relatively small proof-of-concept trial,41 2 types of concurrent combined antidepressant therapy were compared with SSRI monotherapy. Although response rates did not differ among the groups, remission rates were significantly higher for mirtazapine plus fluoxetine (52%) and mirtazapine plus venlafaxine (58%) compared with fluoxetine alone (25%). When placebo was substituted for one of the paired agents during a double-blind discontinuation phase, relapse occurred in approximately 40% of patients. In a previous 6-week trial, the same group reported remission rates of 19% with mirtazapine alone, 26% with paroxetine alone, and 43% with the combination.42 However, the Combining Medications to Enhance Depression Outcomes trial38 did not confirm an advantage for concurrent combined antidepressant therapy. The investigators evaluated outcomes with escitalopram plus placebo, escitalopram plus bupropion, and venlafaxine plus mirtazapine and found no difference in response or remission rates in acute or continuation phases of the treatment.38 The reasons for these disparities are not yet understood but warrant investigation.
Residual Symptoms
The presence and importance of residual symptoms, even in remitted patients, are frequently overlooked. In a study of patients who attained remission while taking fluoxetine, more than 90% had residual symptoms.43 Sleep disturbance, fatigue, and anhedonia were the most prevalent residual symptoms, which affect neurocognition and function. In the STAR*D trial, a similar proportion of patients who remitted while taking citalopram (90%) had at least 1 residual symptom—most frequently sleep or appetite/weight disturbance.44 In contrast to the previously cited fluoxetine study,43 residual symptoms were predictive of relapse.44
Neurocognition. There is considerable debate as to whether neurocognitive impairment in depression precedes depressive episodes or whether recurrent episodes are associated with a neurodegenerative process. Neuroimaging studies suggest that repeated episodes are associated with hippocampal volume reduction and neuropsychological impairment,45 particularly impaired memory, psychomotor, and executive functions, as well as fatigue. These symptoms are common in patients with MDD and frequently persist after mood symptoms remit.46 In a meta-analysis of antidepressant effects in neurocognitive function, it was concluded that current first-line antidepressants in general do not impair neurocognition, but methodological shortcomings, including small sample sizes in most studies, limit conclusions.47 In contrast, there were residual deficits in sustained and selective attention, memory, and executive function in a subsequent meta-analytic study of remitted patients with MDD,48 and in a 3-year prospective study of depressed patients in primary care, cognitive symptoms, lack of energy, and sleep disturbances were present 39% to 44% of the time during remission.49
Functional outcomes. Although functional impairment is implicit in the criteria for a major depressive episode, relatively few clinical trials report changes in functional outcomes despite the availability of several brief validated scales.50 The Sheehan Disability Scale51 is a brief 3-item self-report instrument designed to evaluate the impact of depression on an individual’s work, leisure, and family life. Several studies comparing antidepressants show differences in functional outcomes despite equivalent efficacy on symptom scales. For example, in a comparison of escitalopram and duloxetine over 24 weeks,52 remission rates were comparable (73% and 70%, respectively), but there were significant differences in favor of escitalopram on the Sheehan Disability Scale.
The relationship between treating to remission and restoring quality of life is illustrated in a study of primary care patients receiving antidepressant treatment.53 After 56 days of treatment, remitters (Montgomery-Asberg Depression Rating Scale score ≤ 12) had significantly better Euro Quality of Life 5-D scores than responders who did not remit and nonresponders. Results of the STAR*D trial showed similar correspondence between remission and improved health-related quality of life.18 These results have particular relevance to the emerging literature on patient self-report outcomes that emphasize the restoration of function and positive affect (Table 4).

