Background: The greater severity and burden of illness in individuals with early onset schizophrenia (ie, before age 18 years) deserves further investigation, specifically regarding its prevalence in community-based treatment and its association with other psychiatric or medical conditions.
Method: A retrospective cohort design was employed using the South Carolina Medicaid claims database covering outpatient and inpatient medical services from January 1, 1999, through December 31, 2013, to identify patients aged ≤ 17 years with a diagnosis of schizophrenia spectrum disorders (ICD-9-CM). Logistic regression was used to examine the factors differentiating childhood- versus adolescent-onset schizophrenia in a community-based system of care.
Results: Early onset schizophrenia was diagnosed in 613 child and adolescent cases during the study epoch or 0.2% of this population-based cohort. The early onset cohort was primarily male (64%) and black (48%). The mean length of time followed in the Medicaid dataset was 12.6 years. Within the early onset cohort, 22.5% were diagnosed at age ≤ 12 years and 77.5% were diagnosed as adolescents. The childhood-onset subgroup was twice as likely to have speech, language, or educational disabilities and an attention-deficit/hyperactivity disorder diagnosis but significantly less likely to have schizophrenia or schizoaffective disorder, an organic brain disorder or mental retardation/intellectual disability, or a substance use disorder (adjusted OR = 2.01, 2.26, 0.38, 0.31, 0.47, and 0.32, respectively) compared to the adolescent-onset subgroup.
Conclusion: Primary care providers should identify and maintain surveillance of cases of pediatric neurodevelopmental disorders, which appear to be highly comorbid and genetically related, and refer them early and promptly for specialized treatment.
Factors Differentiating Childhood-Onset
and Adolescent-Onset Schizophrenia:
A Claims Database Study
ABSTRACT
Background: The greater severity and burden of illness in individuals with early onset schizophrenia (ie, before age 18 years) deserves further investigation, specifically regarding its prevalence in community-based treatment and its association with other psychiatric or medical conditions.
Method: A retrospective cohort design was employed using the South Carolina Medicaid claims database covering outpatient and inpatient medical services from January 1, 1999, through December 31, 2013, to identify patients aged ≤ 17 years with a diagnosis of schizophrenia spectrum disorders (ICD-9-CM). Logistic regression was used to examine the factors differentiating childhood- versus adolescent-onset schizophrenia in a community-based system of care.
Results: Early onset schizophrenia was diagnosed in 613 child and adolescent cases during the study epoch or 0.2% of this population-based cohort. The early onset cohort was primarily male (64%) and black (48%). The mean length of time followed in the Medicaid dataset was 12.6 years. Within the early onset cohort, 22.5% were diagnosed at age ≤ 12 years and 77.5% were diagnosed as adolescents. The childhood-onset subgroup was twice as likely to have speech, language, or educational disabilities and an attention-deficit/hyperactivity disorder diagnosis but significantly less likely to have schizophrenia or schizoaffective disorder, an organic brain disorder or mental retardation/intellectual disability, or a substance use disorder (adjusted OR = 2.01, 2.26, 0.38, 0.31, 0.47, and 0.32, respectively) compared to the adolescent-onset subgroup.
Conclusion: Primary care providers should identify and maintain surveillance of cases of pediatric neurodevelopmental disorders, which appear to be highly comorbid and genetically related, and refer them early and promptly for specialized treatment.
Prim Care Companion CNS Disord 2016;18(2):doi:10.4088/PCC.15m01901
© Copyright 2016 Physicians Postgraduate Press, Inc.
aDepartment of Neuropsychiatry and Behavioral Science, University of South Carolina School of Medicine, Columbia
bDepartments of Psychiatry and Pharmacology, University of Toronto, Toronto, Canada
*Corresponding author: Jeanette M. Jerrell, PhD, Department of Neuropsychiatry and Behavioral Science, University of South Carolina School of Medicine,
15 Medical Park, Ste 301, Columbia, SC 29203
([email protected]).
