Review of Evidence for Use of Antidepressants in Bipolar Depression
ABSTRACT
Objective: Depressive episodes predominate over the course of bipolar disorder and cause considerable functional impairment. Antidepressants are frequently prescribed in the treatment of bipolar depression, despite concerns about efficacy and risk of switching to mania. This review provides a critical examination of the evidence for and against the use of antidepressants in bipolar depression.
Data Sources: English-language peer-reviewed literature and evidence-based guidelines published between January 1, 1980, and March 2014, were identified using PubMed, MEDLINE, PsycINFO/PsycLIT, and EMBASE. All searches contained the terms antidepressants, bipolar depression, depressive episodes in bipolar disorder, and treatment guidelines for bipolar depression. Meta-analyses, randomized controlled trials, systematic reviews, and practice guidelines were included. Bibliographies from these publications were used to identify additional articles of interest.
Data Extraction: Studies involving treatment of bipolar depression with antidepressant monotherapy, adjunctive use of antidepressant with a mood stabilizer, and meta-analysis of such studies combined were reviewed.
Conclusions: The body of evidence on the use of antidepressant monotherapy to treat patients with bipolar depression is contentious, but the recommendations from evidence-based guidelines do not support antidepressant monotherapy for bipolar depression. Only when mood stabilizer or atypical antipsychotic monotherapy has failed should adjunctive treatment with an antidepressant be considered.
Prim Care Companion CNS Disord 2014;16(5):doi:10.4088/PCC.14r01653
© Copyright 2014 Physicians Postgraduate Press, Inc.
Submitted: March 10, 3014; accepted July 9, 2014.
Published online: October 16, 2014.
Corresponding author: Sidney H. Kennedy, MD, Department of Psychiatry, University Health Network, EN-8-238, 200 Elizabeth St, Toronto, Ontario, Canada M5G2C4 ([email protected]).
Antidepressants are used in up to 50% of patients with bipolar depression,1,2 despite concerns about limited efficacy and the potential risk of inducing mania.3 The 2 main questions to be addressed in this brief review are (1) Do antidepressants work in bipolar depression? and (2) Do benefits exceed risks, particularly in relation to induction of (hypo)mania and rapid cycling? This review aims to provide an evaluation of the risk-benefit ratio associated with antidepressant use in the treatment of bipolar depression that will be beneficial to the primary care physician.
Despite the emergence of mood stabilizers such as lamotrigine and quetiapine, both of which display greater antidepressant than antimanic properties, there is mixed evidence that these medications effectively treat the frequently prolonged depressive episodes experienced in the course of bipolar disorder.3,4 Justification for antidepressant usage in bipolar depression requires evidence of efficacy in the absence of significant adverse effects on the overall course of illness.5 The ISBD (International Society for Bipolar Disorders) Task Force rated the level of evidence for antidepressant monotherapy as poor (D), while the evidence for adjunctive antidepressant usage with a mood stabilizer was given a higher rating (B).6
METHOD
English-language, peer-reviewed literature and evidence-based guidelines published between January 1, 1980, and March 2014, were identified using PubMed, MEDLINE, PsycINFO/PsycLIT, and EMBASE. All searches contained the terms antidepressants, bipolar depression, depressive episodes in bipolar depression, and treatment guidelines for bipolar depression. Meta-analyses, randomized controlled trials, systematic reviews, and practice guidelines were included. Bibliographies from these publications were used to identify additional articles of interest.
Burden of Illness in Bipolar I and II Depression
Estimates of the lifetime prevalence of bipolar I and II disorder in the general population are between 3.7% and 3.9%.7,8 While the diagnosis of bipolar disorder is based on the presence of hypomania or mania, depressive symptoms and episodes predominate over the course of bipolar I and II disorder. Greater than 50% of bipolar patients present with depression as the index mood episode rather than a manic or mixed mood state,9,10 often because patients misidentify or underreport previous manic/hypomanic episodes.11 Depressive symptoms are also 3 times more prevalent than manic symptoms in bipolar disorder.12,13
Given the fact that bipolar II depression is characterized by multiple, often prolonged, depressive episodes, it can be argued that this illness is as severe as bipolar I depression.14 In addition to being longer and more persistent, the depressive phase of bipolar disorder is associated with higher rates of morbidity and mortality15 and with greater disability and decreased quality of life than any other phase of the illness.10,12,16,17 Also, a 35 times greater risk of suicide has been reported in bipolar patients during depression than during mania.18 With this degree of illness burden, it is essential that effective and appropriate treatment is provided for bipolar depression as early in the course of the illness as possible.
EVIDENCE FOR AND AGAINST ANTIDEPRESSANT USE IN BIPOLAR DEPRESSION
The modern controversy about antidepressant efficacy in bipolar depression is well illustrated by 2 studies reported in 2000 and 2001. The first study19 compared the effect of adding a second mood stabilizer (lithium carbonate or divalproex sodium) versus adding an antidepressant (paroxetine) to a preexisting mood stabilizer in patients with bipolar depression. While both groups improved, there was less discontinuation of treatment in the antidepressant-treated group.19 The second study20 compared the addition of paroxetine, imipramine, or placebo to lithium in outpatients with bipolar depression. There was no difference in efficacy across the 3 groups, but paroxetine and imipramine were superior to placebo for patients with serum lithium levels below 0.8mEq/L. In other words, when lithium levels exceeded 0.8mEq/L, neither paroxetine nor imipramine conferred additional benefit. The authors also reported a lower switch to mania in the paroxetine group compared to the imipramine group.20

- Mood stabilizers should be used as first-line treatment for bipolar depression, and adjunctive antidepressant treatment should be considered only if this strategy fails.
- Particular caution is required when considering an antidepressant if there is agitation, evidence of rapid cycling, manic episodes, prior antidepressant-induced worsening of the condition, or family history of mania.
- Mixed depressive conditions are more likely to have more episodes, rapid cycling, and hypomanic switches with antidepressant treatment compared with a unipolar illness.
