Abstract
Objective: To evaluate the efficacy and safety of transcranial direct current stimulation (tDCS) in reducing autism spectrum disorder (ASD) symptoms in children and adolescents.
Data sources: A systematic search of multiple databases including PubMed, Ovid MEDLINE, Embase, Web of Science, Scopus, Cochrane CENTRAL, and Google Scholar was conducted through March 2025 using keywords and MeSH terms related to ASD and tDCS.
Study selection: Eligible studies included randomized controlled trials (RCTs), nonrandomized controlled trials (non-RCTs), and case reports that examined the effects of tDCS in individuals with ASD aged ≤18 years.
Data extraction: Two reviewers independently extracted data and assessed bias using Cochrane tools. Meta-analyses were performed with RevMan Web, and the certainty of evidence was rated with Grading of Recommendations Assessment, Development, and Evaluation profiler.
Results: Of the 266 studies screened, thirty studies (22 RCTs, 6 non-RCTs, and 2 case reports; N = 963) were analyzed. Significant improvements were observed in Social Responsiveness Scale scores (mean difference = −14.95; 95% CI, −26.07 to −3.83; P = .007; d = −0.42) after tDCS. Autism Treatment Evaluation Checklist scores showed moderate-to-large effect (d = –0.75), increasing to d = –0.95 after sensitivity analysis. Dorsolateral prefrontal cortex stimulation was most effective for behavioral improvements, while ventromedial prefrontal cortex stimulation improved emotional regulation. No major adverse effects were reported.
Conclusion: tDCS appears to be a safe and potentially effective adjunctive therapy for ASD. However, further large-scale, long-term RCTs are needed to confirm efficacy and optimize stimulation parameters.
Prim Care Companion CNS Disord 2026;28(2):25r04049
Author affiliations are listed at the end of this article.
Autism spectrum disorder (ASD) is usually diagnosed in early childhood, characterized by repetitive behaviors, restricted activities, and social communication impairments. As per the Autism and Developmental Disabilities Monitoring Network, for 2020, the overall ASD prevalence was 27.6 per 1,000 (1 in 36) children aged 8 years and was 3.8 times more prevalent among boys compared to girls (43.0 versus 11.4).1 Current treatments for ASD include behavioral and speech therapies, educational support, and psychopharmacologic interventions.2 The 2 US Food and Drug Administration–approved medications (risperidone and aripiprazole) target irritability but have limited effect on the core symptoms and may cause side effects. Behavioral interventions can improve adaptive skills but are resource intensive, require prolonged engagement, and often fail to generalize to daily life. Consequently, a therapeutic gap persists, as neither approach directly targets underlying neural dysfunctions such as atypical cortical connectivity and excitatory-inhibitory imbalance. Noninvasive brain stimulation could be a promising treatment modality to address the therapeutic gap between the pharmacologic, behavioral, and combination interventions. Transcranial direct current stimulation (tDCS) is a lightweight, inexpensive, noninvasive, and safe brain stimulation method that delivers electrical current to the brain using electrodes on the scalp. It is estimated that electric currents of 1 mA, for 7 to 13 minutes, can produce anodal stimulation (excitatory) or cathodal stimulation (inhibitory effects) on the motor cortex.3,4 tDCS modulates synaptic transmission by increasing or decreasing the neurons’ action potential frequency.5 In addition, the currents alter the transmembrane neuronal potential and change the excitability level, thus modulating the neurons’ firing rate in response to additional inputs.6 Studies suggest that the effects of tDCS at the molecular level are associated with several different mechanisms, local changes in ionic hydrogen, calcium concentrations, levels of cyclic adenosine monophosphate, modifications in protein synthesis, and modulation of N-methyl-D-aspartate receptor efficacy.7–9 Magnetic resonance spectroscopy has shown that anodal stimulation reduces local concentrations of the inhibitory neurotransmitter γ-aminobutyric acid (GABA), whereas cathodal stimulation reduces excitatory glutamate levels.10,11
Several systematic reviews have previously explored the effects of tDCS on brain connectivity in adults with various psychiatric disorders, including depression, anxiety, ASD, and obsessive-compulsive disorder. However, research on the application of tDCS in children with ASD remains relatively limited. To address this gap, we conducted a systematic review specifically focused on evaluating the impact of tDCS on neural connectivity, cognitive functions, social functioning, and behavioral outcomes in children with ASD. This review aims to synthesize existing findings, highlight potential therapeutic benefits, and identify key areas for future research in this emerging field.
METHODS
This systematic review and meta-analysis adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines.12 The protocol was registered with the International Prospective Register of Systematic Reviews (registration no. CRD420251033515).
Eligibility Criteria
Included studies enrolled participants aged ≤18 years with a primary diagnosis of ASD, confirmed by standardized diagnostic criteria. There were no restrictions based on sex, ethnicity, or comorbid conditions. Eligible studies were conducted in hospital or outpatient settings. Studies involving participants >18 years of age or those with conditions other than ASD were excluded.
Intervention
The intervention involved tDCS of varying intensities and brain targets, delivered alone or alongside stable pharmacologic regimens or behavioral therapies (eg, occupational, speech, or animal-assisted therapy).
Comparisons
Comparisons included sham tDCS, as well as combinations such as tDCS plus cognitive training versus cognitive training with or without sham stimulation and tDCS compared to repetitive transcranial magnetic stimulation (rTMS).
Outcomes
The primary outcomes included changes in core and associated ASD symptoms measured using validated tools: Autism Treatment Evaluation Checklist (ATEC),13 Autism Behavioral Checklist (ABC),14 Childhood Autism Rating Scale (CARS),15 Social Responsiveness Scale (SRS),16 Emotion Regulation Checklist (ERC),17 Emotion Recognition Task (ERT),18 and Theory of Mind (ToM).19 These tools measured domains like behavior, language/ speech communication, cognition, emotional regulation, and social functioning. Secondary outcomes included safety, adverse events, and biomarker changes related to tDCS. Improvement was indicated by reductions in ATEC, ABC, CARS, and SRS scores and increases in ERC, ERT, inhibitory control, and ToM scores.
Study Design
Randomized controlled trials (RCTs), nonrandomized clinical trials, and case reports were included. Excluded were systematic reviews, editorials, unpublished studies, and trials where tDCS was not used specifically for ASD.
Search Strategy and Selection Process
A comprehensive literature search was conducted on December 10, 2024, and updated in March 2025, using the following databases: PubMed, Ovid MEDLINE, Embase, Web of Science, Scopus, Cochrane CENTRAL, and Google Scholar. Keywords and MeSH terms included combinations of “Autism” OR “Autism Spectrum Disorder” AND “Transcranial Direct Current Stimulation” OR “tDCS.” Additional records were identified through manual screening of reference lists. All retrieved citations were imported into Rayyan.ai for deduplication and blinded screening. Two reviewers (M.S. and M.A.) independently screened titles, abstracts, and full texts. Discrepancies were resolved through discussion or consultation with a senior reviewer.
Data Extraction and Synthesis
An Excel-based extraction sheet was used by both reviewers independently to collect data on study design, participant demographics, sample size, tDCS parameters, cointerventions, outcome measures, and results. Disagreements were resolved by consensus.
Statistical Analysis
For continuous outcomes, mean differences (MDs) were calculated when the same scale was used, and standardized MDs (SMDs) when different scales measured the same outcome. Cohen d was computed for effect sizes, and all analyses used 95% CIs. For studies with multiple experimental arms, pooled means and SDs were calculated to generate a single composite experimental group for analysis. Analyses were conducted using RevMan Web, applying the inverse variance method under a random-effects model.20
Assessment of Risk of Bias
Risk of bias (RoB) for included RCTs was assessed using the revised Cochrane RoB 2 tool,21 evaluating the following domains:
- Randomization process
- Deviations from intended interventions
- Missing outcome data
- Measurement of outcomes
- Selection of the reported result.
Each domain was rated as low risk, some concerns, or high RoB, leading to an overall RoB judgment per study.