About 75% of all mental disorders have observable characteristics that manifest before the age of 25 years, including schizophrenia and bipolar disorder.1 Over time, the disease progression of schizophrenia leads to a slow decline in cognitive function, marked personality change, social isolation, occupational disability, poor cardiometabolic health, and various forms of morbidity and mortality.2 In 2010, schizophrenia accounted for 7.4% of disability-adjusted life-years caused by mental and substance use disorders, with the highest proportion of total disability-adjusted life-years occurring in young people aged 10–29 years.3
Furthermore, recent prevalence studies of early onset schizophrenia in children and adolescents are relatively scarce.4–7 Population-based incidence rates reported previously in children and adolescents for early onset schizophrenia have ranged widely from 0.018%4 to 0.0052%5 to 1.8%.6 Although not based on epidemiologic methods, an incidence rate of ≤ 0.04% was reported by National Institute of Mental Health (NIMH) researchers7 for the “most homogeneous form” of childhood-onset schizophrenia. Gender differences have been reported, but were not consistent across studies.4,5 Moreover, in-depth clinical characterization by NIMH researchers7 points to premorbid pervasive developmental disorder/autism spectrum disorder (PDD/ASD) and disturbances in social, motor, and language functional domains, as well as learning disabilities, and comorbid mood and anxiety disorders as important covariates to investigate. As the symptoms and syndromes of schizophrenia are continuous (ie, the same) across age groups, the primary differentiating factors between childhood-onset and adolescent-onset schizophrenia are likely to be the comorbid conditions developing in these children over time and their age at onset. Since none of the previous epidemiologic studies examined the influence of comorbidities, their presence in childhood- and adolescent-onset cases needs to be systematically replicated and better understood in a population-based, heterogeneous cohort.
Given the substantially higher rates of long-term impairment and morbidity associated with age at onset of schizophrenia, this area of inquiry deserves further investigation. In this analysis, we compare the demographic and comorbidity correlates for incident schizophrenia in childhood versus adolescence and elucidate the potential significance of these factors in earlier diagnosis and treatment of affected individuals in community-based care systems.
METHOD
Subject Ascertainment Procedures
Data for this study were obtained retrospectively from the South Carolina Medicaid database during a 15-year period from January 1, 1999, through December 31, 2013. Medical claims were used to identify a service encounter, date of service, and the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) diagnosis codes related to that visit. Selection criteria were aged ≤ 17 years, continuous enrollment in Medicaid for a minimum of 9 months in each calendar year, and at least 1 service encounter with an ICD-9-CM diagnostic code of schizophrenia spectrum disorders (295.xx). These files were subjected to a wide variety of data-checking analyses as well as procedures to maximize the integrity of the data and minimize missing data. The methods involved in this study were approved by the University of South Carolina Institutional Review Board as exempt from human subject research guidelines (45 Code of Federal Regulations part 46).
For all individuals meeting the inclusion criteria for this early onset cohort, the following categories of diagnosed conditions were evaluated as being present or not present in each child’s medical service history due to their association with schizophrenia spectrum disorders: organic psychosis (291.xx–294.xx); affective psychoses, major depressive disorder (MDD), or bipolar disorder (296.xx); paranoid, delusional, or other nonorganic psychoses (297.xx–298.xx); PDD/ASD (299.xx), anxiety (300.0–300.09); conduct disorder/oppositional defiant disorder (CD/ODD) (309.3, 312xx, 313.81); attention-deficit/hyperactivity disorder (ADHD) (314.0–2); mental retardation/intellectual disability (MR/ID) (317, 318.x, 319); speech, language, or educational disabilities (315.xx); cerebrovascular and central nervous system disorders (320.x–326.x, 330.x–337.x, 340.x–344.x); epilepsy (345.x), seizure disorder (780.3); organic brain disorders (310.xx); traumatic brain injury (850.x–854.x); chromosomal abnormalities and syndromes (277.2, 279.11, 282.6, 334.x, 747.29, 754.8, 756.83, 758.0–1, 758.6, 759.3, 759.5, 759.81–2, 759.89); or a substance use disorder (304.xx–30.5xx).8–14
Statistical Analyses
Descriptive statistical analyses were performed to determine the prevalence and incidence of each comorbid psychiatric or medical condition and any bivariate likelihood ratio associations (χ2 and P value) between the independent predictor variables of interest and the dependent variable: age at first diagnosis of schizophrenia spectrum disorders.