Support for the efficacy and safety of antidepressants in the treatment of bipolar depression comes from a meta-analysis of 12 trials.21 While there was a 1.86 risk ratio for response to antidepressants in the 5 placebo-controlled studies that compared 1 or more antidepressants with placebo, this result should be treated with caution, as 1 large study accounted for 69% (456/662) of the total number of patients in the comparison.22 It should also be noted that, in 3 of the studies, patients received a concurrent mood stabilizer (lithium) or antipsychotic agent (olanzapine), so the comparison was not between antidepressant monotherapy and placebo.22 A much earlier report that was also included in the meta-analysis, on the use of tranylcypromine (a classical monoamine oxidase inhibitor), included both patients with anergic bipolar depression and patients with anergic major depressive disorder (MDD).23 The combined anergic sample had a risk ratio of 5.54 in favor of treatment with tranylcypromine versus placebo. Since the authors did not separate the patients with bipolar depression from the patients with MDD in their analysis, the result favoring antidepressant treatment may be accounted for by the MDD anergic group.23 The relapse rate in a cohort of bipolar I and II depressed patients treated with an antidepressant and a mood stabilizer and followed for 1 year was 70% for the antidepressant discontinuation group (less than 6 months of antidepressant treatment) compared with 36% of the antidepressant continuation group (antidepressant treatment beyond 6 months).24 In a later review of the clinical responses to antidepressants among acute patients with bipolar depression or MDD,5 the study findings indicated that antidepressants in combination with mood stabilizers yielded favorable acute clinical outcomes, with responder rates of 71% for bipolar I and 77% for bipolar II patients. These clinical outcomes were independent of mood switching and compared favorably with response rates of 61.7% for MDD, perhaps reflecting the heterogeneity of the pole of the illness in particular.5
Contrary to these reports, in the STEP-BD trial (Systematic Treatment Enhancement Program for Bipolar Disorder),25 bipolar I or II depressed patients received either adjunctive antidepressant medication (bupropion or paroxetine, n = 179) or mood stabilizer plus placebo (n = 187) treatment for up to 26 weeks. Adjunctive antidepressant treatment was not associated with increased efficacy, with 23.5% recovery (defined as 8 consecutive weeks of euthymia) for those who received a mood stabilizer plus adjunctive antidepressant as opposed to 27.3% for those who received a mood stabilizer plus placebo.25 The naturalistic trial design allowed for the inclusion of patients with coexisting anxiety disorders, substance use disorders, or psychotic symptoms, and patients could also receive additional pharmacotherapy or psychotherapy.
In a systematic review and meta-analysis of 6 randomized controlled trials (n = 1,034) examining antidepressant use in the acute treatment of bipolar depression,26 antidepressant usage was not associated with a statistically significant increase in efficacy compared with placebo or other pharmacologic treatments. Three of the trials included by Gijsman et al21 were excluded from this meta-analysis due to the inclusion of MDD patients23,27 or lack of a placebo control group.28 In 4 of the studies included by Gijsman et al,21 100% of patients received an antidepressant coadministered with a mood stabilizer (standard mood stabilizer or atypical antipsychotic), and, overall, 68% of the patients from the 6 trials received this combined treatment.
A lack of effectiveness of antidepressant medication was shown also in the EMBOLDEN (Efficacy of Monotherapy Quetiapine in Bipolar Depression) II trial,29 which included paroxetine antidepressant monotherapy (n = 122) as 1 arm of treatment for bipolar II depressed patients compared to quetiapine at either 300-mg (n = 245) or 600-mg (n = 247) dosages. Following 8 weeks of treatment, patients receiving either dose of quetiapine achieved response on the Montgomery-Asberg Depression Rating Scale, whereas paroxetine-treated patients did not.29 In a large multicenter study comparing olanzapine monotherapy (n = 351) and the combination of olanzapine with fluoxetine (n = 82) in the acute treatment of bipolar I depression,22 rates of response in the olanzapine/fluoxetine combination group (56%) were significantly greater than in the olanzapine (39%) and the placebo (30%) groups. The remission rate for the olanzapine/fluoxetine combination group was 49%, which was significantly greater than that for the olanzapine group (33%) and the placebo group (25%).22 However, this trial lacked a fluoxetine monotherapy comparator arm.
Few investigators have examined the role of antidepressant monotherapy in the treatment of bipolar depression, with the majority preferring to evaluate the addition of an antidepressant to a mood stabilizer. Amsterdam and colleagues30-33 produced 4 articles over recent years examining the role of antidepressant monotherapy. In the first of these,30 bipolar I and II depressed patients were randomized to receive fluoxetine (n = 8), olanzapine monotherapy (n = 9), olanzapine/fluoxetine combination (n = 8), or placebo (n = 9) for up to 8 weeks. All treatment groups produced response, with no difference in efficacy between treatment groups including placebo.30 Amsterdam et al31 compared venlafaxine (n = 43) and lithium (n = 40) in rapid- and non-rapid-cycling patients with bipolar II depression and found that venlafaxine had significantly greater efficacy versus lithium, with no increase in mood conversion, irrespective of rapid-cycling status. In a later small but long-term follow-up study of bipolar II patients who were in remission from a depressive episode,32 the relapse rate was lower for fluoxetine (n = 28) compared to lithium (n = 26) or placebo (n = 27) treatments, with no difference in mood conversion between groups.32 The fourth study,33 which included fluoxetine monotherapy, is discussed in the following section on treatment-emergent affective switches.
