Objective: To review the literature examining the use of mirtazapine with an emphasis on its therapeutic benefits for psychiatric patients with comorbid medical conditions.
Data Sources: MEDLINE, PsycINFO, Global Health, and AGRICOLA were searched using the terms mirtazapine OR Remeron. Limits were English language, human, year 1980-2012, treatment and prevention, and therapy.
Study Selection: Two hundred ninety-three articles were identified.
Data Extraction: Identified articles were reviewed with a focus on indications and therapeutic benefits in patients with medical comorbidities.
Results: Mirtazapine is an effective antidepressant with unique mechanisms of action. It is characterized by a relatively rapid onset of action, high response and remission rates, a favorable side-effect profile, and several unique therapeutic benefits over other antidepressants. Mirtazapine has also shown promise in treating some medical disorders, including neurologic conditions, and ameliorating some of the associated debilitating symptoms of weight loss, insomnia, and postoperative nausea and vomiting.
Conclusions: Mirtazapine offers clinicians multiple therapeutic advantages especially when treating patients with comorbid medical illness.
Prim Care Companion CNS Disord 2013;15(5):doi:10.4088/PCC.13r01525
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: April 29, 2013; accepted July 9, 2013.
Published online: October 10, 2013.
Corresponding author: Abdulkader Alam, MD, 3520 5th Ave, Lower Level, Ste 1, Pittsburgh, PA 15213 ([email protected]).
A Review of Therapeutic Uses of Mirtazapine
in Psychiatric and Medical Conditions
ABSTRACT
Objective: To review the literature examining the use of mirtazapine with an emphasis on its therapeutic benefits for psychiatric patients with comorbid medical conditions.
Data Sources: MEDLINE, PsycINFO, Global Health, and AGRICOLA were searched using the terms mirtazapine OR Remeron. Limits were English language, human, year 1980–2012, treatment and prevention, and therapy.
Study Selection: Two hundred ninety-three articles were identified.
Data Extraction: Identified articles were reviewed with a focus on indications and therapeutic benefits in patients with medical comorbidities.
Results: Mirtazapine is an effective antidepressant with unique mechanisms of action. It is characterized by a relatively rapid onset of action, high response and remission rates, a favorable side-effect profile, and several unique therapeutic benefits over other antidepressants. Mirtazapine has also shown promise in treating some medical disorders, including neurologic conditions, and ameliorating some of the associated debilitating symptoms of weight loss, insomnia, and postoperative nausea and vomiting.
Conclusions: Mirtazapine offers clinicians multiple therapeutic advantages especially when treating patients with comorbid medical illness.
Prim Care Companion CNS Disord 2013;15(5):doi:10.4088/PCC.13r01525
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: April 29, 2013; accepted July 9, 2013.
Published online: October 10, 2013.
Corresponding author: Abdulkader Alam, MD, 3520 5th Ave, Lower Level, Ste 1, Pittsburgh, PA 15213 ([email protected]).
Mirtazapine is a novel antidepressant originally known as Org 3770. It was first synthesized in The Netherlands in 19871 and introduced in the United States in 1996. Mirtazapine has a unique dual mode of action as a noradrenergic and specific serotonergic antidepressant.2 Mirtazapine shows rapid improvement in the symptoms of depression, with minimal anticholinergic or serotonin-related adverse effects. Comparisons of mirtazapine to the selective serotonin reuptake inhibitors (SSRIs) suggest a faster onset of action.3 Most notably, in a trial designed specifically to test onset of action of mirtazapine versus sertraline, a significant difference in favor of mirtazapine was observed as early as the fourth day of treatment.4 The advantages of quick onset of effects and a favorable safety profile have resulted in frequent use of mirtazapine in psychiatric patients with comorbid medical problems. This article reviews the literature examining the use of mirtazapine with an emphasis on its therapeutic benefits for patients with comorbid medical conditions.
METHOD
MEDLINE, PsycINFO, Global Health, and AGRICOLA were searched using the keywords mirtazapine OR Remeron. Limits were English language, human, years 1980–2012, treatment and prevention, and therapy. Two hundred ninety-three articles were identified. The identified articles were reviewed with a focus on indications and therapeutic benefits in patients with medical comorbidities.
PHARMACOLOGIC PROPERTIES
Mirtazapine, a noradrenergic and specific serotonergic antidepressant,5,6 enhances noradrenergic transmission via blockade of central α2-adrenoceptors.7,8 Mirtazapine is a potent serotonin 5-HT2 and 5-HT3 antagonist, thereby increasing serotonergic stimulation via the 5-HT1 receptor.7 Mirtazapine has no significant affinity for dopamine receptors, low affinity for muscarinic cholinergic receptors, and no effect on monoamine reuptake.8,9
Mirtazapine is available in an oral tablet form and as an oral rapidly disintegrating tablet.10 It is well absorbed by the gastrointestinal tract, and its bioavailability does not appear to be affected by the presence of food.11 Mirtazapine is extensively metabolized in the liver, and its metabolites are eliminated primarily in the urine (up to 75%) and in the feces.11 Mirtazapine has a half-life of 20–40 hours, which may increase by 30%–40% in patients with hepatic impairment.11 The drug’s clearance may decrease by 30%–50% in patients with moderate-to-severe renal impairment.11 Mirtazapine does not auto-induce hepatic isoenzymes. In vitro studies show that mirtazapine is not a potent inhibitor or inducer of P450 isoenzymes 1A2, 2D6, and 3A4.11 The most commonly reported adverse effects are transient somnolence, hyperphagia, and weight gain, which may be attributed in part to the antihistaminic activity of mirtazapine at low doses.8
THERAPEUTIC BENEFITS
Depressive Disorders
As an antidepressant, mirtazapine has proven to be equally as effective as the tricyclic antidepressants and trazodone in patients with moderate-to-severe depression in both inpatient and ambulatory settings.12–16 Mirtazapine was found to be as effective as amitriptyline, but it causes fewer and less severe anticholinergic and cardiovascular side effects.17 When compared with the SSRIs fluoxetine,18 citalopram,19 sertraline,4 and paroxetine,20,21 as well as the serotonin-norepinephrine reuptake inhibitor venlafaxine,9 mirtazapine was shown to be equally effective but with a significantly earlier onset of action, with effects seen as early as 1 and 2 weeks after treatment initiation.4,22 Compared to participants taking SSRIs, those taking mirtazapine had a 74% greater likelihood of achieving remission during the first 2 weeks of therapy.23 This early improvement appears to be a specific antidepressant effect that is independent of mirtazapine’s sleep-improving properties,9 and it has been identified as a highly sensitive predictor of later stable response or stable remission.24 Early improvement was also noted in the primary care setting in depressed patients treated with mirtazapine.25 Similarly, in primary care, mirtazapine (30–45 mg/d) showed a statistically significant early improvement over paroxetine (20–30 mg/d), although both were found to be efficacious.26 Benefits for other subtypes of depression are listed in Table 1.