To address our research question regarding the factors that are significantly associated with a higher risk of a child being diagnosed with childhood-onset schizophrenia versus adolescent-onset schizophrenia, a multiple logistic regression equation (full model) was constructed to assess the relative odds associated with having any of the independently diagnosed conditions, including individual risk and demographic factors (dichotomously coded sex and ethnicity), and comorbid psychiatric or medical disorders (dichotomously coded as diagnosed or not diagnosed in the child’s medical history: schizophrenia, schizoaffective disorder, organic psychosis, affective psychoses, MDD, or bipolar disorder; paranoid, delusional, or other nonorganic psychoses; PDD/ASD, anxiety; CD/ODD; ADHD; MR/ID; speech, language, educational disabilities; cerebrovascular and central nervous system disorders; epilepsy, seizures; organic brain disorders; traumatic brain injury; chromosomal abnormalities and syndromes; or substance use disorder). The full regression model was then reduced through a stepwise procedure, using a preset significance level of P ≤ .05, to reflect only the statistically significant variables associated with being diagnosed with childhood-onset schizophrenia. The measure of association reported for these multivariate results is the adjusted odds ratio (OR) with a corresponding 95% confidence interval, and all statistical analyses were performed using SAS version 9.3 (SAS Institute, Cary, North Carolina).
RESULTS
Early onset schizophrenia was diagnosed in 613 child and adolescent cases during the study epoch or 0.2% of this population-based cohort (total population of Medicaid cases ≤ 17 reviewed was N = 278,067 for the defined study period). Descriptive results regarding this cohort (Table 1) indicate that the early onset schizophrenia cohort was primarily male (64%) and black (48%), with a mean age at diagnosis of 14.1 years (SD = 3.0 years, median = 15). The mean length of time included in these patient clinical trajectories (being followed in the Medicaid dataset) was 12.6 years (SD = 3.6 years). Within the early onset schizophrenia cohort, 22.5% were diagnosed at ≤ 12 years of age and 77.5% were diagnosed as adolescents. Bivariate statistical results, ie, the unadjusted likelihood ratios and P values, for the comorbid psychiatric disorders and medical conditions diagnosed as present in the early onset schizophrenia cohort are noted in Table 1.