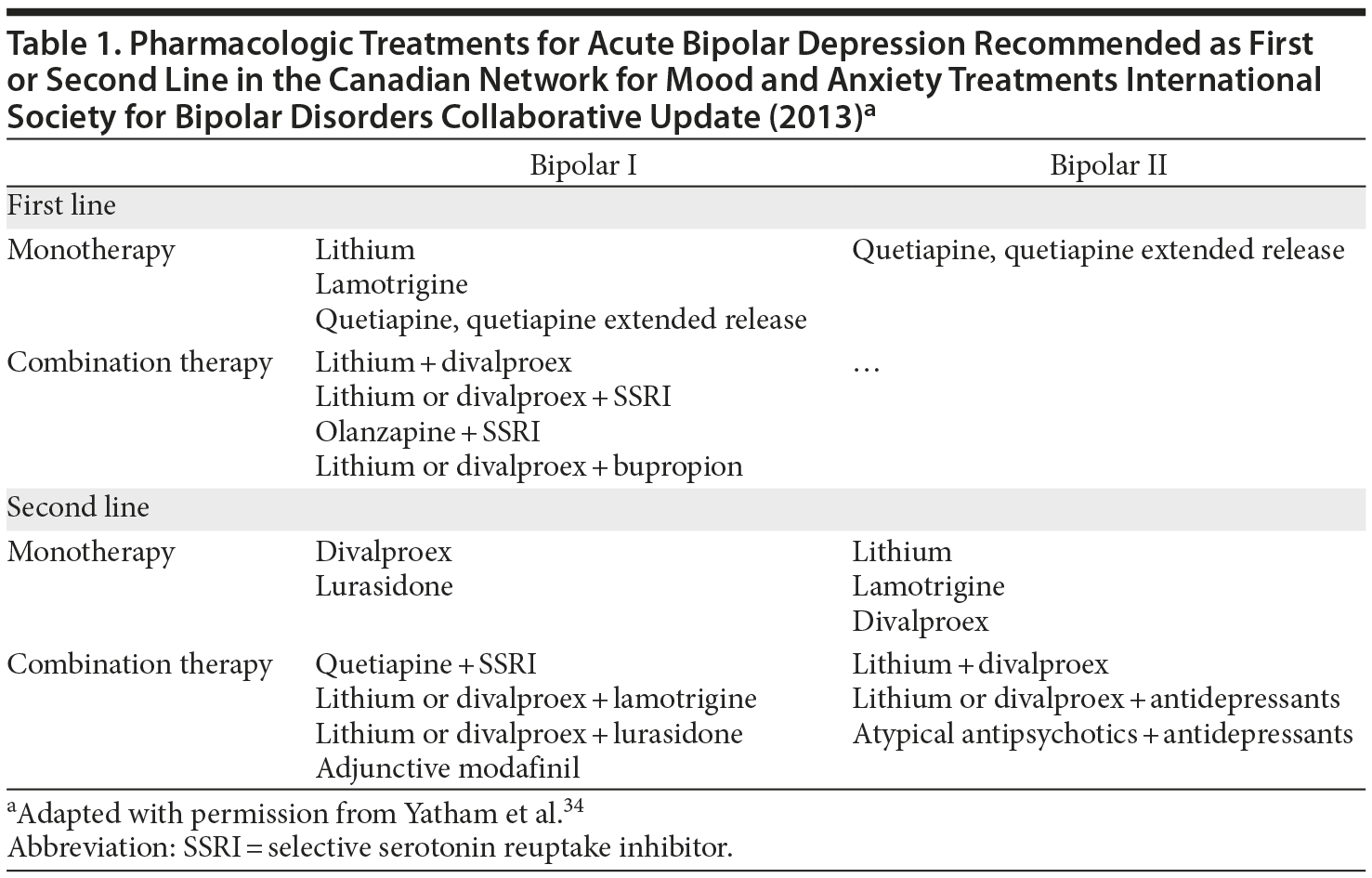
The 2013 Canadian Network for Mood and Anxiety Treatments and ISBD guidelines34 are shown in Table 1. These guidelines recommend against antidepressant monotherapy as a first-line treatment for bipolar depression and rather recommend that mood stabilizers (eg, lithium, divalproex, or lamotrigine) and antipsychotics (eg, olanzapine, quetiapine, and lurasidone) be considered as monotherapy in bipolar depression.
TREATMENT-EMERGENT AFFECTIVE SWITCHES
The risk of switch into mania is often a significant concern when treating bipolar depression with an antidepressant, and it requires careful monitoring. In a 10-week trial designed to assess the effects of 3 second-generation antidepressants (bupropion: n = 51, sertraline: n = 58, or venlafaxine: n = 65) added to mood stabilizer treatment for bipolar patients (73% bipolar I, 26% bipolar II, 1% bipolar disorder not otherwise specified), the switch rate based on a Young Mania Rating Scale (YMRS) score ≥ 13 was significantly elevated in the venlafaxine group (15%) compared to the bupropion (4%) and sertraline (7%) groups. No placebo control group was used in this study. Interestingly, with the rapid-cycling group excluded, this difference was no longer present between antidepressants.35 On the other hand, in the previously cited study of a bipolar I and II cohort,24 there was no increase in the rate of switching to mania in the antidepressant continuation group compared to the discontinuation group. There were a limited number of patients receiving tricyclic antidepressant treatment (2%) or venlafaxine (12%), with the majority taking either selective serotonin reuptake inhibitors (SSRIs) (31%) or bupropion (19%), which may account for the low switch rate into mania. The authors offered the term antidepressant discontinuation-related mania as one possible explanation for this result.24
The meta-analysis by Gijsman et al21 found that in the 6 studies that had predefined criteria for switching to mania, antidepressants did not induce more switching to mania than placebo. The rate of switching for tricyclic antidepressants was 10% compared with 3.2% for all other antidepressants combined.21 Results from 2 additional trials suggest a 4-fold switch rate for venlafaxine compared with SSRIs and bupropion when used concurrently with a mood stabilizer. In the first study,36 the rate of switch was 13% (n = 4) with venlafaxine compared to 3% (n = 1) with paroxetine. In the second study,37 the switch rate was 29% with venlafaxine compared to sertraline (9%) and bupropion (10%). The olanzapine/fluoxetine combination in the study by Tohen et al22 discussed earlier did not cause any switching into mania or hypomania.
In Sidor and Macqueen’s meta-analysis26 on the use of antidepressants in bipolar depression, the overall switch rates were not different (approximately 7%) for adjunctive antidepressant or placebo treatment. Nor was there an increased switch rate for patients treated with antidepressants compared to recognized mood stabilizers (lithium, lamotrigine, or divalproex).26 The consensus for these studies is that antidepressants with dual-action serotonergic-noradrenergic activity, such as tricyclics and venlafaxine, are more likely to induce hypomania or mania than serotonergic agents.36 They should not, therefore, be used as monotherapy, and other agents should be considered for adjunctive treatment for bipolar depression before an antidepressant is considered.