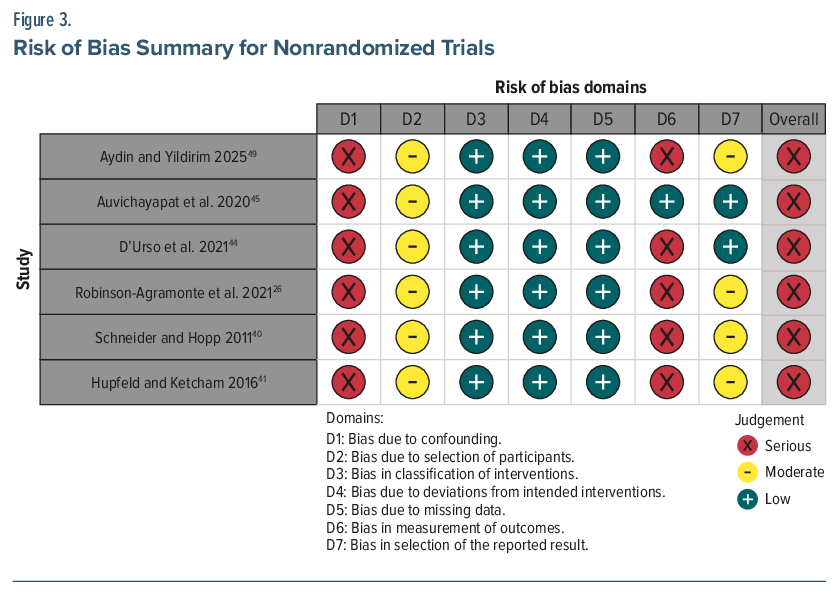
For nonrandomized, open-label studies, the Risk of Bias In Nonrandomized Studies of Interventions (ROBINS-I) tool was applied.22 ROBINS-I evaluates 7 domains of potential bias:
- Bias due to confounding
- Bias in selection of participants
- Bias in classification of interventions
- Bias due to deviations from intended interventions
- Bias due to missing data
- Bias in measurement of outcomes
- Bias in selection of the reported result.
Each domain was judged as having low, moderate, serious, or critical RoB or no information. Overall RoB was derived from the highest domain-level judgment, in accordance with ROBINS-I guidelines.
Measure of Treatment Effect
To evaluate treatment effects, we used the MD for outcomes measured on the same scale and SMDs with 95% CIs for continuous outcomes assessed using different scales. Postintervention means and SDs were extracted from each group when available. Cohen d was computed to quantify the overall effect size of the intervention on the main outcomes.
Assessment of Heterogeneity
A random-effects meta-analysis was performed to synthesize the data by pooling the postintervention scores and the proportion of patients with symptomatic improvement in autistic symptoms. Heterogeneity among included studies was assessed using the Cochrane Q-test and I2 statistic.
Missing Data
When SD data were not reported in the studies, we calculated SDs using CIs and SEs. Some studies did not report raw scores (ie, means) in their publications, and thus their data could not be included in the final analysis.
Subgroup Analysis
Subgroup analysis was performed to investigate the effect of potential variables leading to sources of heterogeneity. These variables were the study design, target used for tDCS, number of treatment sessions, and effects of simultaneous cognitive training used in certain studies.
Assessment of the Certainty of the Evidence
The Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach was used to rate the certainty of evidence in this systematic review.23 The GRADE evaluates the RoB, inconsistency, indirectness, imprecision, and publication bias to rate the quality of the evidence to the meta-analyses for the outcome as high, moderate, low, or very low. The rating for each domain was evaluated as not serious, serious, or very serious depending on the quality of the articles. A summary of findings table was created using GRADEpro and is available in Supplementary Table 1.
RESULTS
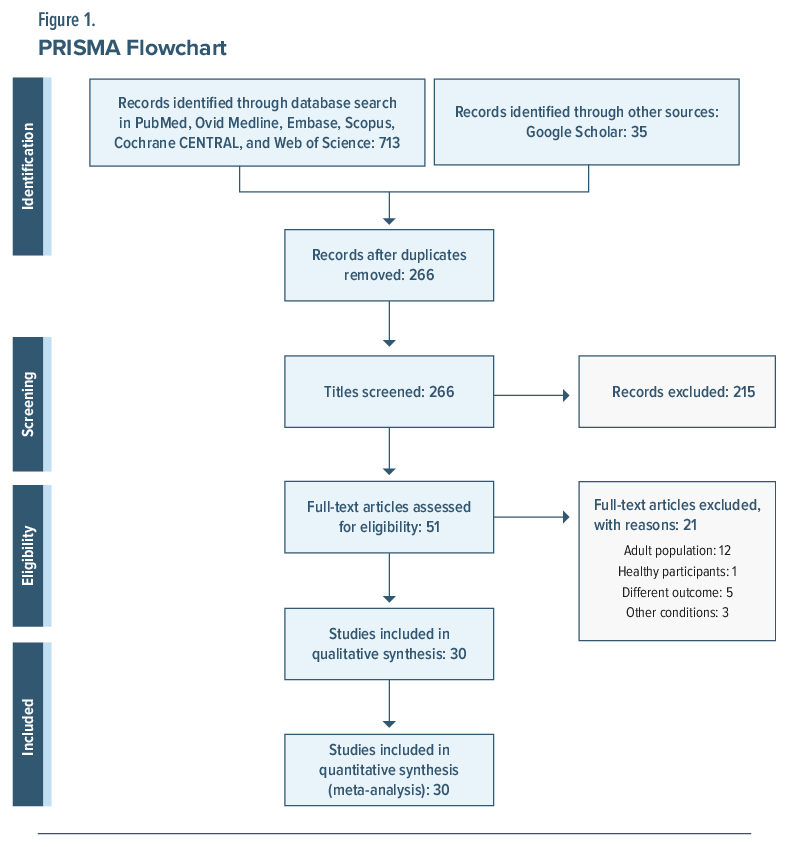
The literature search retrieved 777 articles. After removal of duplicates and screening, 30 studies24–53 met the inclusion criteria for this systematic review: 22 RCTs, 6 open-label nonrandomized studies, and 2 case reports—all in English. Except for the Toscano et al (2019)43 abstract, all were available in full text. Figure 1 illustrates the search and selection process (PRISMA flowchart).
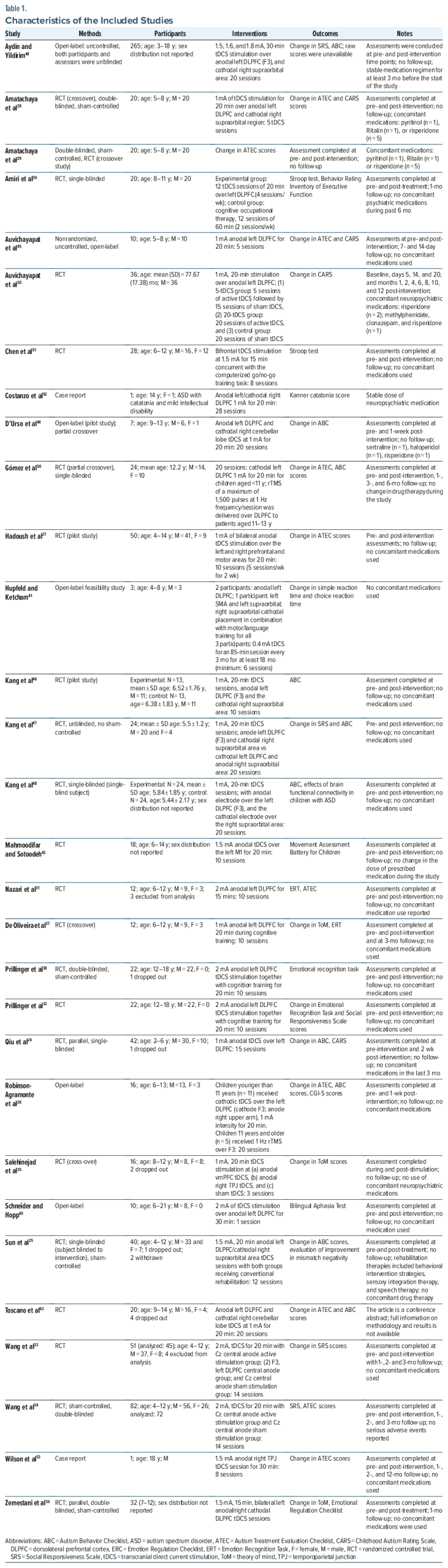
Table 1 shows the baseline characteristics of the 30 included studies.24–53 A total of 942 participants met the eligibility criteria and were included in the final analysis, with 716 in the treatment group and 226 in the control group. Among them, 521 were male; however, some studies did not report sex distribution. The studies were published between 2011 and March 2025 with a follow-up period ranging from 2 weeks to 12 months postintervention.