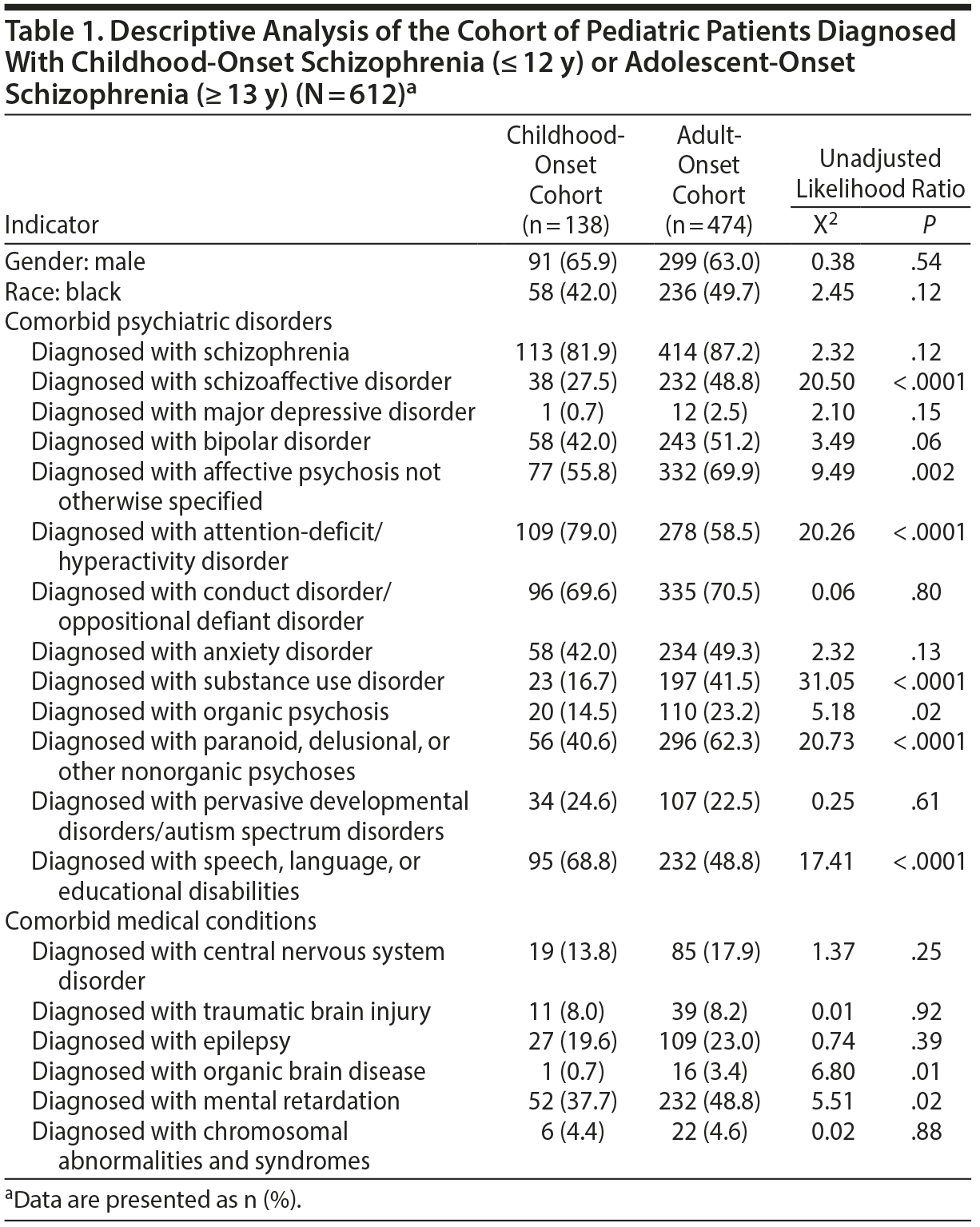
Comparing Childhood-Onset and Adolescent-Onset Schizophrenia Diagnostic Features
In the logistic regression analysis, patients in the childhood-onset schizophrenia subgroup were twice as likely to have speech, language, or educational disabilities and an ADHD diagnosis but were significantly less likely to have a schizophrenia or schizoaffective diagnosis, an organic brain disorder or MR/ID, or a substance use disorder diagnosis (adjusted OR = 2.01, 2.26, 0.38, 0.31, 0.47, and 0.32, respectively) (Table 2) compared to the adolescent-onset schizophrenia subgroup.