Although there is some evidence that the risk of hypomanic switch or cycle acceleration with antidepressants may be lower in bipolar II patients,38,39 this has not been consistently reported.5,40-42 Antidepressants may also induce mixed symptoms, in particular, agitation, irritability, rapid thoughts, and distractibility, which are often untreated due to lack of diagnostic clarity.43,44
Antidepressant treatment in bipolar patients is sometimes associated with new or worsening irritability or agitation termed antidepressant-associated chronic irritable dysphoria. In the STEP-BD study,45 the occurrence of irritable dysphoria was related to previous history of antidepressant-induced affective switch and female gender, rather than use of a current antidepressant.5 In addition, agitated depression in bipolar patients, which often presents with new-onset insomnia, impulsivity, and suicidality, has been associated with new antidepressant treatment.46 Mixed depressive conditions are more likely to have more episodes, rapid cycling, and hypomanic switches with antidepressant treatment than those with a unipolar illness.47 In a further study from the STEP-BD sample,48 antidepressant use was not higher among the subjects with mixed features than among those with pure depressive presentations; indeed, the total mean number of threshold-level manic symptoms was actually lower among subjects taking an antidepressant than among those not taking an antidepressant, suggesting that the presence of concomitant manic symptoms was not simply a matter of antidepressant use. Eppel49 has raised concerns that the relatively short follow-up period of 6 to 12 weeks may miss cycle acceleration and a switch into hypomania or mania, which in clinical experience is more likely to occur after 1 to 2 years. Eppel49 also raises concern about the long-term use of antidepressants in bipolar depression, as the evidence presented here suggests that, though antidepressants may produce an acute improvement in mood, this change is not sustained over the long term, and there is the risk of mood cycling in the future.
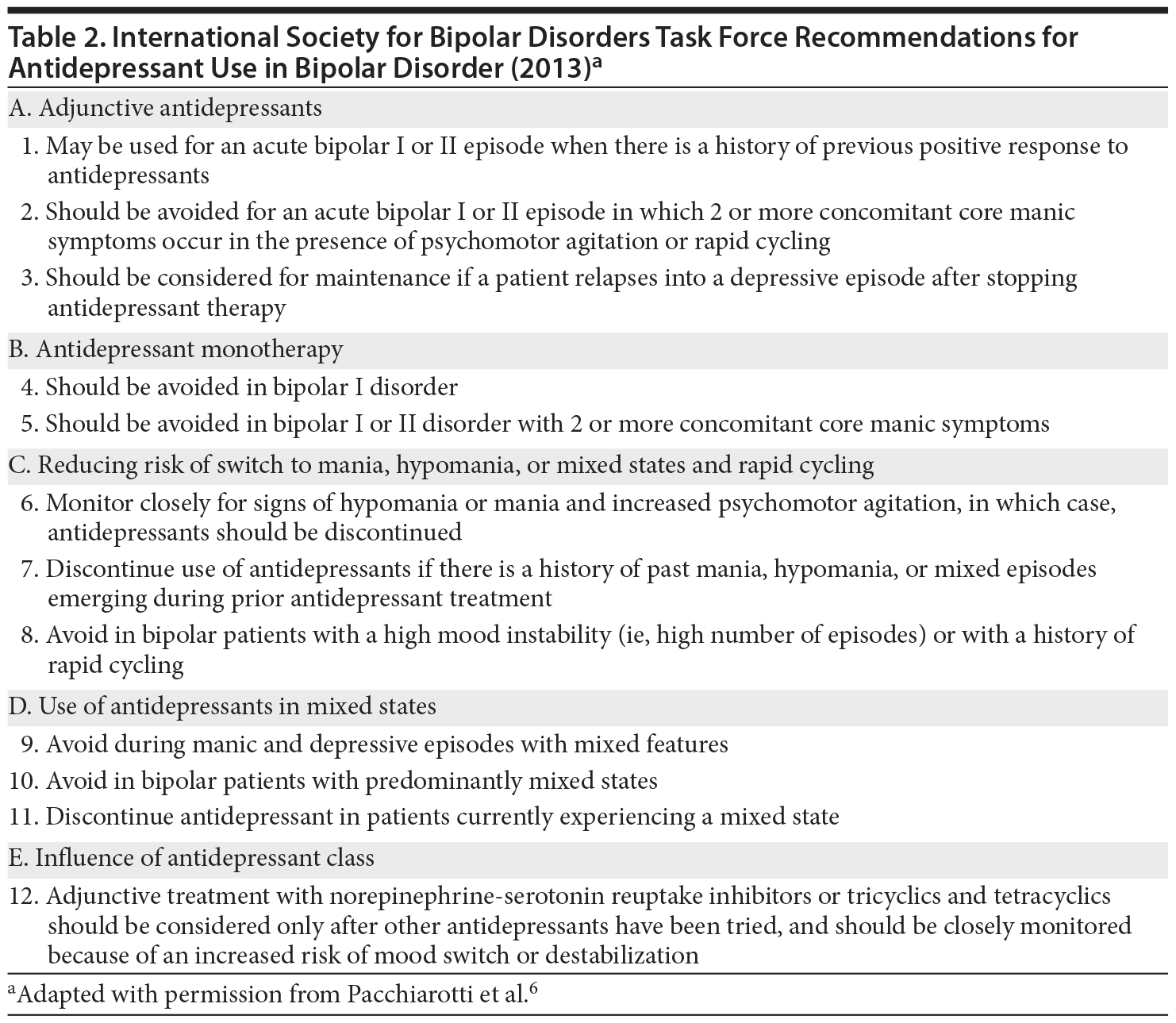
In the DSM-5, the new specifier “with mixed features” can be applied to bipolar I and II disorder and MDD. This change from DSM-IV was made to reflect the clinical phenomenon of “mixed” mood states that do not meet full criteria for a mixed episode of bipolar I, reflected by co-occurrence of full mania and MDD. To be diagnosed with the “with mixed features” specifier, a person has to meet the full criteria for 1 mood episode (depression, mania, or hypomania) and have 3 or more symptoms of the other mood pole.50 Stetka and Correll50 raise concern that classifying a major depressive episode with 3 relevant mania-like symptoms as MDD rather than a bipolar spectrum disorder may be misleading, and that treatment recommendations are unclear for patients with MDD and mixed manic or hypomanic features. A helpful summary of the ISBD Task Force recommendations for antidepressant use in bipolar depression is provided in Table 2.