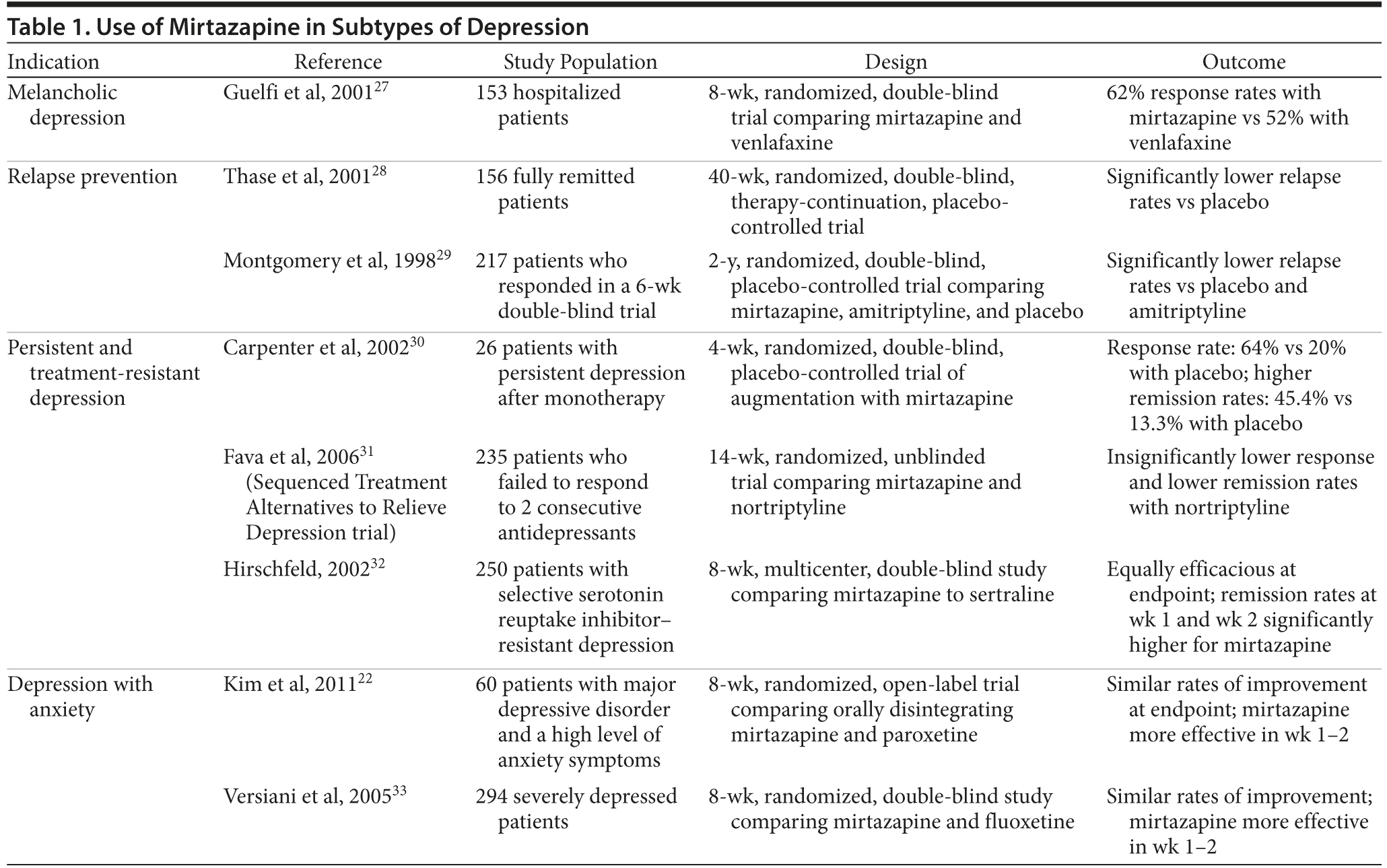
Depression and
Comorbid Medical Conditions
Treating depression in patients with general medical conditions can be done safely and effectively with antidepressants, including mirtazapine, with no additional adverse effects or tolerability burden.34 One study suggests that mirtazapine and venlafaxine are effective not only for the treatment of overall symptoms of depression, but may also be useful for the treatment of somatic symptoms in depressed patients.35
Post–myocardial infarction depression. Depression after myocardial infarction is associated with increased cardiac morbidity and mortality, while antidepressant treatment in patients with cardiac disease and depression may result in significant therapeutic benefit.36 Additionally, there is evidence that nonresponse to treatment of post–myocardial infarction depression may be associated with more cardiac events.37
Honig et al38 examined mirtazapine’s efficacy in post–myocardial infarction depressed patients in a randomized, double-blind, placebo-controlled, 24-week trial and found that mirtazapine (15–45 mg) exhibited safety and efficacy on primary and secondary depression measures in the treatment of post–myocardial infarction depression. There were no differences between the control and treatment groups in serious adverse events, although patients reported more fatigue and appetite changes with mirtazapine.38 Additionally, antidepressive response to mirtazapine (30 mg) in post–myocardial infarction patients was associated with significant decrease in inflammatory markers in contrast to placebo and nonresponder groups.39
Poststroke depression. Niedermaier et al40 found that prophylactic treatment with mirtazapine (30 mg) begun 1 day poststroke significantly reduced the rate of developing poststroke depression, with 5.7% (2/35) of treated patients becoming depressed compared to 40% (14/35) of nontreated patients. Mirtazapine was also shown to be effective in treating poststroke depression in the nontreated patients who developed depression in the first phase of the study.40
Temporal lobe epilepsy and comorbid depression. Mirtazapine, citalopram, and reboxetine were each evaluated for the treatment of patients with temporal lobe epilepsy and depression in an inpatient setting.41 Treatment was efficacious with all 3 antidepressants. No serious adverse events, drug interactions, or increase in frequency or severity of seizures occurred. However, the endpoint dropout rate for patients treated with mirtazapine was significantly higher than for those treated with either citalopram or reboxetine.41
Substance dependence and comorbid depression. In a study of patients with alcohol dependence and comorbid depressive disorder, Altintoprak et al42 demonstrated significant improvement in depression and alcohol craving scores with both mirtazapine and amitriptyline, although mirtazapine was better tolerated. Another study looked at treating depressed cocaine-dependent subjects with either mirtazapine (45 mg daily) or placebo for 12 weeks.43 Urine concentrations of benzoylecgonine (the main cocaine metabolite) and self-reports did not show mirtazapine to be more effective than a placebo in reducing cocaine use or improving symptoms of depression.43
Geriatric depression. A 10-week, open-label trial of mirtazapine in 16 elderly patients with depression and 1 or more serious comorbid medical illnesses found that mirtazapine improves depression, insomnia, anxiety, somatic symptoms, and certain quality-of-life measures.44 A 12-week open-label trial suggested that mirtazapine orally disintegrating tablets were effective and well tolerated in depressed nursing home residents aged 85 years or older.45 A comparison study of depressed subjects aged 65 years or older found both mirtazapine and paroxetine to be effective during acute (8 weeks) and extension (16 weeks) phases of the trial.46 However, mirtazapine demonstrated a faster median response time of 26 days compared to 40 days for paroxetine and was associated with greater reduction in anxiety/somatization and sleep disturbance scores.46 Additionally, mirtazapine (15–45 mg) was found to be similar in efficacy and safety when compared to amitriptyline (30–90 mg) in the treatment of depressed patients aged 60–85 years.47
Dementia with comorbid depression and agitation. A study assessed the use of antidepressants for depression associated with dementia in participants in England and found no difference in depression scores at 13 weeks between 111 controls and 107 participants receiving sertraline or mirtazapine or between the participants in the mirtazapine and sertraline arms.48 For agitation, an open-label prospective pilot study of 16 patients with severe agitation associated with Alzheimer’s dementia found that mirtazapine was associated with significant improvement in agitation and appetite without significant side effects or cognitive deterioration.49
Substance Use Disorders
Alcohol dependence. Liappas et al50 demonstrated that mirtazapine significantly improved the effects of cognitive-behavioral therapy on social anxiety symptoms in patients with alcohol dependence after a detoxification protocol. Mirtazapine also reduced anxiety and depressive symptoms more quickly when administered in combination with psychotherapy during the postwithdrawal phase.51 Compared to venlafaxine, mirtazapine significantly improved anxiety and depression scores in patients undergoing detoxification from alcohol.52
Methamphetamine use disorders. A randomized controlled trial found that treatment with mirtazapine in actively using methamphetamine-dependent men who have sex with men resulted in decreased methamphetamine use and decreased sexual risk behaviors despite low-to-moderate medication adherence.53 In a double-blind, randomized, placebo-controlled trial of withdrawal treatment in 31 participants,54 mirtazapine was not associated with a significant difference in participant retention or symptom reduction. In another study of withdrawal treatment,55 mirtazapine was associated with a less severe withdrawal than pericyazine, although modafinil resulted in the mildest withdrawal.