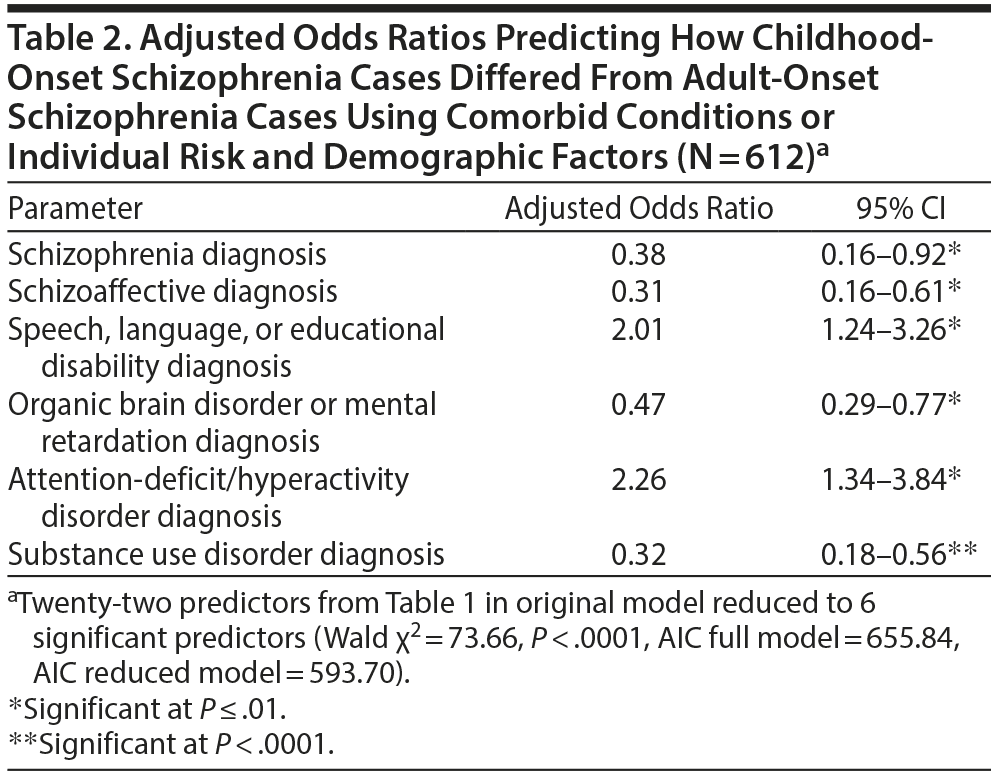
DISCUSSION
Early onset schizophrenia was diagnosed in 0.2% of this population-based cohort at a mean age of 14.1 years, which is generally in the midrange of incidence rates reported previously in children and adolescents: 0.018%,4 0.0052%,5 and 1.8%.6 Our childhood-onset schizophrenia incidence rate of 0.05% (n = 138) was more comparable to the ≤ 0.04% rate noted by NIMH researchers.7 The basic demographics of our early onset schizophrenia cohort (ie, primarily male and ≤ 15 years old at diagnosis) are also similar to previous early onset cohorts, with the exception of the 1994–2010 primarily female Danish subgroup in the study by Okkels et al.5 The discrepancies between our findings and those of other investigators may be due to methodological differences, as the previous cohorts were ascertained in national registries, school settings, or highly specialized care clinics, whereas our cohort was composed of cases referred by primary care physicians, nurse practitioners, or family members to public mental health treatment in the United States.