Relapse Prevention
There is robust evidence that antidepressant maintenance treatment prevents relapse. Typically, relapse prevention trials involve treating patients to remission and randomizing half to stay on the antidepressant while the other half receives placebo. Most trials evaluate the percentage of survivors who remain in remission over 6 to 24 months. In a meta-analysis of more than 30 randomized trials,28 continuing treatment with an antidepressant reduced the odds of relapse by 70%. The relapse rate was 41% among patients receiving placebo and 18% among those who continued to receive an antidepressant.28 These findings were confirmed in an updated meta-analysis of relapse prevention antidepressant trials.29
Other relapse prevention trials with venlafaxine,55 duloxetine,56 and escitalopram57 have demonstrated significantly reduced relapse rates and low discontinuation rates due to adverse events. Another analysis from the extended Prevention of Recurrent Episodes of Depression with Venlafaxine Extended Release for Two Years trial demonstrated similar relapse prevention and discontinuation rates between venlafaxine extended release and fluoxetine,58 and superiority of venlafaxine extended release over placebo after 1 and 2 years.55
Treatment-Resistant Depression
The current definition of treatment-resistant depression requires that 2 or more first-line antidepressants prescribed at adequate doses for an adequate duration fail to produce a response. When treatment-resistant depression is suspected, misdiagnosis (eg, bipolar disorder), comorbid diagnoses (eg, substance abuse disorder), and nonadherence should first be ruled out.58 There is no consensus on optimal next steps, although augmentation and combination strategies are routinely used.34 Lithium is an established first-line augmentation strategy59 despite limited information about its efficacy and safety in combination with SSRIs or SNRIs. Triiodothyronine (T3) appears to have similar efficacy to lithium with better tolerability.60 Adjunctive methylphenidate improved apathy and fatigue in patients with treatment-resistant MDD but did not significantly reduce depression scores over 5 weeks.61 Nevertheless, a systematic review of 32 trials found limited and inconsistent results for most strategies.62 The most convincing evidence for augmentation strategies comes from several atypical antipsychotic studies.63 Aripiprazole is now indicated in the United States as an adjunctive treatment for MDD on the basis of superiority to placebo with low rates of adverse event discontinuation (< 10%),64 and olanzapine in combination with fluoxetine is approved for treatment-resistant depression.65 However, the improvement in depressive symptoms comes at the cost of potentially severe neuroendocrine, metabolic, and extrapyramidal side effects.63
Adherence and Side Effects
In a review of adherence and persistence in patients prescribed branded and generic SSRIs and SNRIs, 30% of patients had discontinued treatment regardless of the drug prescribed within the first month.66 At 12 months, adherence rates were 25% for SSRIs, 34% for venlafaxine extended release, and 38.1% for duloxetine, and persistence ranged from 129.1 days to 158.5 days.66 These findings are consistent with an analysis performed in Quebec, Canada, wherein more than 60% of patients were found to be nonpersistent with antidepressants after 6 months.10
The reasons for nonadherence and nonpersistence can be diverse; however, physicians may underestimate the incidence of side effects and their impact on a patient’s adherence to treatment.67 One approach to increasing patient-reported side effects is to use a patient checklist. In a recent trial, patients endorsed 20 times more side effects than were recorded on physician clinical notes, and about half described at least 1 side effect as very or extremely troubling.67 By understanding the patient view of side effects, treatment choices can better meet the needs of individual patients.
Weight gain and sexual dysfunction are most concerning to patients during antidepressant maintenance therapy.68 Although weight gain during SSRI or SNRI treatment is moderate overall, excessive weight gain is most likely to occur with mirtazapine and paroxetine.69,70 Rates of sexual side effects associated with SSRIs and SNRIs range from 25% to 73%, although lower rates in the range of 10% to 25% are associated with bupropion and trazodone.71,72 For bupropion, the difference is thought to be related to higher available dopamine levels, and trazodone blocks the activation of serotonin 5-HT2 receptors that are responsible for sexual side effects with SSRIs and SNRIs.73 In general, agents that are 5-HT2A and 5-HT2C antagonists74–76 and 5-HT1A agonists77 have favorable sexual profiles.
Individualizing Antidepressant Therapy:
Future Directions
While the overall effect size for antidepressant response in the acute phase of treatment is modest,78 there are several publications showing distinct response trajectories differentiating responders and nonresponders to a range of antidepressants.58,79 The challenge for researchers and clinicians is to identify markers of response at the earliest possible stage of treatment.
Clinical subtypes (eg, melancholic vs atypical) or profiles (symptom severity, childhood trauma) have limited utility in individualizing treatment strategies. While research has demonstrated that depression with seasonal patterns responds to light therapy and psychotic depression benefits from the use of atypical antipsychotics, there are few other examples of how clinical subtypes have led to distinctive treatment pathways.