Anxiety and Related Disorders
Multiple studies have looked into the benefits of mirtazapine in treating anxiety disorders. Those benefits are summarized in Table 2.
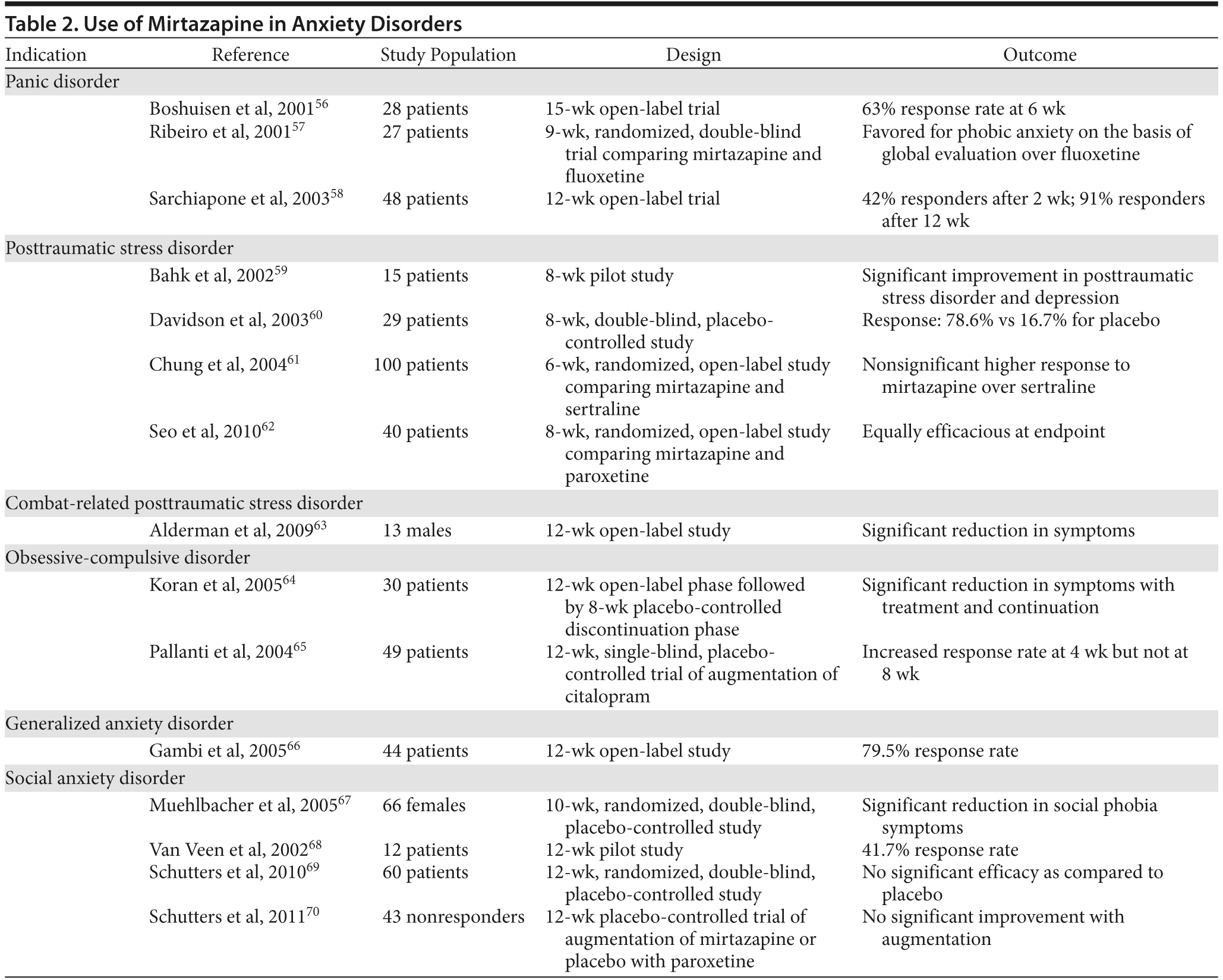
Neurologic Disorders
The use of mirtazapine in contemporary neurology is based on the improved understanding of disease mechanisms and from the drug’s effectiveness seen with certain movement disorders.
Progressive multifocal leukoencephalopathy. Progressive multifocal leukoencephalopathy is a fatal demyelinating disease caused by the reactivation of latent infection with human John Cunningham polyomavirus, predominantly affecting patients infected with human immunodeficiency virus (HIV).71 In 2004, Elphick et al72 published a seminal article on 5-HT2A receptor mediation of John Cunningham polyomavirus entry into cells, spurring the empiric use of mirtazapine in progressive multifocal leukoencephalopathy therapy on the basis of the drug’s 5-HT2 receptor antagonism. Cettomai and McArthur73 reported on a case series of 4 HIV-infected patients diagnosed with progressive multifocal leukoencephalopathy treated with highly active antiretroviral therapy followed by the addition of mirtazapine (15 mg), in which 3 of the 4 patients demonstrated clinical improvement, while the patient who failed to show improvement was not fully adherent with treatment. Also, a 49-year-old man with HIV on highly active antiretroviral therapy who was diagnosed with progressive multifocal leukoencephalopathy and given mirtazapine and mefloquine hydrochloride showed subjective functional and cognitive improvement.74 However, a 55-year-old man with hepatitis C virus treated with pegylated interferon-α and ribavirin and subsequently diagnosed with progressive multifocal leukoencephalopathy resulted in a mortal outcome despite a 5-day course of cytarabine and mirtazapine.75
In a review of 28 cases of patients with multiple sclerosis who developed natalizumab-associated progressive multifocal leukoencephalopathy, Clifford et al76 found that mirtazapine was used as a part of adjunctive therapy in 39% of the cases, although it is difficult to comment on mirtazapine’s contribution in the setting of multidrug regimens.
Movement Disorders
The use of mirtazapine in treating tremors started with the description by Pact and Giduz77 of the reduction or elimination of parkinsonian tremor, action tremor, and levodopa-induced dyskinesias in 5 patients taking mirtazapine (30 mg). Tremor and dyskinesias reemerged on discontinuation of mirtazapine in 2 patients and once again abated when mirtazapine was restarted.77 A double-blind, placebo-controlled, cross-over pilot study of mirtazapine as an add-on therapy in reducing essential tremor in 17 patients, 13 of whom were already treated with other antitremor medications, showed global improvement in 3 patients but no significant improvement compared to baseline.78 Uccellini et al79 conducted an open-label observer-blind study of 30 patients with untreated essential tremor and found that 85% of those who remained on mirtazapine treatment therapy after 1 month demonstrated good control of essential tremor. After 1 year of treatment, 55% of patients continued to show benefit,79 which is comparable to the estimated 50% improvement rate seen with propranolol and primidone.80
Tension-type headache. A randomized, double-blind, placebo-controlled, crossover trial in 24 patients with chronic tension-type headache found mirtazapine to be effective in reducing headache severity81 at a rate comparable to treatment with amitriptyline.82 In a follow-up double-blind, placebo-controlled, parallel trial of mirtazapine, ibuprofen, or the combination of both in 93 patients with chronic tension-type headache, low-dose mirtazapine alone was found to reduce headache severity by 20%, with a noted dose-response effect on efficacy and tolerance.83
Other Possible Therapeutic Benefits
Pain in advanced cancer. Interest in the use of mirtazapine as an adjuvant treatment in the palliation of advanced cancer was originally spurred by the hope that it could offer an alternative to tricyclic antidepressants in pain management. In fact, a double-blind cross-over trial demonstrated that a single dose of mirtazapine could significantly increase the pain threshold of healthy participants.84 A pilot open-label trial,85 which targeted advanced cancer patients who were experiencing moderate-to-severe residual pain despite opioid maintenance, identified only a small insignificant improvement in pain and pain relief scores with mirtazapine, although there were significant dose-independent improvements in self-rated depression and functional assessment measures.