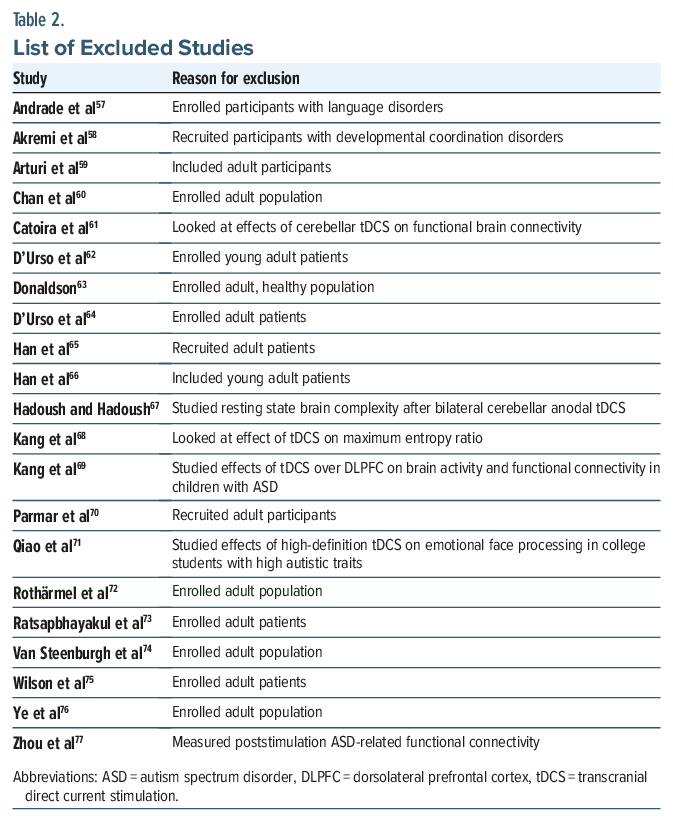
Several studies were excluded during the screening process due to use of interventions that were not of interest (ie, other neuromodulation techniques), recruitment of participants older than 18 years of age, or use of tDCS for treatment of other neuropsychiatric conditions (Table 2).
RoB in the Included Studies
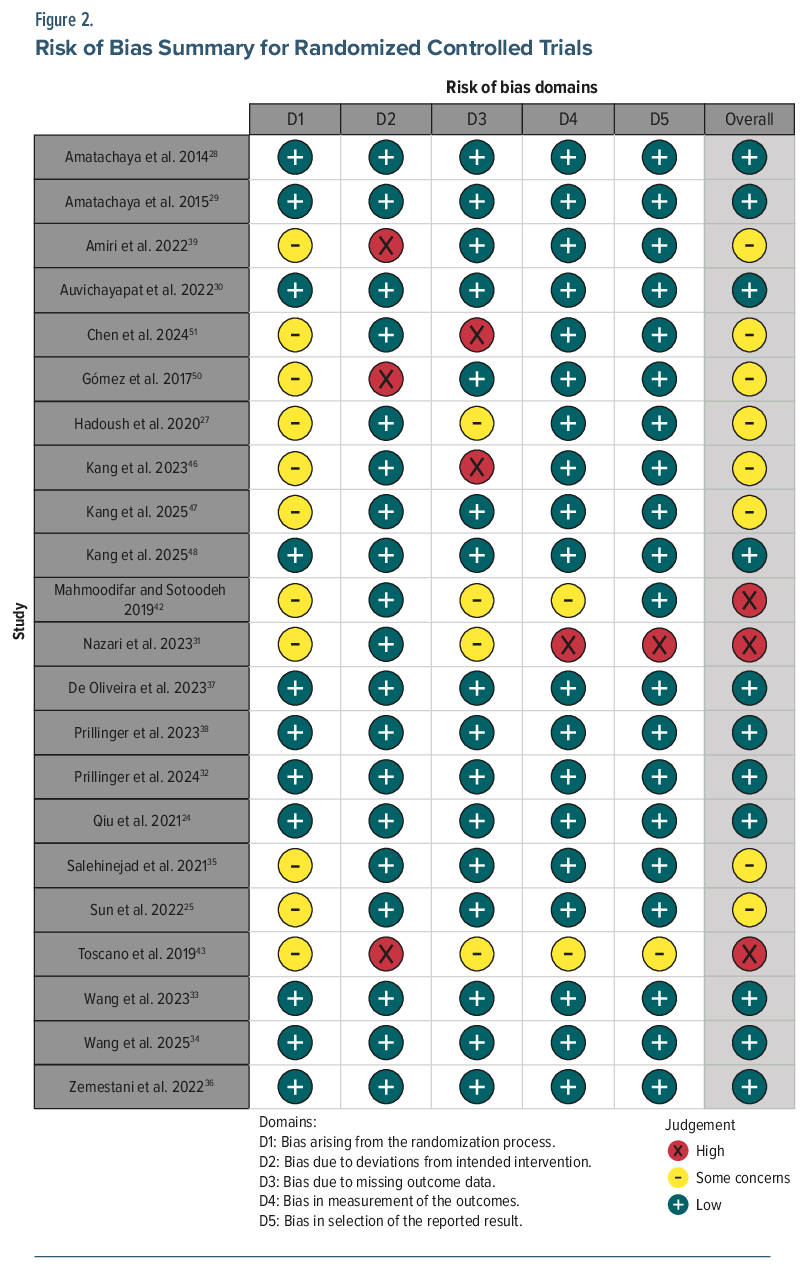
We assessed RoB in all studies and summarized the findings in Figures 2 and 3. Figure 2 shows the summary of RoB for RCTs, and Figure 3 shows a summary of the RoB for non-RCTs. A summary of the analyses for meta-analytic outcomes is provided in Supplementary Table 2.
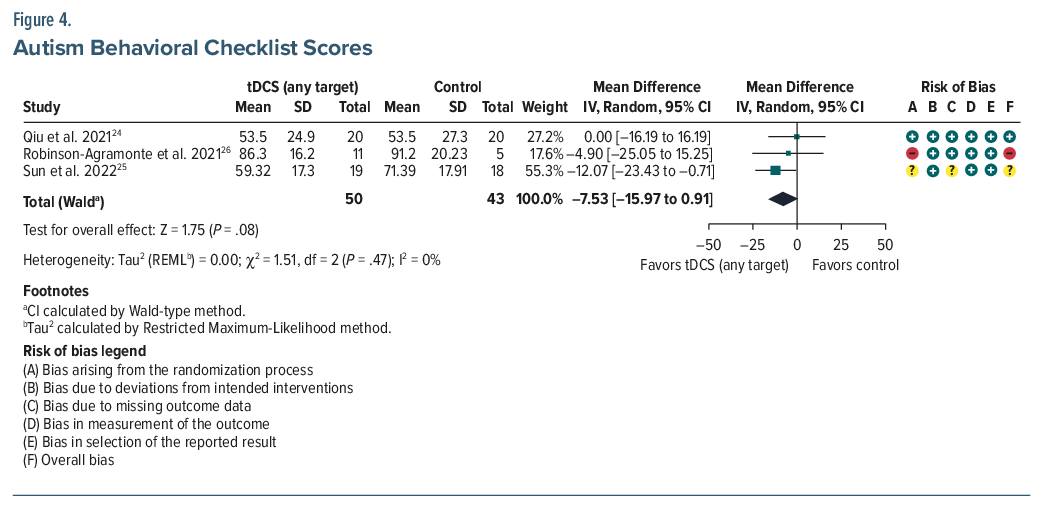
ABC. Three studies involving 93 participants yielded a pooled MD of −7.53 (95% CI, −15.97 to 0.91; P =.08), indicating no statistically significant changes between the active and the control groups.24–26 However, the pooled Cohen d of −0.43 indicated a small-to-moderate effect size favoring tDCS (Figure 4).