Biomarkers also have not been helpful at the individual patient level to identify diagnostic subgroups or to select specific treatments. Neuroimaging offers an opportunity to examine neural pathways in patients at rest and in association with emotional challenges (eg, positive or negatively valenced pictures, words, faces) and to identify differences in brain responses. McCabe et al80 demonstrated differences in how brain circuits associated with pleasure react to pictures and the taste of chocolate with an SSRI or a norepinephrine-acting antidepressant. Furthermore, data arising from the fusion of structural and functional brain imaging in patients with recurrent episodes of depression support the role of certain regions (eg, hippocampus, orbitofrontal cortex, subgenual cingulate) in the pathophysiology of depression and risk of recurrence.45,81
Other advances have come from the field of molecular psychiatry, wherein a combination of biomarkers reflecting monoamine neurotransmitters, neurotrophic factors, and inflammatory markers is able to distinguish patients with MDD from healthy controls.82 Although it is unlikely that any one of these putative biomarkers will identify unique subpopulations or predict treatment response, integrated biomarkers involving neuroimaging, proteomic, and genomic data, as well as clinical profiles, may help to individualize treatment strategies.83
Antidepressant Development
The discussion so far has focused on identifying and applying currently accepted approaches to the use of first-line antidepressants, specifically dose optimization and switching strategies. However, there is growing support to explore mechanisms that go beyond the classical serotonin transporter and/or norepinephrine transporter targets.84 There is also evidence that genetic factors related to patient susceptibility to MDD may correlate with treatment response85 and potentially improve prevention, diagnosis, and treatment. New therapeutic targets include brain regions regulating circadian rhythms, the immune system,86–88 and neurotrophins.89–91 In addition, N-methyl-d-aspartate antagonists to target stress-related perturbation of the hypothalamic pituitary axis are currently being explored.92–95 Moreover, a serotonin reuptake stimulator (tianeptine) has shown efficacy in MDD and is currently marketed for that indication in Europe and Asia, although this drug appears to act primarily as a glutamatergic agent.96
New molecular entities that target multiple receptors are in development and potentially would obviate the need for using multiple agents. Lu AA21004 (vortioxetine) has an affinity for the 5-HT1A, 5-HT1B, 5-HT3, and 5-HT7 receptors, as well as the serotonin transporter.97 Preliminary data from pivotal trials have recently been published.98,99 Triple reuptake inhibitors that act at serotonin, norepinephrine, and dopaminergic transporters also are being studied.100
SUMMARY AND CONCLUSIONS
Depressed patients with MDD seen in primary care tend to have sociodemographic and clinical features placing them at high risk for incomplete response and recurrence or relapse. This risk can be reduced by ensuring adequate dosing of antidepressant treatment during acute and maintenance therapy. Patient-directed treatment goals should be geared to selecting appropriate treatment and achieving remission. Patients should not continue on any antidepressant at the same dose for more than 4 weeks if there is no evidence of at least partial symptomatic improvement. Primary care physicians should have experience prescribing several current first-line antidepressants and several add-on strategies. The emphasis during maintenance treatment is on adherence and psychosocial and functional rehabilitation, as well as maintaining symptom remission. To maximize remission in the real world, individualized patient treatment is an important research avenue to improve antidepressant selection. New antidepressant therapies with novel mechanisms that limit unwanted side effects will offer alternative therapeutic options to treat depressed patients in primary care as well as specialist settings.
Drug names: bupropion (Wellbutrin, Aplenzin, and others), buspirone (BuSpar and others), citalopram (Celexa and others), duloxetine (Cymbalta), escitalopram (Lexapro and others), fluoxetine (Prozac and others), fluvoxamine (Luvox and others), lithium (Lithobid and others), methylphenidate (Focalin, Daytrana, and others), mirtazapine (Remeron and others), olanzapine (Zyprexa), paroxetine (Paxil, Pexeva, and others), sertraline (Zoloft and others), trazodone (Oleptro and others), venlafaxine (Effexor and others).
Author affiliation: University Health Network, University of Toronto, Ontario, Canada.
Potential conflicts of interest: Dr Kennedy has served as a consultant to AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen-Ortho, Lundbeck, Pfizer, Servier, and St Jude Medical; has received grant/research support from AstraZeneca, Brain Cells Inc, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen-Ortho, Lundbeck, and St Jude Medical; and has served on the speakers or advisory boards of AstraZeneca, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen-Ortho, Lundbeck, Servier, and St Jude Medical.
Funding/support: This review was sponsored and funded by the Takeda Pharmaceutical Company, Ltd, Osaka, Japan, as part of a joint clinical development program with H. Lundbeck A/S, Deerfield, Illinois.
Acknowledgments: The author thanks Ann Sherwood, PhD, and The Medicine Group, Hope, Pennsylvania, who provided a preliminary outline and literature search, which was sponsored and funded by Takeda Pharmaceutical Company, Ltd as part of a joint clinical development program with H. Lundbeck A/S. Dr Sherwood reports no other conflicts of interest related to the subject of this article.