Our childhood-onset subgroup was twice as likely to have speech, language, or educational disabilities and an ADHD diagnosis, whereas the adolescent-onset subgroup was significantly more likely to have a diagnosis of schizophrenia or schizoaffective disorder, a substance use disorder diagnosis, or an organic brain disorder or MR/ID. These foregoing differences comport with previous reports in the literature that although clinicians may be reluctant to code a schizophrenia spectrum or mood disorder diagnosis in children, early speech, language, or educational disabilities and attentional or hyperactivity problems are being recognized.14–16 A formal ADHD diagnosis is often noted as the first indication that an individual’s development is “off course” in children with otherwise normal development trajectories. In addition, PDD/ASD or intellectual disabilities and the associated behaviors, eg, uncontrollable, disruptive or self-harmful behavior, may be the primary reason for a referral from primary care to specialty mental health services. Once in specialized care, more severe comorbid psychiatric disorders can be recognized, such as schizophrenia or schizoaffective disorder, a substance use disorder, an organic brain disorder, or MR/ID. Our findings also indicate that schizoaffective disorder and symptoms may be much more prevalent in population-based early onset schizophrenia cohorts than was apparent in the NIMH study.7
Conceptualized as a neurodevelopmental abnormality (ie, an illness that has its origins in disturbed central nervous system development), schizophrenia may be viewed as just one manifestation of a wider group of overlapping syndromes.17 Genetic studies have found evidence of significant sharing of relatively common risk variants between schizophrenia and bipolar disorder, schizophrenia and MDD, ADHD and MDD, schizophrenia and intellectual disability, and schizophrenia and PDD/ASD.18 These foregoing syndromes may manifest at different times in an individual’s growth trajectory and in relation to their severity or impact on brain structure and functioning. However, intensive diagnostic, neuroimaging, and genetic studies over the past 2 decades have not yielded a reliable premorbid phenotype for early onset schizophrenia and no single therapeutic target or biomarker that can reliably distinguish patients with particular diagnostic disorders or healthy controls from clinical populations.7,19
Although the knowledge base in this area of inquiry has expanded greatly over the past 2 decades and the combined overall risk of a child developing a severe mental or emotional disorder appears to be less than 10%, treatment of these disorders may be delayed for up to 10 years.1,20 The declaration of a severe and persisting brain disorder (eg, schizophrenia) may be preceded by nonspecific symptomatology that is often pleiomorphic in presentation (ie, able to change in size and shape as a function of where the child is along the developmental trajectory and in relation to environmental forces). The pleiomorphic phenomenology that often predates the requisite symptoms of schizophrenia is one of several factors that contribute to significant diagnostic delay. The diagnostic delay and, consequently, the progression of the underlying illness invites the need for a more refined understanding of the phenomenology of early onset brain illnesses among primary care and specialty practitioners. Recognition of the constellation of comorbid psychiatric conditions associated with childhood-onset schizophrenia as demonstrated herein may assist primary care and specialty practitioners to identify and diagnose this progressive illness earlier and mitigate its severity over time.
Strengths and Limitations
The cohort represents a relatively heterogeneous group of children and adolescents diagnosed with schizophrenia. Although observational (Medicaid) databases provide much less detailed information on individuals than would a structured research interview, the coded medical and psychiatric diagnoses made by licensed physicians or nurse practitioners and patient utilization data are more reliable than patient or family self-reports.21 Moreover, administrative claims data have been used to accurately identify positive predictive values for diagnostic patterns and clinical outcomes, such as antipsychotic treatment in children.22 Furthermore, the cohort is generally representative of pediatric patients in routine care settings in the southeastern US states and other small states with predominantly small-city and rural populations in terms of age, sex, racial demographics, and Medicaid eligibility, but our results may not be generalizable to other patient groups.23–25
Several limitations also must be considered. No structured research or clinical interviews were employed. Identification of psychiatric and nonpsychiatric medical conditions was based on spontaneous self-reporting to or observation by a primary care physician or psychiatrist and their accurate designation of each diagnosis in the Medicaid billing system; consequently, the prevalence of these conditions may be an underestimate, which we cannot quantify. Data regarding established risk factors, such as family history of psychiatric disorders, were not available to the investigators and are not modeled in these analyses. Children and adolescents who dropped out of treatment, who were periodically ineligible for Medicaid, or who died before they were registered in Medicaid coverage are not represented in this dataset. Finally, these results report associations, and, as a result, directions of causality cannot be inferred.
CONCLUSION
It is important that primary care practitioners identify and maintain vigilant surveillance of any patients with symptoms of the pediatric neurodevelopmental disorders, which appear to be highly comorbid and genetically related, and refer them early and promptly for specialized treatment.
Submitted: October 29, 2015; accepted January 11, 2016.