Fibromyalgia
In a 6-week open-label trial of mirtazapine,86 54% of the 26 fibromyalgia patients who completed the study demonstrated a clinically significant reduction in pain intensity and in mean weekly dosage of acetaminophen. Additionally, there was a significant improvement in sleep quality and somatic symptoms, including cold extremities, dry mouth, sweating, dizziness, and headache. Of note, the magnitude of reduction in major fibromyalgia symptoms was significantly correlated with the magnitude of reduction in depression.86
Weight Loss or Appetite Stimulation
Trials of mirtazapine in the treatment of weight loss were motivated by weight gain being one of the most common side effects, which was attributed to increased appetite87 and food craving.88 A 6-week, open-label, crossover trial of mirtazapine in 36 advanced cancer patients with pain found a significant increase in weight at weeks 4 and 7, along with improvement in appetite.85 Another open-label trial of mirtazapine in 17 patients with cancer-related cachexia/anorexia found weight gain in 24% of patients and weight maintenance in 6%, which was encouraging given that patients were losing weight prior to enrollment.89 In an uncontrolled trial of mirtazapine administered over a range of 29 to 412 days in 5 patients with cystic fibrosis,90 all demonstrated an increase in weight, body fat, and growth velocity at the end of the study. In a retrospective analysis of 6 patients with cystic fibrosis aged 10–19 years who were treated with mirtazapine over a range of 8 to 28 months,91 all patients demonstrated an increase in body mass index percentile for age. One instance of cystic fibrosis-related diabetes reported in the trial by Boas et al90 contributed to the lack of support for the drug as an appetite stimulant in cystic fibrosis.92 On the other hand, in a small naturalistic study of 11 adults, Himmerich et al93 found that inpatients with major depression actually showed improved glucose tolerance after 2 to 6 weeks of mirtazapine treatment despite the associated weight gain.
Insomnia
The high incidence of somnolence, 53.3% reported across US trials of mirtazapine,94 spurred an interest in leveraging this side effect for the treatment of insomnia. A 2-week parallel trial of 15 mg versus 30 mg daily mirtazapine in 130 depressed patients with insomnia found persistent improvement in sleep quality and quantity, ease of getting to sleep, and daytime alertness with both doses.95 The initial 10% incidence of somnolence decreased as the trial progressed, suggesting rapid development of tolerance to the sleep induction effect of H1 antagonism.95 In a double-blind placebo-controlled study of 20 young healthy volunteers given 1 dose of 30 mg of mirtazapine, Aslan et al96 confirmed the findings of increased sleep continuity and efficiency in the acute setting while reporting additional benefit of prolonged slow-wave sleep attributed to 5-HT2A/C antagonism. Of note, 5-HT2A/C antagonism has not been implicated in unwanted sedation or tolerance.96 A double-blind study of 19 depressed patients with insomnia compared mirtazapine versus fluoxetine over an 8-week period and found that the mirtazapine group alone demonstrated significant improvements in total sleep time and sleep latency without the unwanted rapid eye movement suppression that is seen with many antidepressants, including fluoxetine.97
Obstructive Sleep Apnea
Although a manufacturer-sponsored, double-blind, 3-arm, crossover trial of placebo versus mirtazapine in 12 newly diagnosed patients with uncomplicated obstructive sleep apnea showed significant reduction of apnea-hypopnea index,98 2 randomized, double-blind, placebo-controlled trials of mirtazapine in patients with obstructive sleep apnea found no measurement of sleep apnea improved with mirtazapine.99
Nausea and Vomiting
Mirtazapine’s blockade of 5-HT3 receptors may help to prevent nausea and vomiting in a manner similar to ondansetron. Initial reports from the obstetrics literature demonstrated its successful use in the treatment of depression, anxiety, and hyperemesis gravidarum during pregnancy.100,101 In a double-blind randomized trial of mirtazapine plus dexamethasone versus dexamethasone alone in 80 gynecologic surgery patients with risk factors for postoperative nausea and vomiting, Chen et al102 found that 80% of the mirtazapine group did not experience postoperative nausea and vomiting or require administration of rescue antiemetic versus 50% of the placebo group. Furthermore, patients in the mirtazapine group experienced significantly lower anxiety 1 hour after mirtazapine administration without significant impact on most anesthesia parameters and recovery times.102 In a randomized, placebo-controlled trial of mirtazapine given 1 hour prior to orthopedic surgery in 84 men and 16 women,103 regardless of risk factors, the incidence of postoperative nausea and vomiting was again significantly lower in the mirtazapine group when compared to placebo.
Sexual Dysfunction
Although mirtazapine may be associated with a lower incidence of sexual side effects than other antidepressants,104 a study of 148 premenopausal women with sexual dysfunction associated with fluoxetine did not find the addition of mirtazapine to be efficacious in reducing sexual dysfunction.105 However, a study of augmentation in generalized anxiety disorder found that the 21 patients treated with the combination of mirtazapine and paroxetine experienced less sexual dysfunction than the 22 patients treated with paroxetine and placebo.106 For lifelong early ejaculation, a double-blind fixed-dose study of 24 healthy men did not show mirtazapine to be effective in prolonging ejaculation latency time.107
CONCLUSION
Mirtazapine is a noradrenergic and specific serotonergic antidepressant that is approved for use in the treatment of major depressive disorder. Its unique pharmacologic properties are thought to be responsible for its excellent tolerability. Monotherapy with mirtazapine 15–45 mg/d leads to rapid and sustained improvements in depressive symptoms. This efficacy has been demonstrated in patients treated in the hospital, as outpatients, and in primary care. Mirtazapine was also effective in subgroups of depressed patients, particularly anxious patients and those with melancholic depression, treatment-resistant depression, geriatric depression, and depression and anxiety associated with alcohol dependence and agitated elderly patients. Mirtazapine appears to be safe and effective in the treatment of post–myocardial infarction depression, as well as in the prevention and treatment of depression after stroke and in association with temporal lobe epilepsy. A number of rather small trials have suggested the efficacy of mirtazapine in the treatment of patients with anxiety disorders including posttraumatic stress disorder (see Table 2), but larger-scale studies are needed to support these conclusions.
Our results also suggest a range of clinically useful applications including improved sleep, antiemetic benefits, improved appetite, and pain management. The results for pain management are intriguing and should encourage further trials of mirtazapine in other patient populations with pain. The weight gain associated with mirtazapine as a side effect was used in treating cancer-related cachexia as well as weight loss with cystic fibrosis.