Whether antidepressants can accelerate episode frequency or induce rapid cycling in bipolar patients is also a controversial issue. In 1 nonrandomized 12-month follow-up study of bipolar I and II outpatients, prior antidepressant treatment was an independent predictor of polarity changes and mixed symptoms.51 In contrast, the study by Amsterdam et al33 found no evidence for mood conversion in bipolar II patients with respect to prior rapid-cycling status irrespective of treatment with fluoxetine, lithium, or placebo.33
Three studies report an association of lifetime mixed episodes and higher rates of antidepressant use with increased risk of suicide,52-54 and, in a study of 290 patients with bipolar I or II depression,55 suicidal ideation or acts were associated with a greater number of previous antidepressant treatment trials.
ANTIPSYCHOTICS AS ANTIDEPRESSANTS IN BIPOLAR DEPRESSION
While there is modest evidence that mood stabilizers such as valproate and lamotrigine are effective in the treatment of bipolar depression, there is considerably stronger evidence to support the use of atypical antipsychotics. Efficacy of an atypical antipsychotic as monotherapy for bipolar depression offers several potential advantages compared with combination or antidepressant strategies. Documented antimanic effects, potential for reduced risk of manic relapses, and reduced risk of treatment-emergent affective switches are all benefits that might lessen the likelihood of nonadherence to treatment.56 A meta-analysis of antipsychotic agents used to treat bipolar depression57 suggests that these agents should not be grouped together as a class but rather that individual agents such as quetiapine show differential benefits. Lamotrigine had been given very favorable status with regard to monotherapy for bipolar depression, but this has also been called into question by a meta-analysis of 5 studies,58 in which efficacy in the acute treatment of bipolar depression was not demonstrated.
In contrast, the BOLDER (BipOLar DEpRession) I and II studies found that quetiapine at both 300-mg and 600-mg doses was effective as monotherapy in the treatment of bipolar depression.4,56 Similarly, in the Embolden I trial, quetiapine but not lithium-treated patients showed significant improvements in remission rates at week 8 compared to placebo.59 In EMBOLDEN II,29 quetiapine-treated patients achieved response, while paroxetine-treated patients did not. As previously mentioned, there is evidence that the olanzapine/fluoxetine combination was significantly more effective than either olanzapine monotherapy or placebo in a trial of patients with bipolar depression.22 Both olanzapine/fluoxetine combination and quetiapine have since been approved by the US Food and Drug Administration (FDA) for the treatment of bipolar depression. The World Federation of Societies of Biological Psychiatry Update 2012 on the treatment of acute bipolar depression reported that it is difficult to give a firm recommendation on the use of antidepressants as a group in bipolar depression, given the number of medications involved and the evidence from trials.60
EMERGING TREATMENTS FOR BIPOLAR DISORDER
In 2012, an atypical antipsychotic received FDA approval for treatment of bipolar depression. Lurasidone, a potent 5-HT7 antagonist, demonstrated efficacy in bipolar depression based on 2 positive double-blind, randomized, placebo-controlled, 6-week clinical trials. In PREVAIL I (PRogram to EValuate the Antidepressant Impact of Lurasidone), lurasidone was effective as adjunctive therapy (added to background treatment with lithium or valproate)60 and as monotherapy in the treatment of bipolar I depression (PREVAIL 2).62
Both pramipexole, a dopamine agonist (for treatment of bipolar II depression), and modafinil, a stimulant (in studies of bipolar I depression), have shown promise in small trials.63,64 There is also preliminary evidence that intravenous ketamine may provide a rapid response in treatment-resistant patients with bipolar depression, but the role of ketamine, especially in an oral format useful for outpatient treatment of bipolar depression, requires further research.65
CONCLUSION
We return to the 2 questions that this review sought to answer: (1) Do antidepressants work in bipolar depression? and (2) Do benefits exceed risks, particularly in relation to induction of (hypo)mania and rapid cycling?
The balance of evidence as described in this report confirms the need for caution with use of antidepressant monotherapy for bipolar depression. While there are some small trials that show favor for SSRI monotherapy, other trials and clinical consensus suggest that antidepressant monotherapy for bipolar depression should not be used. Indeed, adjunctive antidepressant treatment with a mood stabilizer should only be used when standard mood stabilizer (lithium, lamotrigine, or divalproex) or antipsychotic treatment (quetiapine, olanzapine, or lurasidone) have failed.
With regard to the benefits exceeding the risk of mood switch, the evidence presented here confirms that antidepressant use in the acute stage of bipolar depression requires a thorough history taking, including evidence of rapid cycling or severe manic episodes, history of prior antidepressant-induced worsening of the condition, and any family history of mania.66 In the longer term, pharmacotherapy for patients with bipolar disorder should focus on aggressive prevention of depressive episodes, with use of antidepressants as an available second-line option in conjunction with a mood stabilizer when other strategies have failed. Studies in this review have provided evidence that the risk of mood conversion may not actually occur in the current episode but rather lead to a lifetime risk of polarity change and mixed episodes.51-54
Drug names: bupropion (Wellbutrin, Aplenzin, and others), divalproex (Depakote and others), fluoxetine (Prozac and others), imipramine (Tofranil and others), ketamine (Ketalar and others), lamotrigine (Lamictal and others), lithium (Lithobid and others), lurasidone (Latuda), modafinil (Provigil), olanzapine (Zyprexa), olanzapine/fluoxetine (Symbyax and others), paroxetine (Paxil, Pexeva, and others), pramipexole (Mirapex and others), quetiapine (Seroquel), sertraline (Zoloft and others), tranylcypromine (Parnate and others), venlafaxine (Effexor and others).
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, bupropion, divalproex, fluoxetine, imipramine, ketamine, lamotrigine, lithium, modafinil, olanzapine, paroxetine, pramipexole, sertraline, tranylcypromine, and venlafaxine are not approved by the US Food and Drug Administration for the treatment of bipolar depression.