Published online: March 3, 2016.
Potential conflicts of interest: Dr Jerrell has received research grants from and served on national advisory boards for National Institutes of Health, Eli Lilly, and Bristol-Myers Squibb/Otsuka. Dr McIntyre has received honoraria for speaking and consulting from AstraZeneca, Janssen-Ortho, Actavis, Eli Lilly, Lundbeck, Merck, Pfizer, Bristol-Myers Squibb/Otsuka, CME Outfitters, and Physicians Postgraduate Press, Inc., and research grants from Eli Lilly, Shire, Janssen-Ortho, AstraZeneca, Lundbeck, Pfizer, Bristol-Myers Squibb/Otsuka, Forest, Sunovion, Stanley Medical Research Institute, and National Alliance for Research on Schizophrenia and Depression and has served on advisory boards for AstraZeneca, Bristol-Myers Squibb/Otsuka, Janssen-Ortho, Eli Lilly, Lundbeck, Pfizer, Merck, Sepracor, and Shire.
Funding support: None reported.
Disclaimer: The views expressed do not necessarily represent official findings of the South Carolina Department of Health and Human Services (Medicaid).
Additional information: The Medicaid dataset used in these analyses is not available to other investigators due to the confidentiality restrictions imposed by the South Carolina Department of Health and Human Services.
REFERENCES
1. Kessler RC, Berglund P, Demler O, et al. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):593–602. doi:10.1001/archpsyc.62.6.593 PubMed
2. Millier A, Schmidt U, Angermeyer MC, et al. Humanistic burden in schizophrenia: a literature review. J Psychiatr Res. 2014;54:85–93. doi:10.1016/j.jpsychires.2014.03.021 PubMed.
3. Whiteford HA, Degenhardt L, Rehm J, et al. Global burden of disease attributable to mental and substance use disorders: findings from the Global Burden of Disease Study 2010. Lancet. 2013;382(9904):1575–1586. doi:10.1016/S0140-6736(13)61611-6 PubMed
4. Häfner H, Nowotny B. Epidemiology of early onset schizophrenia. Eur Arch Psychiatry Clin Neurosci. 1995;245(2):80–92. doi:10.1007/BF02190734 PubMed
5. Okkels N, Vernal DL, Jensen SO, et al. Changes in the diagnosed incidence of early onset schizophrenia over four decades. Acta Psychiatr Scand. 2013;127(1):62–68. doi:10.1111/j.1600-0447.2012.01913.x PubMed
6. Malhotra S, Kohli A, Kapoor M, et al. Incidence of childhood psychiatric disorders in India. Indian J Psychiatry. 2009;51(2):101–107. doi:10.4103/0019-5545.49449 PubMed
7. Driver DI, Gogtay N, Rapoport JL. Childhood-onset schizophrenia and early onset schizophrenia spectrum disorders. Child Adolesc Psychiatr Clin N Am. 2013;22(4):539–555. doi:10.1016/j.chc.2013.04.001 PubMed
8. Morgan VA, Leonard H, Bourke J, et al. Intellectual disability co-occurring with schizophrenia and other psychiatric illness: population-based study. Br J Psychiatry. 2008;193(5):364–372. doi:10.1192/bjp.bp.107.044461 PubMed
9. Greenwood CM, Husted J, Bomba MD, et al. Elevated rates of schizophrenia in a familial sample with mental illness and intellectual disability. J Intellect Disabil Res. 2004;48(Pt 6):531–539. doi:10.1111/j.1365-2788.2004.00621.x PubMed
10. Rapoport J, Chavez A, Greenstein D, et al. Autism spectrum disorders and childhood-onset schizophrenia: clinical and biological contributions to a relation revisited. J Am Acad Child Adolesc Psychiatry. 2009;48(1):10–18. doi:10.1097/CHI.0b013e31818b1c63 PubMed