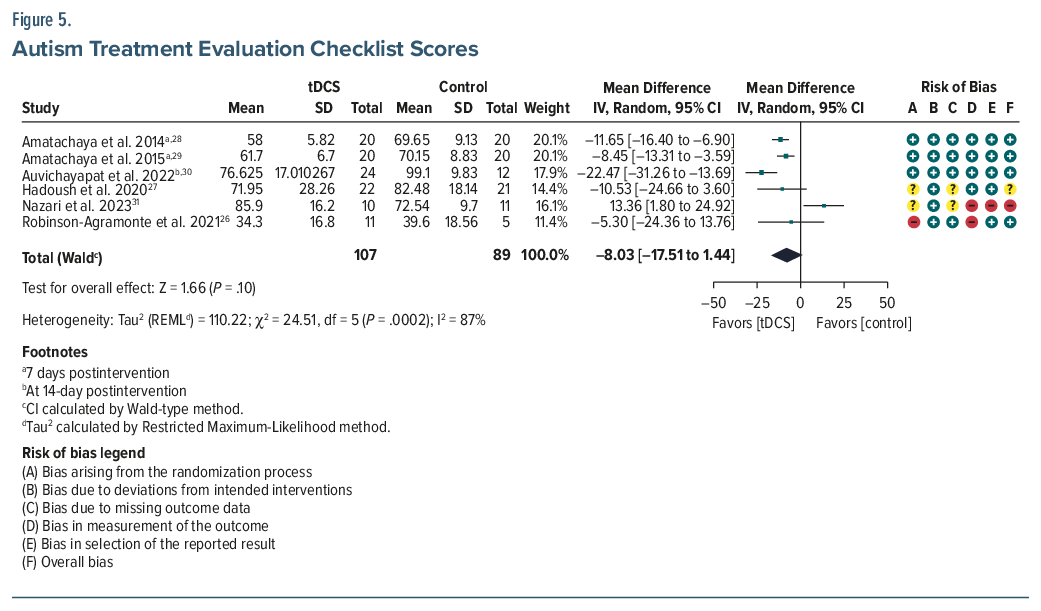
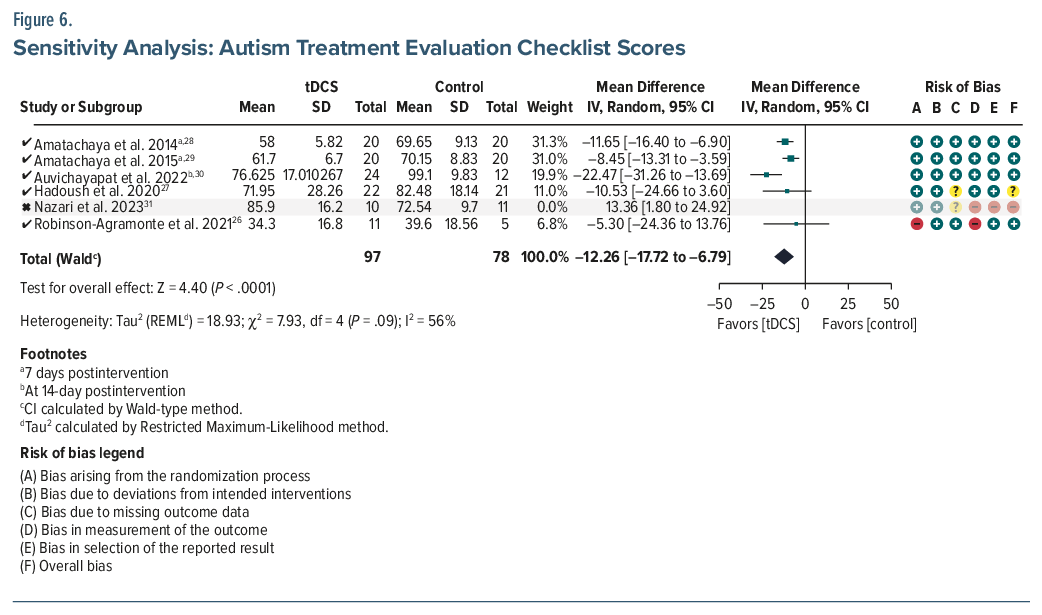
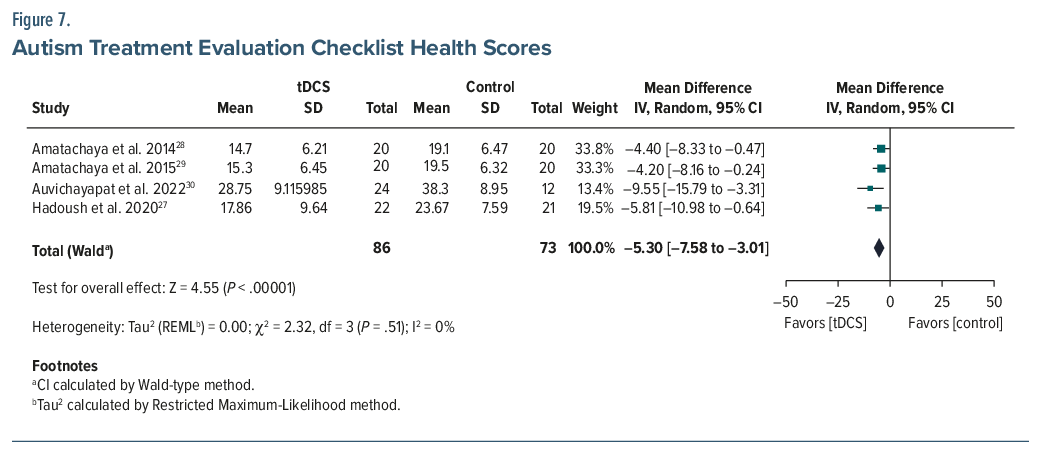
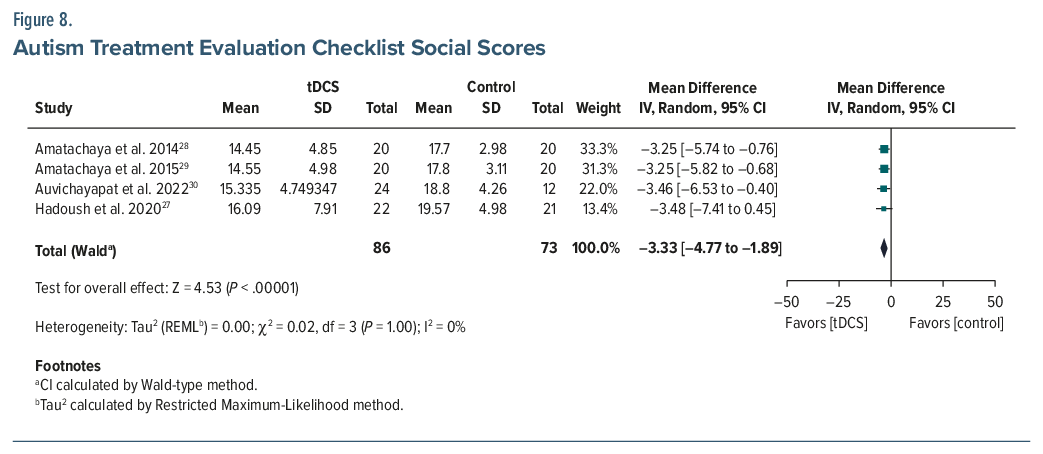
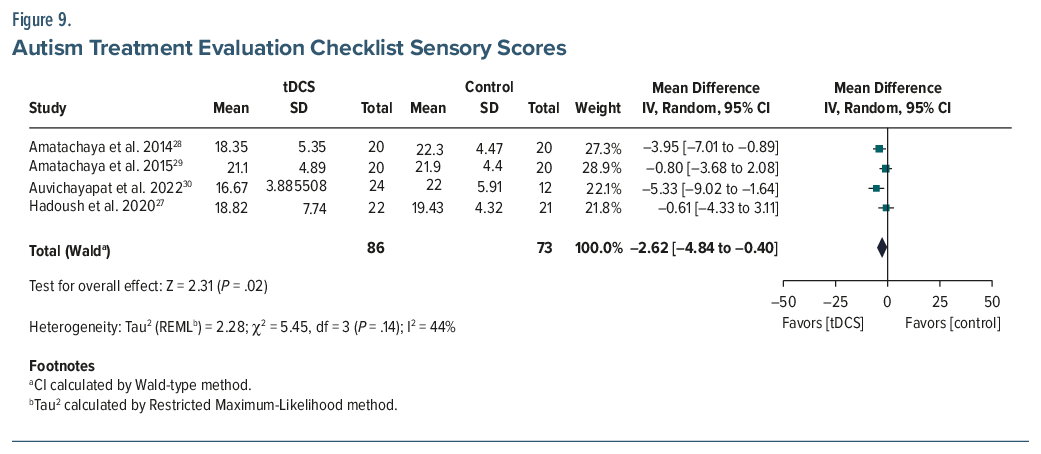
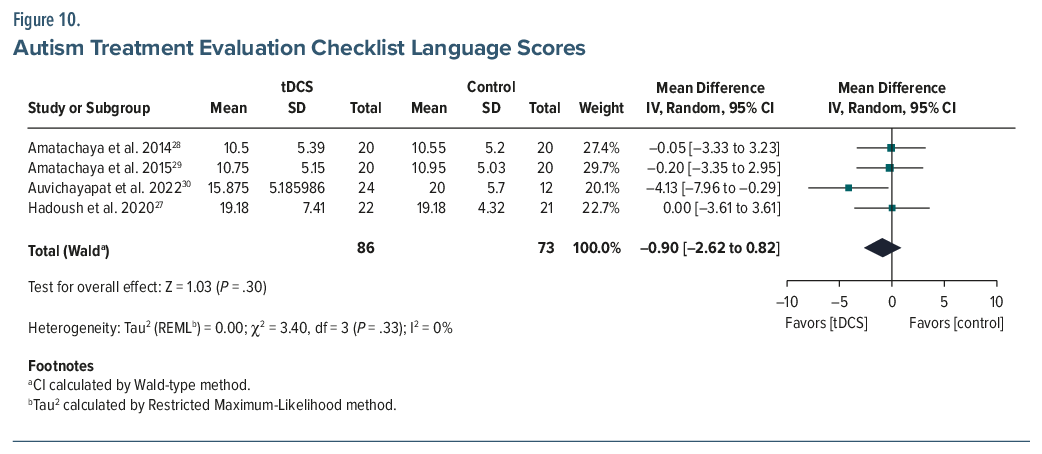
ATEC. Six studies (n=196) reported a pooled MD of –8.03 (95% CI, –17.51 to 1.44; P =.10), with high heterogeneity (I2=87%).26–31 While not statistically significant, the pooled Cohen d was –0.75, indicating a moderate-to-large effect favoring tDCS. A sensitivity analysis was conducted to assess the influence of the Nazari et al (2023) study,31 which misinterpreted an increase in ATEC scores postintervention in the experimental group from 76.36 (±14.1) to 85.9 (±16.2) as an improvement, which is incorrect. Excluding this study from the meta-analysis reduced heterogeneity (I2 from 87% to 56%) and increased the magnitude of the treatment effect (MD changed to–12.26; 