REFERENCES
1. Lopez AD, Mathers CD, Ezzati M, et al. Global and regional burden of disease and risk factors, 2001: systematic analysis of population health data. Lancet. 2006;367(9524):1747–1757. doi:10.1016/S0140-6736(06)68770-9 PubMed
2. Collins PY, Patel V, Joestl SS, et al; Scientific Advisory Board and the Executive Committee of the Grand Challenges on Global Mental Health. Grand challenges in global mental health. Nature. 2011;475(7354):27–30. doi:10.1038/475027a PubMed
3. Langlieb AM, Guico-Pabia CJ. Beyond symptomatic improvement: assessing real-world outcomes in patients with major depressive disorder. Prim Care Companion J Clin Psychiatry. 2010;12(2): e1–e14. 10.4088/PCC.09r00826blu PubMed
4. Eaton WW, Shao H, Nestadt G, et al. Population-based study of first onset and chronicity in major depressive disorder. Arch Gen Psychiatry. 2008;65(5):513–520. doi:10.1001/archpsyc.65.5.513 PubMed
5. Simon GE, Revicki D, Heiligenstein J, et al. Recovery from depression, work productivity, and health care costs among primary care patients. Gen Hosp Psychiatry. 2000;22(3):153–162. doi:10.1016/S0163-8343(00)00072-4 PubMed
6. Simon GE, Khandker RK, Ichikawa L, et al. Recovery from depression predicts lower health services costs. J Clin Psychiatry. 2006;67(8):1226–1231. doi:10.4088/JCP.v67n0808 PubMed
7. Kessler RC, Chiu WT, Demler O, et al. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):617–627. doi:10.1001/archpsyc.62.6.617 PubMed
8. Shim RS, Baltrus P, Ye J, et al. Prevalence, treatment, and control of depressive symptoms in the United States: results from the National Health and Nutrition Examination Survey (NHANES), 2005–2008. J Am Board Fam Med. 2011;24(1):33–38. doi:10.3122/jabfm.2011.01.100121 PubMed
9. Spitzer RL, Kroenke K, Williams JB. Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. Primary Care Evaluation of Mental Disorders. Patient Health Questionnaire. JAMA. 1999;282(18):1737–1744. doi:10.1001/jama.282.18.1737 PubMed
10. Béland SG, Tournier M, Galbaud du Fort G, et al. Economic impact of nonpersistence with antidepressant treatment in the adult population of Quebec: a comparative cost-effectiveness approach. Value Health. 2011;14(4):492–498. doi:10.1016/j.jval.2010.11.015 PubMed
11. Warden D, Rush AJ, Trivedi MH, et al. The STAR*D Project results: a comprehensive review of findings. Curr Psychiatry Rep. 2007;9(6):449–459. doi:10.1007/s11920-007-0061-3 PubMed
12. Mattisson C, Bogren M, Horstmann V, et al. The long-term course of depressive disorders in the Lundby study. Psychol Med. 2007;37(6):883–891. doi:10.1017/S0033291707000074 PubMed
13. Mueller TI, Leon AC, Keller MB, et al. Recurrence after recovery from major depressive disorder during 15 years of observational follow-up. Am J Psychiatry. 1999;156(7):1000–1006. PubMed
14. Paykel ES, Ramana R, Cooper Z, et al. Residual symptoms after partial remission: an important outcome in depression. Psychol Med. 1995;25(6):1171–1180. doi:10.1017/S0033291700033146 PubMed
15. Judd LL, Paulus MJ, Schettler PJ, et al. Does incomplete recovery from first lifetime major depressive episode herald a chronic course of illness? Am J Psychiatry. 2000;157(9):1501–1504. doi:10.1176/appi.ajp.157.9.1501 PubMed
16. Nierenberg AA, Wright EC. Evolution of remission as the new standard in the treatment of depression. J Clin Psychiatry. 1999;60(suppl 22):7–11. PubMed
17. Cipriani A, Furukawa TA, Salanti G, et al. Comparative efficacy and acceptability of 12 new-generation antidepressants: a multiple-treatments meta-analysis. Lancet. 2009;373(9665):746–758. doi:10.1016/S0140-6736(09)60046-5 PubMed
18. Trivedi MH, Rush AJ, Wisniewski SR, et al; STAR*D Study Team. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry. 2006;163(1):28–40. doi:10.1176/appi.ajp.163.1.28 PubMed
19. Williamson PS, Yates WR. The initial presentation of depression in family practice and psychiatric outpatients. Gen Hosp Psychiatry.1989;11(3):188–193; discussion 216-121.