11. Simonoff E, Pickles A, Charman T, et al. Psychiatric disorders in children with autism spectrum disorders: prevalence, comorbidity, and associated factors in a population-derived sample. J Am Acad Child Adolesc Psychiatry. 2008;47(8):921–929. doi:10.1097/CHI.0b013e318179964f PubMed
12. Simonoff E, Pickles A, Wood N, et al. ADHD symptoms in children with mild intellectual disability. J Am Acad Child Adolesc Psychiatry. 2007;46(5):591–600. doi:10.1097/chi.0b013e3180323330 PubMed
13. Rommelse NN, Franke B, Geurts HM, et al. Shared heritability of attention-deficit/hyperactivity disorder and autism spectrum disorder. Eur Child Adolesc Psychiatry. 2010;19(3):281–295. doi:10.1007/s00787-010-0092-x PubMed
14. Nicolson R, Lenane M, Singaracharlu S, et al. Premorbid speech and language impairments in childhood-onset schizophrenia: association with risk factors. Am J Psychiatry. 2000;157(5):794–800. doi:10.1176/appi.ajp.157.5.794 PubMed
15. Jerrell JM, McIntyre RS, Park YM. Correlates of incident bipolar disorder in children and adolescents diagnosed with attention-deficit/hyperactivity disorder. J Clin Psychiatry. 2014;75(11):e1278–e1283. doi:10.4088/JCP.14m09046 PubMed
16. Jerrell JM, McIntyre RS, Park YM. Risk factors for incident major depressive disorder in children and adolescents with attention-deficit/hyperactivity disorder. Eur Child Adolesc Psychiatry. 2015;24(1):65–73. doi:10.1007/s00787-014-0541-z PubMed
17. Owen MJ, O’Donovan MC, Thapar A, et al. Neurodevelopmental hypothesis of schizophrenia. Br J Psychiatry. 2011;198(3):173–175. doi:10.1192/bjp.bp.110.084384 PubMed
18. Owen MJ. New approaches to psychiatric diagnostic classification. Neuron. 2014;84(3):564–571. doi:10.1016/j.neuron.2014.10.028 PubMed
19. Craddock N, Owen MJ. The Kraepelinian dichotomy: going, going … but still not gone. Br J Psychiatry. 2010;196(2):92–95. doi:10.1192/bjp.bp.109.073429 PubMed
20. Wang PS, Berglund P, Olfson M, et al. Failure and delay in initial treatment contact after first onset of mental disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):603–613. doi:10.1001/archpsyc.62.6.603 PubMed
21. Holmberg SK, Kane C. Health and self-care practices of persons with schizophrenia. Psychiatr Serv. 1999;50(6):827–829. doi:10.1176/ps.50.6.827 PubMed
22. Jerrell JM, Shugart MA. Community-based care for youths with early and very early onset bipolar I disorder. Bipolar Disord. 2004;6(4):299–304. doi:10.1111/j.1399-5618.2004.00129.x PubMed
23. Health Insurance Coverage for Children
Under 19 Years of Age, at or Below 200% of Poverty by State, 2006–2008 and 2009–2011. United States Census Bureau Web site. http://www.census.gov/hhes/www/hlthins/data/children/low-income.html. Updated September 17, 2013. Accessed
January 30, 2013.
24. Tables D1. Children Under 18 and Their Designated Parents and Table D2: Children Under 18 and Their Designated Parents: Characteristics of Families (including region). United States Census Bureau Web site. http://www.census.gov/hhes/socdemo/children/data/sipp/well2009/tables.html. Updated August 11, 2011. Accessed January 30, 2013.
25. Census 2010, Summary File 2, Demographic Profile 1 by State. United States Census Bureau Web site. http://factfinder2.census.gov/faces/tableservices/jsf/pages/productview.xhtml?pid=DEC_10_SF2_SF2DP1&prodType=table. Accessed
February 4, 2013.
Enjoy this premium PDF as part of your membership benefits!