Author affiliations: Department of Psychiatry, University Health Network, University of Toronto (Drs McInerney and Kennedy), and Arthur Sommer-Rotenberg Chair in Suicide Studies (Dr Kennedy) and Department of Psychiatry (Dr McInerney), St Michael’s Hospital, Toronto, Ontario, Canada.
Financial disclosure: Dr McInerney receives support from a CANMAT-Pfizer Clinical Fellowship. Dr Kennedy has served as an advisor, consultant, or speaker for AA Pharma, Abbott Laboratories, AstraZeneca, Bristol-Myers Squibb, Eli Lilly, Elsevier, Forest, Glia Communications, Lundbeck, Lundbeck Institute, Pfizer, Servier, Spimaco, Symbiote Medical Communications, and Spirant Communication and has received grant/research support from Bristol-Myers Squibb, BrainCells, Canadian Institutes of Health Research, Clera, Eli Lilly, Lundbeck, Ontario Brain Institute, Pfizer, Servier, and St Jude Medical.
Funding/support: No funding support was received by Drs McInerney or Kennedy for the writing of this review.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
1. Baldessarini RJ, Leahy L, Arcona S, et al. Patterns of psychotropic drug prescription for US patients with diagnoses of bipolar disorders. Psychiatr Serv. 2007;58(1):85-91. PubMed doi:10.1176/appi.ps.58.1.85
2. Ventimiglia J, Kalali AH, McIntyre R. Treatment of bipolar disorder. Psychiatry (Edgmont). 2009;6(10):12-14. PubMed
3. Swartz HA, Thase ME. Pharmacotherapy for the treatment of acute bipolar II depression: current evidence. J Clin Psychiatry. 2011;72(3):356-366. PubMed doi:10.4088/JCP.09r05192gre
4. Calabrese JR, Keck PE Jr, Macfadden W, et al. A randomized, double-blind, placebo-controlled trial of quetiapine in the treatment of bipolar I or II depression. Am J Psychiatry. 2005;162(7):1351-1360. PubMed doi:10.1176/appi.ajp.162.7.1351
5. Tondo L, Baldessarini RJ, Vázquez G, et al. Clinical responses to antidepressants among 1036 acutely depressed patients with bipolar or unipolar major affective disorders. Acta Psychiatr Scand. 2013;127(5):355-364. PubMed doi:10.1111/acps.12023
6. Pacchiarotti I, Bond DJ, Baldessarini RJ, et al. The International Society for Bipolar Disorders (ISBD) task force report on antidepressant use in bipolar disorders. Am J Psychiatry. 2013;170(11):1249-1262. PubMed doi:10.1176/appi.ajp.2013.13020185
7. Kessler RC, Berglund P, Demler O, et al. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):593-602. PubMed doi:10.1001/archpsyc.62.6.593
8. Hirschfeld RM, Calabrese JR, Weissman MM, et al. Screening for bipolar disorder in the community. J Clin Psychiatry. 2003;64(1):53-59. PubMed doi:10.4088/JCP.v64n0111
9. Kupfer DJ, Frank E, Grochocinski VJ, et al. Demographic and clinical characteristics of individuals in a bipolar disorder case registry. J Clin Psychiatry. 2002;63(2):120-125. PubMed doi:10.4088/JCP.v63n0206
10. Angst J, Sellaro R. Historical perspectives and natural history of bipolar disorder. Biol Psychiatry. 2000;48(6):445-457. PubMed doi:10.1016/S0006-3223(00)00909-4
11. Bowden CL. Strategies to reduce misdiagnosis of bipolar depression. Psychiatr Serv. 2001;52(1):51-55. PubMed doi:10.1176/appi.ps.52.1.51
12. Judd LL, Akiskal HS, Schettler PJ, et al. The long-term natural history of the weekly symptomatic status of bipolar I disorder. Arch Gen Psychiatry. 2002;59(6):530-537. PubMed doi:10.1001/archpsyc.59.6.530
13. Post RM, Denicoff KD, Leverich GS, et al. Morbidity in 258 bipolar outpatients followed for 1 year with daily prospective ratings on the NIMH life chart method. J Clin Psychiatry. 2003;64(6):680-690, quiz 738-739. PubMed doi:10.4088/JCP.v64n0610
14. Mantere O, Suominen K, Valtonen HM, et al. Differences in outcome of DSM-IV bipolar I and II disorders. Bipolar Disord. 2008;10(3):413-425. PubMed doi:10.1111/j.1399-5618.2007.00502.x
15. Hoang U, Stewart R, Goldacre MJ. Mortality after hospital discharge for people with schizophrenia or bipolar disorder: retrospective study of linked English hospital episode statistics, 1999-2006. BMJ. 2011;343(1):d5422. PubMed doi:10.1136/bmj.d5422
16. Yatham LN, Lecrubier Y, Fieve RR, et al. Quality of life in patients with bipolar I depression: data from 920 patients. Bipolar Disord. 2004;6(5):379-385. PubMed doi:10.1111/j.1399-5618.2004.00134.x
17. Hlastala SA, Frank E, Mallinger AG, et al. Bipolar depression: an underestimated treatment challenge. Depress Anxiety. 1997;5(2):73-83. PubMed doi:10.1002/(SICI)1520-6394(1997)5:2<73::AID-DA3>3.0.CO;2-6
18. Dilsaver SC, Chen YW, Swann AC, et al. Suicidality, panic disorder and psychosis in bipolar depression, depressive-mania and pure-mania. Psychiatry Res. 1997;73(1-2):47-56. PubMed doi:10.1016/S0165-1781(97)00109-1
19. Young LT, Joffe RT, Robb JC, et al. Double-blind comparison of addition of a second mood stabilizer versus an antidepressant to an initial mood stabilizer for treatment of patients with bipolar depression. Am J Psychiatry. 2000;157(1):124-126. PubMed
20. Nemeroff CB, Evans DL, Gyulai L, et al. Double-blind, placebo-controlled comparison of imipramine and paroxetine in the treatment of bipolar depression. Am J Psychiatry. 2001;158(6):906-912. PubMed doi:10.1176/appi.ajp.158.6.906
21. Gijsman HJ, Geddes JR, Rendell JM, et al. Antidepressants for bipolar depression: a systematic review of randomized, controlled trials. Am J Psychiatry. 2004;161(9):1537-1547. PubMed doi:10.1176/appi.ajp.161.9.1537
22. Tohen M, Vieta E, Calabrese J, et al. Efficacy of olanzapine and olanzapine-fluoxetine combination in the treatment of bipolar I depression. Arch Gen Psychiatry. 2003;60(11):1079-1088. PubMed doi:10.1001/archpsyc.60.11.1079
23. Himmelhoch JM, Fuchs CZ, Symons BJ. A double-blind study of tranylcypromine treatment of major anergic depression. J Nerv Ment Dis. 1982;170(10):628-634. PubMed doi:10.1097/00005053-198210000-00007