20. Schwenk TL, Coyne JC, Fechner-Bates S. Differences between detected and undetected patients in primary care and depressed psychiatric patients. Gen Hosp Psychiatry. 1996;18(6):407–415. doi:10.1016/S0163-8343(96)00062-X PubMed
21. Cooper-Patrick L, Crum RM, Ford DE. Characteristics of patients with major depression who received care in general medical and specialty mental health settings. Med Care. 1994;32(1):15–24. doi:10.1097/00005650-199401000-00002 PubMed
22. Gaynes BN, Rush AJ, Trivedi MH, et al. Major depression symptoms in primary care and psychiatric care settings: a cross-sectional analysis. Ann Fam Med. 2007;5(2):126–134. doi:10.1370/afm.641 PubMed
23. Stegenga BT, Kamphuis MH, King M, et al. The natural course and outcome of major depressive disorder in primary care: the PREDICT-NL study. Soc Psychiatry Psychiatr Epidemiol. 2012;47(1):87–95. doi:10.1007/s00127-010-0317-9 PubMed
24. Rucci P, Frank E, Calugi S, et al. Incidence and predictors of relapse during continuation treatment of major depression with SSRI, interpersonal psychotherapy, or their combination. Depress Anxiety. 2011;28(11):955–962. doi:10.1002/da.20894 PubMed
25. Rush AJ, Warden D, Wisniewski SR, et al. STAR*D: revising conventional wisdom. CNS Drugs. 2009;23(8):627–647. PubMed
26. Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23(1):56–62. doi:10.1136/jnnp.23.1.56 PubMed
27. Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry. 1979;134(4):382–389. doi:10.1192/bjp.134.4.382 PubMed
28. Geddes JR, Carney SM, Davies C, et al. Relapse prevention with antidepressant drug treatment in depressive disorders: a systematic review. Lancet. 2003;361(9358):653–661. doi:10.1016/S0140-6736(03)12599-8 PubMed
29. Glue P, Donovan MR, Kolluri S, et al. Meta-analysis of relapse prevention antidepressant trials in depressive disorders. Aust N Z J Psychiatry. 2010;44(8):697–705. doi:10.3109/00048671003705441 PubMed
30. Patten SB, Kennedy SH, Lam RW, et al; Canadian Network for Mood and Anxiety Treatments (CANMAT). Canadian Network for Mood and Anxiety Treatments (CANMAT) clinical guidelines for the management of major depressive disorder in adults, 1: classification, burden and principles of management. J Affect Disord. 2009;117(suppl 1):S5–S14. doi:10.1016/j.jad.2009.06.044 PubMed
31. Kupfer DJ. Long-term treatment of depression. J Clin Psychiatry. 1991;52(suppl):28–34. PubMed
32. National Institute for Health and Clinical Excellence. Depression: The Treatment and Management of Depression in Adults. Leicester, UK: British Psychological Society; 2010.
33. Gelenberg AJ, Freeman MP, Markowitz JC, et al; American Psychiatric Association. Practice Guideline for the Treatment of Patients With Major Depressive Disorder. 3rd ed. Arlington, VA: American Psychiatric Association; 2010.
34. Lam RW, Kennedy SH, Grigoriadis S, et al; Canadian Network for Mood and Anxiety Treatments (CANMAT). Canadian Network for Mood and Anxiety Treatments (CANMAT) clinical guidelines for the management of major depressive disorder in adults, 3: pharmacotherapy. J Affect Disord. 2009;117(suppl 1):S26–S43. doi:10.1016/j.jad.2009.06.041 PubMed
35. Anderson IM, Ferrier IN, Baldwin RC, et al. Evidence-based guidelines for treating depressive disorders with antidepressants: a revision of the 2000 British Association for Psychopharmacology guidelines. J Psychopharmacol. 2008;22(4):343–396. doi:10.1177/0269881107088441 PubMed
36. Gartlehner G, Hansen RA, Morgan LC, et al. Comparative benefits and harms of second-generation antidepressants for treating major depressive disorder: an updated meta-analysis. Ann Intern Med. 2011;155(11):772–785. PubMed
37. Gaynes BN, Warden D, Trivedi MH, et al. What did STAR*D teach us? results from a large-scale, practical, clinical trial for patients with depression. Psychiatr Serv. 2009;60(11):1439–1445. doi:10.1176/appi.ps.60.11.1439 PubMed
38. Rush AJ, Trivedi MH, Stewart JW, et al. Combining Medications to Enhance Depression Outcomes (CO-MED): acute and long-term outcomes of a single-blind randomized study. Am J Psychiatry. 2011;168(7):689–701. doi:10.1176/appi.ajp.2011.10111645 PubMed
39. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905–1917. doi:10.1176/appi.ajp.163.11.1905 PubMed
40. Thase ME, Friedman ES, Biggs MM, et al. Cognitive therapy versus medication in augmentation and switch strategies as second-step treatments: a STAR*D report. Am J Psychiatry. 2007;164(5):739–752. doi:10.1176/appi.ajp.164.5.739 PubMed
41. Blier P, Ward HE, Tremblay P, et al. Combination of antidepressant medications from treatment initiation for major depressive disorder: a double-blind randomized study. Am J Psychiatry. 2010;167(3):281–288. doi:10.1176/appi.ajp.2009.09020186 PubMed
42. Blier P, Gobbi G, Turcotte JE, et al. Mirtazapine and paroxetine in major depression: a comparison of monotherapy versus their combination from treatment initiation. Eur Neuropsychopharmacol. 2009;19(7):457–465. doi:10.1016/j.euroneuro.2009.01.015 PubMed
43. Iovieno N, van Nieuwenhuizen A, Clain A, et al. Residual symptoms after remission of major depressive disorder with fluoxetine and risk of relapse. Depress Anxiety. 2011;28(2):137–144. doi:10.1002/da.20768 PubMed
44. Nierenberg AA, Husain MM, Trivedi MH, et al. Residual symptoms after remission of major depressive disorder with citalopram and risk of relapse: a STAR*D report. Psychol Med. 2010;40(1):41–50. doi:10.1017/S0033291709006011 PubMed
45. McKinnon MC, Yucel K, Nazarov A, et al. A meta-analysis examining clinical predictors of hippocampal volume in patients with major depressive disorder. J Psychiatry Neurosci. 2009;34(1):41–54. PubMed
46. Fava M, Graves LM, Benazzi F, et al. A cross-sectional study of the prevalence of cognitive and physical symptoms during long-term antidepressant treatment. J Clin Psychiatry. 2006;67(11):1754–1759. doi:10.4088/JCP.v67n1113 PubMed
47. Biringer E, Rongve A, Lund A. A review of modern antidepressants’ effects on neurocognitive function. Curr Psychiatry Rev. 2009;5(3):1–11.