Mirtazapine showed potential in the treatment of some medical and neurologic conditions. Treatment of progressive multifocal leukoencephalopathy was very promising, although only a randomized trial could establish the role of mirtazapine in the treatment of this disease. Mirtazapine may also present an alternate treatment for essential tremor with a different side effect profile and improved tolerability. Although studies that looked at perioperative use of mirtazapine included fairly narrow patient populations, the combination of their results suggests that mirtazapine can be useful in perioperative settings.
There is no conclusive evidence to support the use of mirtazapine in treatment of depression associated with dementia, treatment of depression with cocaine dependence, or for treatment of obstructive sleep apnea. Mirtazapine may improve sexual function in some patients taking SSRIs, but that finding was not supported in premenopausal women taking an SSRI, nor was it effective in treating males with early ejaculation sexual dysfunctions.
Drug names: citalopram (Celexa and others), cytarabine (Depocyt, Cytosar-U, and others), fluoxetine (Prozac and others), mirtazapine (Remeron and others), modafinil (Provigil), nortriptyline (Pamelor, Aventyl, and others), ondansetron (Zofran and others), paroxetine (Paxil, Pexeva, and others), propranolol (Inderal, InnoPran, and others), sertraline (Zoloft and others), trazodone (Oleptro and others), venlafaxine (Effexor and others).
Author affiliations: Departments of Psychiatry and Medicine (Dr Alam), University of Pittsburgh School of Medicine (Ms Voronovich), Pittsburgh, Pennsylvania; and University of Alabama School of Medicine, Birmingham (Dr Carley).
Potential conflicts of interest: None reported.
Funding/support: None reported.
REFERENCES
1. Delbressine LP, Moonen ME, Kaspersen FM, et al. Pharmacokinetics and biotransformation of mirtazapine in human volunteers. Clin Drug Investig. 1998;15(1):45–55. doi:10.2165/00044011-199815010-00006 PubMed
2. de Boer T. The effects of mirtazapine on central noradrenergic and serotonergic neurotransmission. Int Clin Psychopharmacol. 1995;10
(suppl 4):19–23. doi:10.1097/00004850-199512004-00004 PubMed
3. Montgomery S, Schutte A, Reimitz P. Mirtazapine and onset of action of antidepressant activity. Int J Neuropsychopharmacol. 2000;3:249.
4. Behnke K, Søgaard J, Martin S, et al. Mirtazapine orally disintegrating tablet versus sertraline: a prospective onset of action study. J Clin Psychopharmacol. 2003;23(4):358–364. doi:10.1097/01.jcp.0000085408.08426.05 PubMed
5. de Boer T, Ruigt GS. The selective [alpha]2-adrenoceptor antagonist mirtazapine (org 3770) enhances noradrenergic and 5-HT1A-mediated serotonergic neurotransmission. CNS Drugs. 1995;4(suppl 1):29–38. doi:10.2165/00023210-199500041-00006
6. De Montigny C, Haddjeri N, Mongeau R, et al. The effects of mirtazapine on the interactions between central noradrenergic and serotonergic systems. CNS Drugs. 1995;4(suppl 1):13–17. doi:10.2165/00023210-199500041-00004
7. Bengtsson HJ, Kele J, Johansson J, et al. Interaction of the antidepressant mirtazapine with alpha2-adrenoceptors modulating the release of 5-HT in different rat brain regions in vivo. Naunyn Schmiedebergs Arch Pharmacol. 2000;362(4–5):406–412. doi:10.1007/s002100000294 PubMed
8. Barkin RL, Schwer WA, Barkin SJ. Recognition and management of depression in primary care: a focus on the elderly: a pharmacotherapeutic overview of the selection process among the traditional and new antidepressants. Am J Ther. 2000;7(3):205–226. doi:10.1097/00045391-200007030-00008 PubMed
9. de Boer T. The pharmacologic profile of mirtazapine. J Clin Psychiatry. 1996;57(suppl 4):19–25. PubMed
10. Benkert O, Szegedi A, Philipp M, et al. Mirtazapine orally disintegrating tablets versus venlafaxine extended release: a double-blind, randomized multicenter trial comparing the onset of antidepressant response in patients with major depressive disorder. J Clin Psychopharmacol. 2006;26(1):75–78. doi:10.1097/01.jcp.0000194622.99986.d6 PubMed
11. Timmer CJ, Sitsen JM, Delbressine LP. Clinical pharmacokinetics of mirtazapine. Clin Pharmacokinet. 2000;38(6):461–474. doi:10.2165/00003088-200038060-00001 PubMed
12. Bremner JD. A double-blind comparison of Org 3770, amitriptyline, and placebo in major depression. J Clin Psychiatry. 1995;56(11):519–525. PubMed
13. Zivkov M, De Jongh GD. Org 3770 versus amitriptyline: a 6-week randomized double-blind multicentre trial in hospitalized depressed patients. Human Psychopharmacology: Clinical and Experimental. 1995;10(3):173–180. doi:10.1002/hup.470100302
14. Mullin J, Lodge A, Bennie E, et al. A multicentre, double-blind, amitriptyline-controlled study of mirtazapine in patients with major depression. J Psychopharmacol. 1996;10(3):235–240. doi:10.1177/026988119601000310 PubMed
15. Halikas JA. Org 3770 (mirtazapine) versus trazodone: a placebo controlled trial in depressed elderly patients. Human Psychopharmacology: Clinical and Experimental. 1995;10(S2):S125–S133. doi:10.1002/hup.470100807
16. van Moffaert M, de Wilde J, Vereecken A, et al. Mirtazapine is more effective than trazodone: a double-blind controlled study in hospitalized patients with major depression. Int Clin Psychopharmacol. 1995;10(1):3–9. doi:10.1097/00004850-199503000-00001 PubMed
17. Stahl S, Zivkov M, Reimitz PE, et al. Meta-analysis of randomized, double-blind, placebo-controlled, efficacy and safety studies of mirtazapine versus amitriptyline in major depression. Acta Psychiatr Scand suppl. 1997;391(s391):22–30. doi:10.1111/j.1600-0447.1997.tb05955.x PubMed
18. Wheatley DP, van Moffaert M, Timmerman L, et al; Mirtazapine-Fluoxetine Study Group. Mirtazapine: efficacy and tolerability in comparison with fluoxetine in patients with moderate to severe major depressive disorder. J Clin Psychiatry. 1998;59(6):306–312. doi:10.4088/JCP.v59n0606 PubMed
19. Leinonen E, Skarstein J, Behnke K, et al; Nordic Antidepressant Study Group. Efficacy and tolerability of mirtazapine versus citalopram: a double-blind, randomized study in patients with major depressive disorder. Int Clin Psychopharmacol. 1999;14(6):329–337. doi:10.1097/00004850-199911000-00002 PubMed
20. Benkert O, Szegedi A, Kohnen R. Mirtazapine compared with paroxetine in major depression. J Clin Psychiatry. 2000;61(9):656–663. doi:10.4088/JCP.v61n0911 PubMed