95% CI, –17.72 to –6.79; P<.001; d =-0.95), suggesting that inclusion of this study may have underestimated the observed treatment benefit of tDCS. Subscale analyses from 4 studies (n=159) demonstrated statistically significant improvements (P <.05 for all) in the health (MD =–5.30; 95% CI, –7.58 to –3.01), social (MD =–3.33; 95% CI, –4.77 to –1.89), and sensory (MD =–2.62; 95% CI, –4.84 to –0.40) domains but no improvement (MD =–0.90; 95% CI, –2.62–0.82) in the language domain (Figures 5, 6, 7, 8, 9, and 10).
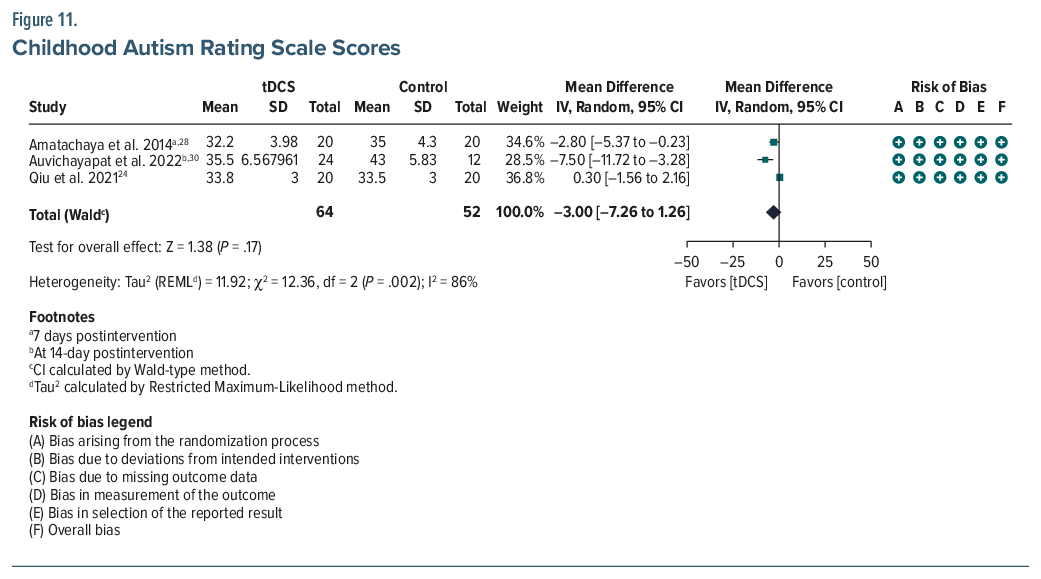
CARS. Three studies (n=116) yielded an MD of –3.00 (95% CI, –7.26 to 1.26; P =.17), indicating no statistically significant changes between the active and the control groups24,28,30 (Figure 11). However, the pooled Cohen d of –0.55 suggests a moderate effect. Individual trials reported significant within-group differences (P<.05 for all) after real tDCS treatment.