48. Hasselbalch BJ, Knorr U, Kessing LV. Cognitive impairment in the remitted state of unipolar depressive disorder: a systematic review. J Affect Disord. 2011;134(1–3):20–31. doi:10.1016/j.jad.2010.11.011 PubMed
49. Conradi HJ, Ormel J, de Jonge P. Presence of individual (residual) symptoms during depressive episodes and periods of remission: a 3-year prospective study. Psychol Med. 2010;8:1–10. PubMed
50. Lam RW, Filteau MJ, Milev R. Clinical effectiveness: the importance of psychosocial functioning outcomes. J Affect Disord. 2011;132(suppl 1):S9–S13. doi:10.1016/j.jad.2011.03.046 PubMed
51. Sheehan DV. The Anxiety Disease. New York, NY: Charles Scribner and Sons; 1983.
52. Wade A, Gembert K, Florea I. A comparative study of the efficacy of acute and continuation treatment with escitalopram versus duloxetine in patients with major depressive disorder. Curr Med Res Opin. 2007;23(7):1605–1614. doi:10.1185/030079907X210732 PubMed
53. Sapin C, Fantino B, Nowicki ML, et al. Usefulness of EQ-5D in assessing health status in primary care patients with major depressive disorder. Health Qual Life Outcomes. 2004;2(1):20. doi:10.1186/1477-7525-2-20 PubMed
54. Zimmerman M, Ruggero CJ, Chelminski I, et al. Developing brief scales for use in clinical practice: the reliability and validity of single-item self-report measures of depression symptom severity, psychosocial impairment due to depression, and quality of life. J Clin Psychiatry. 2006;67(10):1536–1541. doi:10.4088/JCP.v67n1007 PubMed
55. Keller MB, Trivedi MH, Thase ME, et al. The Prevention of Recurrent Episodes of Depression with Venlafaxine for Two Years (PREVENT) study: outcomes from the 2-year and combined maintenance phases. J Clin Psychiatry. 2007;68(8):1246–1256. doi:10.4088/JCP.v68n0812 PubMed
56. Perahia DG, Maina G, Thase ME, et al. Duloxetine in the prevention of depressive recurrences: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2009;70(5):706–716. doi:10.4088/JCP.08m04756 PubMed
57. Kornstein SG, Bose A, Li D, et al. Escitalopram maintenance treatment for prevention of recurrent depression: a randomized, placebo-controlled trial. J Clin Psychiatry. 2006;67(11):1767–1775. doi:10.4088/JCP.v67n1115 PubMed
58. Thase ME, Gelenberg A, Kornstein SG, et al. Comparing venlafaxine extended release and fluoxetine for preventing the recurrence of major depression: results from the PREVENT study. J Psychiatr Res. 2011;45(3):412–420. doi:10.1016/j.jpsychires.2010.07.009 PubMed
59. Bauer M, Adli M, Bschor T, et al. Lithium’s emerging role in the treatment of refractory major depressive episodes: augmentation of antidepressants. Neuropsychobiology. 2010;62(1):36–42. doi:10.1159/000314308 PubMed
60. Nierenberg, AA, Fava M, Trivedi MH, et al. A comparison of lithium and T(3) augmentation following two failed medication treatments for depression: a STAR*D report. Am J Psychiatry, 2006;163(9):1519–1530; quiz 1665.