21. Rodriguez AG, Ferrer CG, Odriozola VN. Efficacy and tolerability of mirtazapine versus paroxetine in the treatment of major depressive disorder. Clin Med Insights Therapeutics. 2012;4:57–63. doi:10.4137/CMT.S7632
22. Kim JE, Yoon SJ, Kim J, et al. Efficacy and tolerability of mirtazapine in treating major depressive disorder with anxiety symptoms: an 8-week open-label randomised paroxetine-controlled trial. Int J Clin Pract. 2011;65(3):323–329. doi:10.1111/j.1742-1241.2010.02624.x PubMed
23. Thase ME, Nierenberg AA, Vrijland P, et al. Remission with mirtazapine and selective serotonin reuptake inhibitors: a meta-analysis of individual patient data from 15 controlled trials of acute phase treatment of major depression. Int Clin Psychopharmacol. 2010;25(4):189–198. doi:10.1097/YIC.0b013e328330adb2 PubMed
24. Szegedi A, Müller MJ, Anghelescu I, et al. Early improvement under mirtazapine and paroxetine predicts later stable response and remission with high sensitivity in patients with major depression. J Clin Psychiatry. 2003;64(4):413–420. doi:10.4088/JCP.v64n0410 PubMed
25. Quitkin FM, Taylor BP, Kremer C. Does mirtazapine have a more rapid onset than SSRIs? J Clin Psychiatry. 2001;62(5):358–361. doi:10.4088/JCP.v62n0509 PubMed
26. Wade A, Crawford GM, Angus M, et al. A randomized, double-blind,
24-week study comparing the efficacy and tolerability of mirtazapine and paroxetine in depressed patients in primary care. Int Clin Psychopharmacol. 2003;18(3):133–141. PubMed
27. Guelfi JD, Ansseau M, Timmerman L, et al; Mirtazapine-Venlafaxine Study Group. Mirtazapine versus venlafaxine in hospitalized severely depressed patients with melancholic features. J Clin Psychopharmacol. 2001;21(4):425–431. doi:10.1097/00004714-200108000-00010 PubMed
28. Thase ME, Nierenberg AA, Keller MB, et al; Relapse Prevention Study Group. Efficacy of mirtazapine for prevention of depressive relapse: a placebo-controlled double-blind trial of recently remitted high-risk patients. J Clin Psychiatry. 2001;62(10):782–788. doi:10.4088/JCP.v62n1006 PubMed
29. Montgomery SA, Reimitz PE, Zivkov M. Mirtazapine versus amitriptyline in the long-term treatment of depression: a double-blind placebo-controlled study. Int Clin Psychopharmacol. 1998;13(2):63–73. doi:10.1097/00004850-199803000-00002 PubMed
30. Carpenter LL, Yasmin S, Price LH. A double-blind, placebo-controlled study of antidepressant augmentation with mirtazapine. Biol Psychiatry. 2002;51(2):183–188. doi:10.1016/S0006-3223(01)01262-8 PubMed
31. Fava M, Rush AJ, Wisniewski SR, et al. A comparison of mirtazapine and nortriptyline following two consecutive failed medication treatments for depressed outpatients: a STAR*D report. Am J Psychiatry. 2006;163(7):1161–1172. doi:10.1176/appi.ajp.163.7.1161 PubMed
32. Hirschfeld RMA. The use of mirtazapine in difficult-to-treat patient populations. Hum Psychopharmacol. 2002;17(suppl 1):S33–S36. doi:10.1002/hup.387 PubMed
33. Versiani M, Moreno R, Ramakers-van Moorsel CJA, et al; Comparative Efficacy Antidepressants Study Group. Comparison of the effects of mirtazapine and fluoxetine in severely depressed patients. CNS Drugs. 2005;19(2):137–146. doi:10.2165/00023210-200519020-00004 PubMed
34. Morris DW, Budhwar N, Husain M, et al. Depression treatment in patients with general medical conditions: results from the CO-MED trial. Ann Fam Med. 2012;10(1):23–33. doi:10.1370/afm.1316 PubMed
35. Kang E-H, Lee IS, Chung SK, et al. Mirtazapine versus venlafaxine for the treatment of somatic symptoms associated with major depressive disorder: a randomized, open-labeled trial. Psychiatry Res. 2009;169(2):118–123. doi:10.1016/j.psychres.2008.06.021 PubMed
36. Dowlati Y, Herrmann N, Swardfager WL, et al. Efficacy and tolerability of antidepressants for treatment of depression in coronary artery disease: a meta-analysis. Can J Psychiatry. 2010;55(2):91–99. PubMed
37. de Jonge P, Honig A, van Melle JP, et al; MIND-IT Investigators. Nonresponse to treatment for depression following myocardial infarction: association with subsequent cardiac events. Am J Psychiatry. 2007;164(9):1371–1378. doi:10.1176/appi.ajp.2007.06091492 PubMed
38. Honig A, Kuyper AM, Schene AH, et al; MIND-IT investigators. Treatment of post-myocardial infarction depressive disorder: a randomized, placebo-controlled trial with mirtazapine. Psychosom Med. 2007;69(7):606–613. doi:10.1097/PSY.0b013e31814b260d PubMed
39. Tulner DM, Smith OR, Schins A, et al. Antidepressive effect of
mirtazapine in post-myocardial infarction depression is associated with soluble TNF-R1 increase: data from the MIND-IT. Neuropsychobiology. 2011;63(3):169–176. doi:10.1159/000321624 PubMed
40. Niedermaier N, Bohrer E, Schulte K, et al. Prevention and treatment of poststroke depression with mirtazapine in patients with acute stroke. J Clin Psychiatry. 2004;65(12):1619–1623. doi:10.4088/JCP.v65n1206 PubMed
41. Kühn K-U, Quednow BB, Thiel M, et al. Antidepressive treatment in patients with temporal lobe epilepsy and major depression: a prospective study with three different antidepressants. Epilepsy Behav. 2003;4(6):674–679. doi:10.1016/j.yebeh.2003.08.009 PubMed
42. Altintoprak AE, Zorlu N, Coskunol H, et al. Effectiveness and tolerability of mirtazapine and amitriptyline in alcoholic patients with co-morbid depressive disorder: a randomized, double-blind study. Hum Psychopharmacol. 2008;23(4):313–319. doi:10.1002/hup.935 PubMed
43. Afshar M, Knapp CM, Sarid-Segal O, et al. The efficacy of mirtazapine in the treatment of cocaine dependence with comorbid depression. Am J Drug Alcohol Abuse. 2012;38(2):181–186. doi:10.3109/00952990.2011.644002 PubMed
44. Varia I, Venkataraman S, Hellegers C, et al. Effect of mirtazapine orally disintegrating tablets on health-related quality of life in elderly depressed patients with comorbid medical disorders: a pilot study. Psychopharmacol Bull. 2007;40(1):47–56. PubMed
45. Roose SP, Nelson JC, Salzman C, et al; Mirtazapine in the Nursing Home Study Group. Open-label study of mirtazapine orally disintegrating tablets in depressed patients in the nursing home. Curr Med Res Opin. 2003;19(8):737–746. doi:10.1185/030079903125002441 PubMed
46. Schatzberg AF, Kremer C, Rodrigues HE, et al; Mirtazapine vs Paroxetine Study Group. Double-blind, randomized comparison of mirtazapine and paroxetine in elderly depressed patients. Am J Geriatr Psychiatry. 2002;10(5):541–550. PubMed
47. Høyberg OJ, Maragakis B, Mullin J, et al. A double-blind multicentre comparison of mirtazapine and amitriptyline in elderly depressed patients. Acta Psychiatr Scand. 1996;93(3):184–190. doi:10.1111/j.1600-0447.1996.tb10629.x PubMed