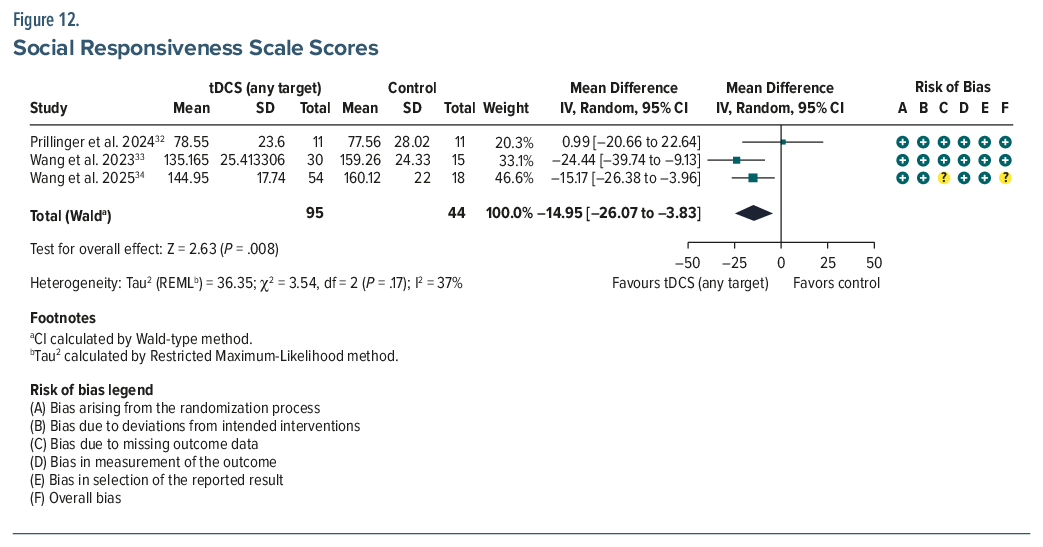
SRS. Three studies (n = 139) showed a significant improvement (MD = −14.95; 95% CI, −26.07 to−3.83; P = .008) in the active treatment group yielding a small-to-medium effect size (Cohen d = –0.42)32–34 (Figure 12). Subscale analysis (n = 117) revealed significant gains in social awareness (MD = –1.77; 95% CI, –2.77 to –0.78; P = .0005). In the Wang et al (2023) study,33 the pooled mean of the Cz (central) and the F3 (left dorsolateral prefrontal cortex [DLPFC]) anodal stimulation groups was calculated to represent the combined experimental group, as the study included 2 active intervention arms. Overall, no differential effects were found across stimulation sites. However, Wang et al33 demonstrated a 19.39% decrease in autistic behavior and a 14.39% decrease in social communication in the F3 stimulation group.
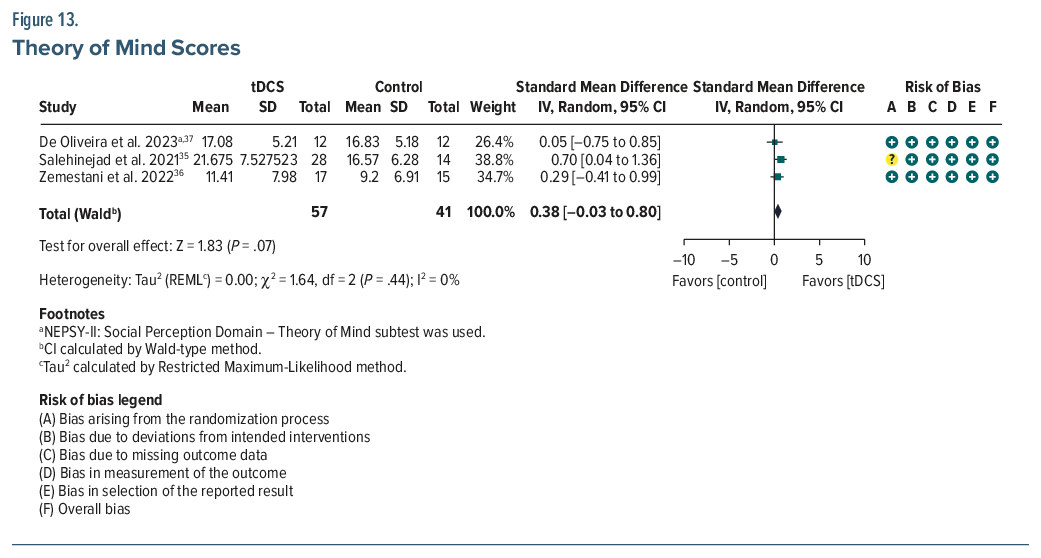
ToM. Three studies (n=98) showed a not significant pooled effect (SMD =0.38; 95% CI, –0.03 to 0.80; P =.07)35–37 (Figure 13). In the study by Salehinejad et al,35 the pooled mean of the ventromedial prefrontal cortex (vmPFC) and right temporoparietal junction tDCS groups was calculated to represent the combined experimental group, as the study included 2 active intervention arms. However, within-group improvements were observed in active groups.
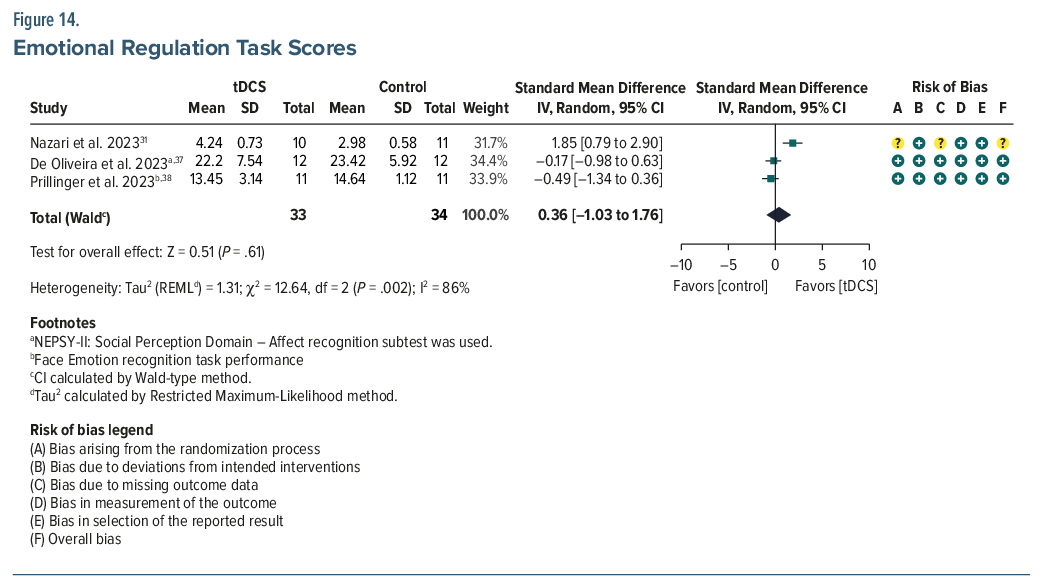
ERT. Three studies (n=74) found no significant between-group differences (SMD=0.36; 95% CI, –1.03 to 1.76; P =.61; I2=86%), though individual studies reported within-group improvements31,37,38 (Figure 14). Subgroup analysis revealed a significant interaction. In Nazari et al,31 tDCS alone showed a large effect (SMD=1.85; P=.0006), while studies combining tDCS with cognitive therapy showed no benefit (pooled SMD=–0.32; P =.28; I2=0%). The subgroup difference was significant (χ2=12.37; P=.0004), suggesting concurrent administration of cognitive therapy may influence, and potentially attenuate, the independent therapeutic effects of tDCS.
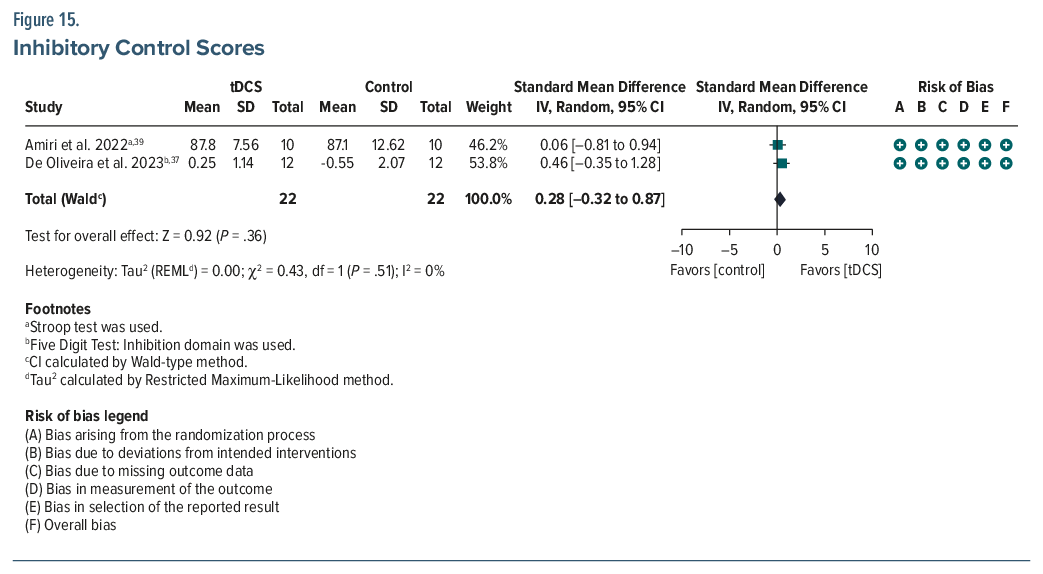
Inhibitory control. Two studies (n=44) found no significant between-group effects.37,39 Nonetheless, both reported within-group gains post-tDCS, measured via the Stroop test and five-digit test (Figure 15).