61. Ravindran AV, Kennedy SH, O’Donovan MC, et al. Osmotic-release oral system methylphenidate augmentation of antidepressant monotherapy in major depressive disorder: results of a double-blind, randomized, placebo-controlled trial. J Clin Psychiatry. 2008;69(1):87–94. doi:10.4088/JCP.v69n0112 PubMed
62. Fleurence R, Williamson R, Jing Y, et al. A systematic review of augmentation strategies for patients with major depressive disorder. Psychopharmacol Bull. 2009;42(3):57–90. PubMed
63. Papakostas GI. Managing partial response or nonresponse: switching, augmentation, and combination strategies for major depressive disorder. J Clin Psychiatry. 2009;70(suppl 6):16–25. doi:10.4088/JCP.8133su1c.03 PubMed
64. Berman RM, Marcus RN, Swanink R, et al. The efficacy and safety of aripiprazole as adjunctive therapy in major depressive disorder: a multicenter, randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2007;68(6):843–853. doi:10.4088/JCP.v68n0604 PubMed
65. Corya SA, Williamson D, Sanger TM, et al. A randomized, double-blind comparison of olanzapine/fluoxetine combination, olanzapine, fluoxetine, and venlafaxine in treatment-resistant depression. Depress Anxiety. 2006;23(6):364–372. doi:10.1002/da.20130 PubMed
66. Liu X, Chen Y, Faries DE. Adherence and persistence with branded antidepressants and generic SSRIs among managed care patients with major depressive disorder. Clinicoecon Outcomes Res. 2011;3:63–72. PubMed
67. Zimmerman M, Galione JN, Attiullah N, et al. Underrecognition of clinically significant side effects in depressed outpatients. J Clin Psychiatry. 2010;71(4):484–490. doi:10.4088/JCP.08m04978blu PubMed
68. Ginsberg LD. Impact of drug tolerability on the selection of antidepressant treatment in patients with major depressive disorder. CNS Spectr. 2009;14(suppl 12):8–14. PubMed
69. Patten SB, Williams JV, Lavorato DH, et al. Weight gain in relation to major depression and antidepressant medication use. J Affect Disord. 2011;134(1–3):288–293. doi:10.1016/j.jad.2011.06.027 PubMed
70. Serretti A, Mandelli L. Antidepressants and body weight: a comprehensive review and meta-analysis. J Clin Psychiatry. 2010;71(10):1259–1272. doi:10.4088/JCP.09r05346blu PubMed
71. Kennedy SH, Rizvi S. Sexual dysfunction, depression, and the impact of antidepressants. J Clin Psychopharmacol. 2009;29(2):157–164. doi:10.1097/JCP.0b013e31819c76e9 PubMed
72. Higgins A, Nash M, Lynch AM. Antidepressant-associated sexual dysfunction: impact, effects, and treatment. Drug Healthc Patient Saf. 2010;2:141–150. doi:10.2147/DHPS.S7634 PubMed
73. Keltner NL, McAfee KM, Taylor CL. Mechanisms and treatments of SSRI-induced sexual dysfunction. Perspect Psychiatr Care. 2002;38(3):111–116. doi:10.1111/j.1744-6163.2002.tb00665.x PubMed
74. Meyer JH, Cho R, Kennedy S, et al. The effects of single dose nefazodone and paroxetine upon 5-HT2A binding potential in humans using [18F]-setoperone PET. Psychopharmacology (Berl). 1999;144(3):279–281. doi:10.1007/s002130051004 PubMed
75. Montejo AL, Llorca G, Izquierdo JA, et al; Spanish Working Group for the Study of Psychotropic-Related Sexual Dysfunction. Incidence of sexual dysfunction associated with antidepressant agents: a prospective multicenter study of 1022 outpatients. J Clin Psychiatry. 2001;62(suppl 3):10–21. PubMed
76. Taylor MJ, Rudkin L, Hawton K. Strategies for managing antidepressant-induced sexual dysfunction: systematic review of randomised controlled trials. J Affect Disord. 2005;88(3):241–254. doi:10.1016/j.jad.2005.07.006 PubMed
77. Clayton AH, Dennerstein L, Pyke R, et al. Flibanserin: a potential treatment for hypoactive sexual desire disorder in premenopausal women. Womens Health (Lond Engl). 2010;6(5):639–653. doi:10.2217/whe.10.54 PubMed
78. Undurraga J, Baldessarini RJ, Valentí M, et al. Bipolar depression: clinical correlates of receiving antidepressants. J Affect Disord. 2012;139(1):89–93. doi:10.1016/j.jad.2012.01.027 PubMed
79. Gueorguieva R, Mallinckrodt C, Krystal JH. Trajectories of depression severity in clinical trials of duloxetine: insights into antidepressant and placebo responses. Arch Gen Psychiatry. 2011;68(12):1227–1237. doi:10.1001/archgenpsychiatry.2011.132 PubMed
80. McCabe C, Mishor Z, Cowen PJ, et al. Diminished neural processing of aversive and rewarding stimuli during selective serotonin reuptake inhibitor treatment. Biol Psychiatry. 2010;67(5):439–445. doi:10.1016/j.biopsych.2009.11.001 PubMed