48. Banerjee S, Hellier J, Dewey M, et al. Sertraline or mirtazapine for depression in dementia (HTA-SADD): a randomised, multicentre, double-blind, placebo-controlled trial. Lancet. 2011;378(9789):403–411. doi:10.1016/S0140-6736(11)60830-1 PubMed
49. Cakir S, Kulaksizoglu IB. The efficacy of mirtazapine in agitated patients with Alzheimer’s disease: a 12-week open-label pilot study. Neuropsychiatr Dis Treat. 2008;4(5):963–966. doi:10.2147/NDT.S3201 PubMed
50. Liappas J, Paparrigopoulos T, Tzavellas E, et al. Alcohol detoxification and social anxiety symptoms: a preliminary study of the impact of mirtazapine administration. J Affect Disord. 2003;76(1–3):279–284. doi:10.1016/S0165-0327(02)00094-0 PubMed
51. Liappas J, Paparrigopoulos T, Malitas P, et al. Mirtazapine improves alcohol detoxification. J Psychopharmacol. 2004;18(1):88–93. doi:10.1177/0269881104040241 PubMed
52. Liappas J, Paparrigopoulos T, Tzavellas E, et al. Mirtazapine and venlafaxine in the management of collateral psychopathology during alcohol detoxification. Prog Neuropsychopharmacol Biol Psychiatry. 2005;29(1):55–60. doi:10.1016/j.pnpbp.2004.10.005 PubMed
53. Colfax GN, Santos GM, Das M, et al. Mirtazapine to reduce methamphetamine use: a randomized controlled trial. Arch Gen Psychiatry. 2011;68(11):1168–1175. doi:10.1001/archgenpsychiatry.2011.124 PubMed
54. Cruickshank CC, Montebello ME, Dyer KR, et al. A placebo-controlled trial of mirtazapine for the management of methamphetamine withdrawal. Drug Alcohol Rev. 2008;27(3):326–333. doi:10.1080/09595230801935672 PubMed
55. McGregor C, Srisurapanont M, Mitchell A, et al. Symptoms and sleep patterns during inpatient treatment of methamphetamine withdrawal: a comparison of mirtazapine and modafinil with treatment as usual. J Subst Abuse Treat. 2008;35(3):334–342. doi:10.1016/j.jsat.2007.12.003 PubMed
56. Boshuisen ML, Slaap BR, Vester-Blokland ED, et al. The effect of mirtazapine in panic disorder: an open label pilot study with a single-blind placebo run-in period. Int Clin Psychopharmacol. 2001;16(6):363–368. doi:10.1097/00004850-200111000-00008 PubMed
57. Ribeiro L, Busnello JV, Kauer-Sant’Anna M, et al. Mirtazapine versus fluoxetine in the treatment of panic disorder. Braz J Med Biol Res. 2001;34(10):1303–1307. doi:10.1590/S0100-879X2001001000010 PubMed
58. Sarchiapone M, Amore M, De Risio S, et al. Mirtazapine in the treatment of panic disorder: an open-label trial. Int Clin Psychopharmacol. 2003;18(1):35–38. doi:10.1097/00004850-200301000-00006 PubMed
59. Bahk W-M, Pae CU, Tsoh J, et al. Effects of mirtazapine in patients with post-traumatic stress disorder in Korea: a pilot study. Hum Psychopharmacol. 2002;17(7):341–344. doi:10.1002/hup.426 PubMed
60. Davidson JRT, Weisler RH, Butterfield MI, et al. Mirtazapine vs placebo in posttraumatic stress disorder: a pilot trial. Biol Psychiatry. 2003;53(2):188–191. doi:10.1016/S0006-3223(02)01411-7 PubMed
61. Chung MY, Min KH, Jun YJ, et al. Efficacy and tolerability of mirtazapine and sertraline in Korean veterans with posttraumatic stress disorder:
a randomized open label trial. Hum Psychopharmacol. 2004;19(7):
489–494. doi:10.1002/hup.615 PubMed
62. Seo H-J, Jung Y-E, Bank W-M, et al. A comparison of mirtazapine and paroxetine for the treatment of patients with posttraumatic stress disorder: a randomized open-label trial. Clin Psychopharmacol Neurosci. 2010;8(2):84–89.
63. Alderman CP, Condon JT, Gilbert AL. An open-label study of mirtazapine as treatment for combat-related PTSD. Ann Pharmacother. 2009;43(7):1220–1226. doi:10.1345/aph.1M009 PubMed
64. Koran LM, Gamel NN, Choung HW, et al. Mirtazapine for obsessive-compulsive disorder: an open trial followed by double-blind discontinuation. J Clin Psychiatry. 2005;66(4):515–520. doi:10.4088/JCP.v66n0415 PubMed
65. Pallanti S, Quercioli L, Bruscoli M. Response acceleration with mirtazapine augmentation of citalopram in obsessive-compulsive disorder patients without comorbid depression: a pilot study. J Clin Psychiatry. 2004;65(10):1394–1399. doi:10.4088/JCP.v65n1015 PubMed
66. Gambi F, De Berardis D, Campanella D, et al. Mirtazapine treatment of generalized anxiety disorder: a fixed dose, open label study. J Psychopharmacol. 2005;19(5):483–487. doi:10.1177/0269881105056527 PubMed
67. Muehlbacher M, Nickel MK, Nickel C, et al. Mirtazapine treatment of social phobia in women: a randomized, double-blind, placebo-controlled study. J Clin Psychopharmacol. 2005;25(6):580–583. doi:10.1097/01.jcp.0000186871.04984.8d PubMed
68. Van Veen JF, Van Vliet IM, Westenberg HGM. Mirtazapine in social anxiety disorder: a pilot study. Int Clin Psychopharmacol. 2002;17(6):315–317. doi:10.1097/00004850-200211000-00008 PubMed
69. Schutters SIJ, Van Megen HJGM, Van Veen JF, et al. Mirtazapine in generalized social anxiety disorder: a randomized, double-blind, placebo-controlled study. Int Clin Psychopharmacol. 2010;25(5):302–304. doi:10.1097/YIC.0b013e32833a4d71 PubMed
70. Schutters SI, van Megen HJ, Van Veen JF, et al. Paroxetine augmentation in patients with generalised social anxiety disorder, non-responsive to mirtazapine or placebo. Hum Psychopharmacol. 2011;26(1):72–76. doi:10.1002/hup.1165 PubMed
71. Major EO, Amemiya K, Tornatore CS, et al. Pathogenesis and molecular biology of progressive multifocal leukoencephalopathy, the JC virus-induced demyelinating disease of the human brain. Clin Microbiol Rev. 1992;5(1):49–73. PubMed
72. Elphick GF, Querbes W, Jordan JA, et al. The human polyomavirus, JCV, uses serotonin receptors to infect cells. Science. 2004;306(5700):1380–1383. doi:10.1126/science.1103492 PubMed
73. Cettomai D, McArthur JC. Mirtazapine use in human immunodeficiency virus-infected patients with progressive multifocal leukoencephalopathy. Arch Neurol. 2009;66(2):255–258. doi:10.1001/archneurol.2008.557 PubMed
74. Moenster RP, Jett RA. Mirtazapine and mefloquine therapy for progressive multifocal leukoencephalopathy in a patient infected with human immunodeficiency virus. Am J Health Syst Pharm. 2012;69(6):496–498. doi:10.2146/ajhp110392 PubMed
75. Lima MA, Auriel E, Wüthrich C, et al. Progressive multifocal leukoencephalopathy as a complication of hepatitis C virus treatment in an HIV-negative patient. Clin Infect Dis. 2005;41(3):417–419. doi:10.1086/431769 PubMed
76. Clifford DB, De Luca A, Simpson DM, et al. Natalizumab-associated progressive multifocal leukoencephalopathy in patients with multiple sclerosis: lessons from 28 cases. Lancet Neurol. 2010;9(4):438–446. doi:10.1016/S1474-4422(10)70028-4 PubMed