Descriptive Analysis of Studies Not Included in the Meta-Analysis
Schneider and Hopp (2011) reported substantial gains in syntax (247%) and vocabulary (16%) post-tDCS.40 Hupfeld et al41 studied the effects of tDCS over DLPFC or spinal muscular atrophy in 3 children during occupational and speech therapy. Improvements were noted in attention, task performance, and compliance, though specific behavioral outcomes were not comprehensively reported.
Mahmoodifar and Sotoodeh42 showed significantly greater improvements in balance (Movement Assessment Battery for Children-2) after anodal tDCS over the primary motor cortex (M1) combined with motor skill training on balance in 18 children with ASD. D’Urso et al44 reported significant reductions in ABC scores, particularly in hyperactivity and lethargy, following right cerebellar cathodal tDCS. Similarly, Toscano et al43 observed decreases in ABC and ATEC scores, along with reduced parental stress on stimulation over the same target. Auvichayapat et al45 also reported a significant reduction in ATEC and CARS scores after 5 consecutive tDCS sessions over the left DLPFC.
In a pilot RCT (n = 26), Kang et al46 reported significant improvements in ABC scores following active tDCS. These findings were replicated in their January 2025 study47 (n = 24), where anodal tDCS over the left DLPFC significantly improved SRS and ABC scores, with notable reductions in social communication (P = .028), social motivation (P = .012), and 4 ABC domains (P < .05); no such effects were seen in the cathodal group. A subsequent March 2025 study with a larger sample (n = 48) confirmed a significant decrease in ABC scores in the active tDCS group (pre: 55.52 ± 7.19; post: 37.67 ± 4.19; P < .05), with no significant change in the sham group.48
Aydin and Yildirim,49 in a large pediatric RCT (n = 256), found that anodal tDCS over the left DLPFC significantly improved verbal communication (Cohen d = 0.52), nonverbal cues (Cohen d = 0.48), emotional regulation (Cohen d = 0.57), and cognitive outcomes including working memory and flexibility (Cohen d = 0.45). Notably, younger children (aged 3–12 years) compared with older adolescents (aged 13–18 years) showed greater gains in nonverbal communication (M = 1.2 ± 0.5 vs. 0.7 ± 0.4, P < .05). Gómez et al50 reported significant reductions in ABC, ADI-R, and ATEC scores up to 6 months postintervention in both tDCS and rTMS groups, with no significant difference between the modalities.
In an RCT involving 28 participants with ASD, Chen et al51 demonstrated that active tDCS significantly improved response inhibition, interference control, and sustained attention, with increased neural activity in the DLPFC and frontopolar area. These cognitive gains also translated into improved classroom behavior. Costanzo et al52 reported full remission of catatonic symptoms such as excitement, stereotypy, and refusal to eat/drink in a 14-year-old girl following 28 sessions of bifrontal tDCS over the DLPFC. Mild improvements were noted in mutism and negativism, though posturing slightly worsened.
Wilson et al53 reported notable gains in social functioning in a patient following stimulation including initiating appropriate spontaneous conversations and context-appropriate conversations, as well as greater emotional awareness, and enhanced perspective taking. Notably, gains persisted with reduced anger 1 year later; the patient remained in a job training program, maintaining most social improvements, though challenges in peer relationships persisted.
Longitudinal Effects and Dosage
Gómez et al50 and Auvichayapat et al45 documented sustained symptom reduction at 6-and 12-months posttreatment, with longer protocols yielding more durable outcomes. However, subgroup analysis of ATEC scores revealed no significant differences based on the number of sessions.
Neurophysiological Correlates
tDCS-induced behavioral improvements correlated with electroencephalogram changes (eg, increased peak α frequency, reduced MMN latency) and biomarker shifts (eg, serum brain-derived neurotrophic factor reductions), suggesting neuroplastic adaptations (Amatachaya et al,29 Sun et al,25 Robinson-Agramonte et al26).
DISCUSSION
We reviewed 30 studies investigating the use of tDCS in children and adolescents with ASD. This review aimed to provide the most accurate estimate of tDCS efficacy based on the current literature. While evidence remains limited across all ASD domains, the findings suggest potential improvements in sociability, behavior, health, and physical functioning. The included studies varied considerably in design, with sample sizes ranging from 1 to 265 participants. Variability in electrode montages, current intensities, and stimulation durations contributed to the heterogeneity of outcomes. The concept of anodal-excitation/cathodal-inhibition theory suggests the direction of plasticity determined by parameters such as session duration, intervals between sessions, course of sessions, individual characteristics, and comedications during the therapy.54 Therefore, it is essential to compare the results in different samples of both anodal excitation and cathodal inhibition for efficacy outcome.
Many studies employed anodal stimulation over specific cortical targets, particularly the DLPFC, to modulate brain activity. The observed improvements may be partly explained by the GABAergic modulation induced by tDCS, as decreased GABA levels—especially in sensorimotor regions—have been implicated in ASD pathology. Several studies utilized unilateral anodal stimulation over the left or right DLPFC, guided by evidence of DLPFC hypoactivation in children with ASD.24,25,28–33,37–40,45–47 These studies commonly reported improvements in core symptoms, particularly in domains related to behavioral regulation, health, and social functioning. Prillinger et al (2023 and 2024) found enhanced dynamic emotion recognition and social brain network activation postintervention.32,38 Notably, Salehinejad et al35 demonstrated that targeting the vmPFC with anodal tDCS enhanced ToM abilities, especially in emotion recognition and first-order mental state reasoning functions centrally involved in social cognition.
The cathodal tDCS reduced glutamate levels with a correlated reduction in GABA.55,56 Cathodal stimulation over the left DLPFC has been associated with reductions in hyperactive behaviors, possibly due to suppression of overactivity in this region. This is supported by findings from Gómez et al50 and Robinson-Agramonte et al,26 who reported significant, sustained improvements in autistic symptoms up to 6 months postintervention with both cathodal tDCS and rTMS, with no significant difference between modalities. In contrast, another study found no significant changes in SRS or ABC scores following cathodal tDCS, highlighting variability in outcomes.45 Hadoush et al,27 via utilization of bilateral anodal tDCS stimulation over left and right mirror neurons, showed the efficacy of tDCS in symptom reduction in ASD symptoms. Chen et al51 noted reduced (response inhibition) and transfer effects (interference control, sustained attention) after receiving bifrontal tDCS.
Age-based subgroup analysis revealed no statistically significant differences in treatment outcomes between children aged ≤12 years and mixed-age samples, suggesting that age alone may not moderate the therapeutic effect of tDCS on ATEC scores. However, substantial heterogeneity was present, indicating the need for age-stratified protocols in future research. Overall, tDCS was well tolerated, with the most frequently reported side effects being transient redness, tingling, and itching at the electrode sites, primarily at the onset of stimulation.
Limitations and Future Directions
This meta-analysis indicates that tDCS may provide small-to-moderate improvements in core ASD symptoms. Pooled results showed significant reductions in ABC and SRS scores and moderate-to-large effects on ATEC scores following sensitivity analysis. However, the findings are limited by the small number of high-quality RCTs, modest sample sizes, and methodological heterogeneity across studies. Variations in tDCS protocols, outcome measures, and follow-up durations complicate comparisons. The use of a uniform 1 mA current across participants of varying ages may have introduced bias, given age-related differences in skull thickness, brain volume, and cerebrospinal fluid composition, which influence current distribution. Additionally, concerns about incomplete blinding and potential publication bias should be acknowledged. While preliminary evidence supports tDCS as a promising adjunctive intervention for ASD, future studies should prioritize larger, rigorously controlled trials to refine stimulation parameters and establish long-term efficacy.