81. Koolschijn PC, van Haren NE, Lensvelt-Mulders GJ, et al. Brain volume abnormalities in major depressive disorder: a meta-analysis of magnetic resonance imaging studies. Hum Brain Mapp. 2009;30(11):3719–3735. doi:10.1002/hbm.20801 PubMed
82. Papakostas GI. Surrogate markers of treatment outcome in major depressive disorder. Int J Neuropsychopharmacol. 2012;15(6):841–854. doi:10.1017/S1461145711001246 PubMed
83. Kennedy SH, Downar J, Evans KR, et al. The Canadian Biomarker Integration Network in Depression (CAN-BIND): advances in response prediction. Curr Pharm Des. 2012;18(36):5976–5989. PubMed
84. Rizvi SJ, Kennedy SH. Emerging drugs for major depressive disorder: an update. Expert Opin Emerg Drugs. 2012;17(3):285–294. doi:10.1517/14728214.2012.681301 PubMed
85. Crisafulli C, Fabbri C, Porcelli S, et al. Pharmacogenetics of antidepressants. Front Pharmacol. 2011;2:6. doi:10.3389/fphar.2011.00006 PubMed
86. Akhondzadeh S, Jafari S, Raisi F, et al. Clinical trial of adjunctive celecoxib treatment in patients with major depression: a double blind and placebo controlled trial. Depress Anxiety. 2009;26(7):607–611. doi:10.1002/da.20589 PubMed
87. Maas DW, Westendorp RG, Willems JM, et al. TNF-α antagonist infliximab in the treatment of depression in older adults: results of a prematurely ended, randomized, placebo-controlled trial. J Clin Psychopharmacol. 2010;30(3):343–345. doi:10.1097/JCP.0b013e3181dcf0de PubMed
88. Müller N. COX-2 inhibitors as antidepressants and antipsychotics: clinical evidence. Curr Opin Investig Drugs. 2010;11(1):31–42. PubMed
89. Duman RS, Monteggia LM. A neurotrophic model for stress-related mood disorders. Biol Psychiatry. 2006;59(12):1116–1127. doi:10.1016/j.biopsych.2006.02.013 PubMed
90. Rantamäki T, Hendolin P, Kankaanpää A, et al. Pharmacologically diverse antidepressants rapidly activate brain-derived neurotrophic factor receptor TrkB and induce phospholipase-Cgamma signaling pathways in mouse brain. Neuropsychopharmacology. 2007;32(10):2152–2162. doi:10.1038/sj.npp.1301345 PubMed
91. Watanabe K, Hashimoto E, Ukai W, et al. Effect of antidepressants on brain-derived neurotrophic factor (BDNF) release from platelets in the rats. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34(8):1450–1454. doi:10.1016/j.pnpbp.2010.07.036 PubMed
92. Sanacora G, Kendell SF, Fenton L, et al. Riluzole augmentation for treatment-resistant depression. Am J Psychiatry. 2004;161(11):2132. doi:10.1176/appi.ajp.161.11.2132 PubMed
93. Preskorn SH, Baker B, Kolluri S, et al. An innovative design to establish proof of concept of the antidepressant effects of the NR2B subunit selective N-methyl-D-aspartate antagonist, CP-101,606, in patients with treatment-refractory major depressive disorder. J Clin Psychopharmacol. 2008;28(6):631–637. doi:10.1097/JCP.0b013e31818a6cea PubMed
94. Zarate CA Jr, Singh JB, Carlson PJ, et al. A randomized trial of an N-methyl-D-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry. 2006;63(8):856–864. doi:10.1001/archpsyc.63.8.856 PubMed
95. Phelps LE, Brutsche N, Moral JR, et al. Family history of alcohol dependence and initial antidepressant response to an N-methyl-D-aspartate antagonist. Biol Psychiatry. 2009;65(2):181–184. doi:10.1016/j.biopsych.2008.09.029 PubMed
96. McEwen BS, Chattarji S, Diamond DM, et al. The neurobiological properties of tianeptine (Stablon): from monoamine hypothesis to glutamatergic modulation. Mol Psychiatry. 2010;15(3):237–249. doi:10.1038/mp.2009.80 PubMed
97. Moore NA, Bang-Andersen B, Brennum LT, et al. P.2.b.015 Lu AA21004: a novel potential treatment for mood disorders. Eur Neuropsychopharmacol. 2008;18(suppl 4):S321. doi:10.1016/S0924-977X(08)70440-1
98. Baldwin DS, Loft H, Dragheim M. A randomised, double-blind, placebo controlled, duloxetine-referenced, fixed-dose study of three dosages of Lu AA21004 in acute treatment of major depressive disorder (MDD). Eur Neuropsychopharmacol. 2012;22(7):482–491. doi:10.1016/j.euroneuro.2011.11.008 PubMed
99. Boulenger JP, Loft H, Florea I. A randomized clinical study of Lu AA21004 in the prevention of relapse in patients with major depressive disorder. J Psychopharmacol. 2012;26(11):1408–1416. doi:10.1177/0269881112441866 PubMed
100. Prins J, Denys DA, Westphal KG, et al. The putative antidepressant DOV 216,303, a triple reuptake inhibitor, increases monoamine release in the prefrontal cortex of olfactory bulbectomized rats. Eur J Pharmacol. 2010;633(1–3):55–61. doi:10.1016/j.ejphar.2010.02.009 PubMed
This PDF is free for all visitors!