24. Altshuler L, Suppes T, Black D, et al. Impact of antidepressant discontinuation after acute bipolar depression remission on rates of depressive relapse at 1-year follow-up. Am J Psychiatry. 2003;160(7):1252-1262. PubMed doi:10.1176/appi.ajp.160.7.1252
25. Sachs GS, Nierenberg AA, Calabrese JR, et al. Effectiveness of adjunctive antidepressant treatment for bipolar depression. N Engl J Med. 2007;356(17):1711-1722. PubMed doi:10.1056/NEJMoa064135
26. Sidor MM, Macqueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry. 2011;72(2):156-167. PubMed doi:10.4088/JCP.09r05385gre
27. Mendlewicz J, Youdim MB. Antidepressant potentiation of 5-hydroxytryptophan by L-deprenil in affective illness. J Affect Disord. 1980;2(2):137-146. PubMed doi:10.1016/0165-0327(80)90013-0
28. De Wilde JE, Doogan DP. Fluvoxamine and chlorimipramine in endogenous depression. J Affect Disord. 1982;4(3):249-259. PubMed doi:10.1016/0165-0327(82)90009-X
29. McElroy SL, Weisler RH, Chang W, et al; EMBOLDEN II (Trial D1447C00134) Investigators. A double-blind, placebo-controlled study of quetiapine and paroxetine as monotherapy in adults with bipolar depression (EMBOLDEN II). J Clin Psychiatry. 2010;71(2):163-174. PubMed doi:10.4088/JCP.08m04942gre
30. Amsterdam JD, Shults J. Comparison of fluoxetine, olanzapine, and combined fluoxetine plus olanzapine initial therapy of bipolar type I and type II major depression: lack of manic induction. J Affect Disord. 2005;87(1):121-130. PubMed doi:10.1016/j.jad.2005.02.018
31. Amsterdam JD, Wang CH, Shwarz M, et al. Venlafaxine versus lithium monotherapy of rapid and non-rapid cycling patients with bipolar II major depressive episode: a randomized, parallel group, open-label trial. J Affect Disord. 2009;112(1-3):219-230. PubMed doi:10.1016/j.jad.2008.03.029
32. Amsterdam JD, Shults J. Efficacy and safety of long-term fluoxetine versus lithium monotherapy of bipolar II disorder: a randomized, double-blind, placebo-substitution study. Am J Psychiatry. 2010;167(7):792-800. PubMed doi:10.1176/appi.ajp.2009.09020284
33. Amsterdam JD, Luo L, Shults J. Efficacy and mood conversion rate during long-term fluoxetine v lithium monotherapy in rapid- and non-rapid-cycling bipolar II disorder. Br J Psychiatry. 2013;202(4):301-306. PubMed doi:10.1192/bjp.bp.111.104711
34. Yatham LN, Kennedy SH, Parikh SV, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) collaborative update of CANMAT guidelines for the management of patients with bipolar disorder: update 2013. Bipolar Disord. 2013;15(1):1-44. PubMed doi:10.1111/bdi.12025
35. Post RM, Altshuler LL, Frye MA, et al. Rate of switch in bipolar patients prospectively treated with second-generation antidepressants as augmentation to mood stabilizers. Bipolar Disord. 2001;3(5):259-265. PubMed doi:10.1034/j.1399-5618.2001.30505.x
36. Vieta E, Martinez-Arán A, Goikolea JM, et al. A randomized trial comparing paroxetine and venlafaxine in the treatment of bipolar depressed patients taking mood stabilizers. J Clin Psychiatry. 2002;63(6):508-512. PubMed doi:10.4088/JCP.v63n0607
37. Post RM, Altshuler LL, Leverich GS, et al. Mood switch in bipolar depression: comparison of adjunctive venlafaxine, bupropion and sertraline. Br J Psychiatry. 2006;189(2):124-131. PubMed doi:10.1192/bjp.bp.105.013045
38. Amsterdam JD, Garcia-Espa×±a F. Venlafaxine monotherapy in women with bipolar II and unipolar major depression. J Affect Disord. 2000;59(3):225-229. PubMed doi:10.1016/S0165-0327(99)00149-4
39. Joffe RT, MacQueen GM, Marriott M, et al. Induction of mania and cycle acceleration in bipolar disorder: effect of different classes of antidepressant. Acta Psychiatr Scand. 2002;105(6):427-430. PubMed doi:10.1034/j.1600-0447.2002.02360.x
40. Ghaemi SN, Hsu DJ, Soldani F, et al. Antidepressants in bipolar disorder: the case for caution. Bipolar Disord. 2003;5(6):421-433. PubMed doi:10.1046/j.1399-5618.2003.00074.x
41. Altshuler LL, Post RM, Leverich GS, et al. Antidepressant-induced mania and cycle acceleration: a controversy revisited. Am J Psychiatry. 1995;152(8):1130-1138. PubMed
42. Benazzi F. Antidepressant-associated hypomania in outpatient depression: a 203-case study in private practice. J Affect Disord. 1997;46(1):73-77. PubMed doi:10.1016/S0165-0327(97)00082-7
43. Koukopoulos A, Faedda G, Proietti R, et al. Mixed depressive syndrome [article in French]. Encephale. 1992;18(Spec No 1):19-21. PubMed
44. Perugi G, Akiskal HS, Micheli C, et al. Clinical characterization of depressive mixed state in bipolar-I patients: Pisa-San Diego collaboration. J Affect Disord. 2001;67(1-3):105-114. PubMed doi:10.1016/S0165-0327(01)00443-8
45. El-Mallakh RS, Ghaemi SN, Sagduyu K, et al; STEP-BD Investigators. Antidepressant-associated chronic irritable dysphoria (ACID) in STEP-BD patients. J Affect Disord. 2008;111(2-3):372-377. PubMed doi:10.1016/j.jad.2008.03.025
46. Koukopoulos A, Sani G, Koukopoulos AE, et al. Melancholia agitata and mixed depression. Acta Psychiatr Scand suppl. 2007;115(433):50-57. PubMed doi:10.1111/j.1600-0447.2007.00963.x
47. Azorin JM, Kaladjian A, Adida M, et al. Self-assessment and characteristics of mixed depression in the French national EPIDEP study. J Affect Disord. 2012;143(1-3):109-117. PubMed doi:10.1016/j.jad.2012.05.036
48. Goldberg JF, Perlis RH, Bowden CL, et al. Manic symptoms during depressive episodes in 1,380 patients with bipolar disorder: findings from the STEP-BD. Am J Psychiatry. 2009;166(2):173-181. PubMed doi:10.1176/appi.ajp.2008.08050746
49. Eppel AB. Antidepressants in the treatment of bipolar disorder: decoding contradictory evidence and opinion. Harv Rev Psychiatry. 2008;16(3):205-209. PubMed doi:10.1080/10673220802160381
50. Stetka B, Correll C. Medscape Psychiatry. A guide to DSM-5. Medscape Psychiatry. http://www.medscape.com/viewarticle/803884. Updated May 21, 2013. Accessed July 30, 2014.