77. Pact V, Giduz T. Mirtazapine treats resting tremor, essential tremor, and levodopa-induced dyskinesias. Neurology. 1999;53(5):1154. doi:10.1212/WNL.53.5.1154-a PubMed
78. Pahwa R, Lyons KE. Mirtazapine in essential tremor: a double-blind, placebo-controlled pilot study. Mov Disord. 2003;18(5):584–587. doi:10.1002/mds.10371 PubMed
79. Uccellini D, Grampa G, La Spina I, et al. Mirtazapine in the treatment of essential tremor: an open-label, observer-blind study. Neuropsychiatr Dis Treat. 2006;2(1):95–100. PubMed
80. Deuschl G, Raethjen J, Hellriegel H, et al. Treatment of patients with essential tremor. Lancet Neurol. 2011;10(2):148–161. doi:10.1016/S1474-4422(10)70322-7 PubMed
81. Bendtsen L, Jensen R. Mirtazapine is effective in the prophylactic treatment of chronic tension-type headache. Neurology. 2004;62(10):1706–1711. doi:10.1212/01.WNL.0000127282.90920.8C PubMed
82. Bendtsen L, Jensen R, Olesen J. A non-selective (amitriptyline), but not a selective (citalopram), serotonin reuptake inhibitor is effective in the prophylactic treatment of chronic tension-type headache. J Neurol Neurosurg Psychiatry. 1996;61(3):285–290. doi:10.1136/jnnp.61.3.285 PubMed
83. Bendtsen L, Buchgreitz L, Ashina S, et al. Combination of low-dose mirtazapine and ibuprofen for prophylaxis of chronic tension-type headache. Eur J Neurol. 2007;14(2):187–193. doi:10.1111/j.1468-1331.2006.01607.x PubMed
84. Arnold P, Vuadens P, Kuntzer T, et al. Mirtazapine decreases the pain feeling
in healthy participants. Clin J Pain. 2008;24(2):116–119. doi:10.1097/AJP.0b013e318159f94a PubMed
85. Theobald DE, Kirsh KL, Holtsclaw E, et al. An open-label, crossover trial of mirtazapine (15 and 30 mg) in cancer patients with pain and other distressing symptoms. J Pain Symptom Manage. 2002;23(5):442–447. doi:10.1016/S0885-3924(02)00381-0 PubMed
86. Samborski W, Lezanska-Szpera M, Rybakowski JK. Open trial of mirtazapine in patients with fibromyalgia. Pharmacopsychiatry. 2004;37(4):168–170. doi:10.1055/s-2004-827172 PubMed
87. Davis R, Wilde MI. Mirtazapine: a review of its pharmacology and therapeutic potential in the management of major depression. CNS Drugs. 1996;5(5):389–402. doi:10.2165/00023210-199605050-00007
88. Sussman N, Ginsberg DL. Weight effects of nefazodone, bupropion, mirtazapine, and venlafaxine: a review of available evidence. Prim Psychiatry. 2000;7:33–48.
89. Riechelmann RP, Burman D, Tannock IF, et al. Phase II trial of mirtazapine for cancer-related cachexia and anorexia. Am J Hosp Palliat Care. 2010;27(2):106–110. doi:10.1177/1049909109345685 PubMed
90. Boas S, McColley S, Danduran M, et al. The role of mirtazapine as an appetite stimulant in malnourished individuals with CF. Pediatr Pulmonol. 2000;30:325–326.
91. Sykes R, Kittel F, Marcus M, et al. Mirtazapine for appetite stimulation in children with cystic fibrosis. Pediatr Pulmonol. 2006;59:389–390.
92. Chinuck RS, Fortnum H, Baldwin DR. Appetite stimulants in cystic fibrosis: a systematic review. J Hum Nutr Diet. 2007;20(6):526–537. doi:10.1111/j.1365-277X.2007.00824.x PubMed
93. Himmerich H, Fulda S, Schaaf L, et al. Changes in weight and glucose tolerance during treatment with mirtazapine. Diabetes Care. 2006;29(1):170–171. doi:10.2337/diacare.29.1.170 PubMed
94. Stimmel GL, Sussman N, Wingard P. Mirtazapine safety and tolerability: analysis of the clinical trials database. Prim Psychiatry. 1997;4:82–90.
95. Radhakishun FS, van den Bos J, van der Heijden BC, et al. Mirtazapine effects on alertness and sleep in patients as recorded by interactive telecommunication during treatment with different dosing regimens. J Clin Psychopharmacol. 2000;20(5):531–537. doi:10.1097/00004714-200010000-00006 PubMed
96. Aslan S, Isik E, Cosar B. The effects of mirtazapine on sleep: a placebo controlled, double-blind study in young healthy volunteers. Sleep. 2002;25(6):677–679. PubMed
97. Winokur A, DeMartinis NA 3rd, McNally DP, et al. Comparative effects
of mirtazapine and fluoxetine on sleep physiology measures in patients with major depression and insomnia. J Clin Psychiatry. 2003;64(10):1224–1229. doi:10.4088/JCP.v64n1013 PubMed
98. Carley DW, Olopade C, Ruigt GS, et al. Efficacy of mirtazapine in obstructive sleep apnea syndrome. Sleep. 2007;30(1):35–41. PubMed
99. Marshall NS, Yee BJ, Desai AV, et al. Two randomized placebo-controlled trials to evaluate the efficacy and tolerability of mirtazapine for the treatment of obstructive sleep apnea. Sleep. 2008;31(6):824–831. PubMed
100. Saks BR. Mirtazapine: treatment of depression, anxiety, and hyperemesis gravidarum in the pregnant patient: a report of 7 cases. Arch Women Ment Health. 2001;3(4):165–170. doi:10.1007/s007370170014
101. Rohde A, Dembinski J, Dorn C. Mirtazapine (Remergil) for treatment resistant hyperemesis gravidarum: rescue of a twin pregnancy. Arch Gynecol Obstet. 2003;268(3):219–221. doi:10.1007/s00404-003-0502-0 PubMed
102. Chen CC, Lin CS, Ko YP, et al. Premedication with mirtazapine reduces preoperative anxiety and postoperative nausea and vomiting. Anesth Analg. 2008;106(1):109–113. doi:10.1213/01.ane.0000289636.09841.bc PubMed
103. Chang F-L, Ho S-T, Sheen MJ. Efficacy of mirtazapine in preventing intrathecal morphine-induced nausea and vomiting after orthopaedic surgery. Anaesthesia. 2010;65(12):1206–1211. doi:10.1111/j.1365-2044.2010.06561.x PubMed
104. Saiz-Ruiz J, Montes JM, Ibáñez A, et al. Assessment of sexual functioning in depressed patients treated with mirtazapine: a naturalistic 6-month study. Hum Psychopharmacol. 2005;20(6):435–440. doi:10.1002/hup.706 PubMed
105. Michelson D, Kociban K, Tamura R, et al. Mirtazapine, yohimbine or olanzapine augmentation therapy for serotonin reuptake-associated female sexual dysfunction: a randomized, placebo controlled trial. J Psychiatr Res. 2002;36(3):147–152. doi:10.1016/S0022-3956(01)00060-7 PubMed
106. Schutters SI, van Megen HJ, Van Veen JF, et al. Paroxetine augmentation in patients with generalised social anxiety disorder, non-responsive to mirtazapine or placebo. Hum Psychopharmacol. 2011;26(1):72–76. doi:10.1002/hup.1165 PubMed
107. Waldinger MD, Zwinderman AH, Olivier B. Antidepressants and ejaculation: a double-blind, randomized, fixed-dose study with mirtazapine and paroxetine. J Clin Psychopharmacol. 2003;23(5):467–470. doi:10.1097/01.jcp.0000088904.24613.e4 PubMed
Enjoy this premium PDF as part of your membership benefits!