51. Strejilevich SA, Martino DJ, Marengo E, et al. Long-term worsening of bipolar disorder related with frequency of antidepressant exposure. Ann Clin Psychiatry. 2011;23(3):186-192. PubMed
52. Valent× M, Pacchiarotti I, Rosa AR, et al. Bipolar mixed episodes and antidepressants: a cohort study of bipolar I disorder patients. Bipolar Disord. 2011;13(2):145-154. PubMed doi:10.1111/j.1399-5618.2011.00908.x
53. Pacchiarotti I, Mazzarini L, Kotzalidis GD, et al. Mania and depression: mixed, not stirred. J Affect Disord. 2011;133(1-2):105-113. PubMed doi:10.1016/j.jad.2011.03.037
54. Sussman M, Friedman M, Korn JR, et al. The relationship between use of antidepressants and resource utilization among patients with manic or mixed bipolar disorder episodes: findings from a managed care setting. J Affect Disord. 2012;138(3):425-432. PubMed doi:10.1016/j.jad.2011.12.048
55. Undurraga J, Baldessarini RJ, Valenti M, et al. Suicidal risk factors in bipolar I and II disorder patients. J Clin Psychiatry. 2012;73(6):778-782. PubMed doi:10.4088/JCP.11m07041
56. Thase ME, Macfadden W, Weisler RH, et al; BOLDER II Study Group. Efficacy of quetiapine monotherapy in bipolar I and II depression: a double-blind, placebo-controlled study (the BOLDER II study). J Clin Psychopharmacol. 2006;26(6):600-609. PubMed doi:10.1097/01.jcp.0000248603.76231.b7
57. Vieta E, Locklear J, Günther O, et al. Treatment options for bipolar depression: a systematic review of randomized, controlled trials. J Clin Psychopharmacol. 2010;30(5):579-590. PubMed doi:10.1097/JCP.0b013e3181f15849
58. Calabrese JR, Huffman RF, White RL, et al. Lamotrigine in the acute treatment of bipolar depression: results of five double-blind, placebo-controlled clinical trials. Bipolar Disord. 2008;10(2):323-333. PubMed doi:10.1111/j.1399-5618.2007.00500.x
59. Young AH, McElroy SL, Bauer M, et al; EMBOLDEN I (Trial 001) Investigators. A double-blind, placebo-controlled study of quetiapine and lithium monotherapy in adults in the acute phase of bipolar depression (EMBOLDEN I). J Clin Psychiatry. 2010;71(2):150-162. PubMed doi:10.4088/JCP.08m04995gre
60. Grunze H, Vieta E, Goodwin GM, et al; WFSBP Task Force on Treatment Guidelines for Bipolar Disorders. The World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the biological treatment of bipolar disorders: update 2012 on the long-term treatment of bipolar disorder. World J Biol Psychiatry. 2013;14(3):154-219. PubMed doi:10.3109/15622975.2013.770551
61. Loebel A, Cucchiaro J, Silva R, et al. Lurasidone as adjunctive therapy with lithium or valproate for the treatment of bipolar I depression: a randomized, double-blind, placebo-controlled study. Am J Psychiatry. 2014;171(2):169-177. PubMed doi:10.1176/appi.ajp.2013.13070985
62. Loebel A, Cucchiaro J, Silva R, et al. Lurasidone monotherapy in the treatment of bipolar I depression: a randomized, double-blind, placebo-controlled study. Am J Psychiatry. 2014;171(2):160-168. PubMed doi:10.1176/appi.ajp.2013.13070984
63. Zarate CA Jr, Payne JL, Singh J, et al. Pramipexole for bipolar II depression: a placebo-controlled proof of concept study. Biol Psychiatry. 2004;56(1):54-60. PubMed doi:10.1016/j.biopsych.2004.03.013
64. Frye MA, Grunze H, Suppes T, et al. A placebo-controlled evaluation of adjunctive modafinil in the treatment of bipolar depression. Am J Psychiatry. 2007;164(8):1242-1249. PubMed doi:10.1176/appi.ajp.2007.06060981
65. Zarate CA Jr, Brutsche NE, Ibrahim L, et al. Replication of ketamine’s antidepressant efficacy in bipolar depression: a randomized controlled add-on trial. Biol Psychiatry. 2012;71(11):939-946. PubMed doi:10.1016/j.biopsych.2011.12.010
66. Yatham LN, Kusumakar V, Parikh SV, et al. Bipolar depression: treatment options. Can J Psychiatry. 1997;42(suppl 2):87S-91S. PubMed