Article Information
Published Online: March 12, 2026. https://doi.org/10.4088/PCC.25r04049
© 2026 Physicians Postgraduate Press, Inc.
Submitted: July 24, 2025; accepted October 31, 2025.
To Cite: Singh M, Adnan M, Husain K, et al. Role of transcranial direct current stimulation in the management of autism spectrum disorder in children and adolescents: a systematic review and meta-analysis. Prim Care Companion CNS Disord 2026;28(2):25r04049.
Author Affiliations: McMaster University, Hamilton, Ontario, Canada (Singh); Department of Psychiatry, Texas Tech University Health Science Center at Odessa/ Permian Basin Odessa, Midland, Texas (Adnan, Husain, Jain); Department of Psychiatry, UT Health, Houston, Texas (Trivedi).
Corresponding Author: Monika Singh, MD, McMaster University, Hamilton, Ontario, Canada ([email protected]).
Financial Disclosure: None.
Funding/Support: None.
Clinical Points
- Transcranial direct current stimulation (tDCS) improves core autism spectrum disorder symptoms, particularly social responsiveness and behavioral regulation, with stimulation of the dorsolateral prefrontal cortex showing the greatest benefit.
- tDCS is safe and well tolerated in children and adolescents, with no serious adverse effects reported across studies.
- tDCS may bridge the therapeutic gap between pharmacologic and behavioral approaches by enhancing cortical plasticity and social-cognitive function.
References (77)

- Maenner MJ, Warren Z, Williams AR, et al. Prevalence and characteristics of autism spectrum disorder among children aged 8 years — autism and Developmental Disabilities Monitoring Network, 11 sites, United States, 2020. MMWR Surveill Summ. 2023;72(2):1–14. PubMed CrossRef
- Qin L, Wang H, Ning W, et al. New advances in the diagnosis and treatment of autism spectrum disorders. Eur J Med Res. 2024;29(1):322. PubMed CrossRef
- Nitsche MA, Paulus W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J Physiology. 2000;527(3):633–639. CrossRef
- Nitsche MA, Fricke K, Henschke U, et al. Pharmacological modulation of cortical excitability shifts induced by transcranial direct current stimulation in humans. J Physiology. 2003;553(1):293–301. PubMed CrossRef
- Woods AJ, Antal A, Bikson M, et al. A technical guide to tDCS, and related non-invasive brain stimulation tools. Clin Neurophysiol. 2015;127(2):1031–1048. PubMed
- Wagner T, Fregni F, Fecteau S, et al. Transcranial direct current stimulation: a computer-based human model study. NeuroImage. 2007;35(3):1113–1124. PubMed CrossRef
- Merzagora AC, Foffani G, Panyavin I, et al. Prefrontal hemodynamic changes produced by anodal direct current stimulation. NeuroImage. 2009;49(3):2304–2310. PubMed
- Islam N, Aftabuddin M, Moriwaki A, et al. Increase in the calcium level following anodal polarization in the rat brain. Brain Res. 1995;684(2):206–208. PubMed CrossRef
- Nitsche MA, Jaussi W, Liebetanz D, et al. Consolidation of human motor cortical neuroplasticity by D-Cycloserine. Neuropsychopharmacology. 2004;29(8):1573–1578. PubMed CrossRef
- Stagg CJ, Best JG, Stephenson MC, et al. Polarity-Sensitive modulation of cortical neurotransmitters by transcranial stimulation. J Neurosci. 2009;29(16):5202–5206. PubMed CrossRef
- Clark VP, Coffman BA, Trumbo MC, et al. Transcranial direct current stimulation (tDCS) produces localized and specific alterations in neurochemistry: a 1H magnetic resonance spectroscopy study. Neurosci Lett. 2011;500(1):67–71. PubMed CrossRef
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;29:n71. CrossRef
- Rimland B, Edelson S. Autism research institute. Autism Treat Eval Checkl (ATEC). 1999.
- Krug DA, Arick J, Almond P. Behavior checklist for identifying severely handicapped individuals with high levels of autistic behavior. J Child Psychol Psychiatry. 1980;21(3):221–229. PubMed CrossRef
- Schopler E, Reichler RJ, DeVellis RF, et al. Toward objective classification of childhood autism: childhood Autism Rating Scale (CARS). J Autism Develop Disord. 1980;10(1):91–103. PubMed CrossRef
- Constantino JN, Davis SA, Todd RD, et al. Validation of a brief quantitative measure of autistic traits: comparison of the social responsiveness scale with the autism diagnostic interview-revised. J Autism Dev Disord. 2003;33(4):427–433. PubMed CrossRef
- Shields A, Cicchetti D. Emotion regulation among school-age children: the development and validation of a new criterion Q-sort scale. Develop Psychol. 1997;33(6):906–916. PubMed CrossRef
- Montagne B, Kessels RP, De Haan EH, et al. The Emotion Recognition Task: a paradigm to measure the perception of facial emotional expressions at different intensities. Percept Mot Skills. 2007;104(2):589–598. PubMed CrossRef
- Long EL, Catmur C, Bird G. The theory of mind hypothesis of autism: a critical evaluation of the status quo. Psychol Rev. 2025. doi:10.1037/rev0000532. CrossRef
- Review Manager (RevMan) [Computer program]. The Cochrane Collaboration; 2025. Available at revman.cochrane.org.
- Sterne JAC, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. PubMed CrossRef
- Sterne JA, Hernán MA, Reeves BC, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919. PubMed CrossRef
- GRADEpro GDT. GRADEpro guideline development tool [Software]. In: McMaster University and Evidence Prime; 2023. Available from gradepro.org.
- Qiu J, Kong X, Li J, et al. Transcranial direct current stimulation (tDCS) over the left dorsal lateral prefrontal cortex in children with autism spectrum disorder (ASD). Neural Plast. 2021;2021:1–11. CrossRef
- Sun C, Zhao Z, Cheng L, et al. Effect of transcranial direct current stimulation on the mismatch negativity features of deviated stimuli in children with autism spectrum disorder. Front Neurosci. 2022;16:721987. PubMed CrossRef
- Robinson-Agramonte M, Michalski B, Fernández LG, et al. Effect of non-invasive brain stimulation on behavior and serum brain-derived neurotrophic factor and insulin-like growth factor-1 levels in autistic patients. Drug Develop Res. 2021;82(5):716–723. PubMed CrossRef
- Hadoush H, Nazzal M, Almasri NA, et al. Therapeutic effects of bilateral anodal transcranial direct current stimulation on prefrontal and motor cortical areas in children with autism spectrum disorders: a pilot study. Autism Res. 2020;13(5):828–836. PubMed CrossRef
- Amatachaya A, Auvichayapat N, Patjanasoontorn N, et al. Effect of anodal transcranial direct current stimulation on autism: a Randomized Double-Blind Crossover trial. Behav Neurol. 2014;2014:1–7. CrossRef
- Amatachaya A, Jensen MP, Patjanasoontorn N, et al. The short-term effects of transcranial direct current stimulation on electroencephalography in children with autism: a randomized crossover controlled trial. Behav Neurol. 2015;2015:1–11. CrossRef
- Auvichayapat P, Intayot K, Udomchat C, et al. Long-term effects of transcranial direct current stimulation in the treatment of autism spectrum disorder: a randomized controlled trial. Develop Med Child Neurology. 2022;65(6):811–820. CrossRef
- Nazari G, Mousavi M, Sadeghi-Firoozabadi V. Effectiveness of anodal transcranial direct current stimulation of left dorsolateral prefrontal cortex in facial emotion recognition and clinical symptoms of boys with autism spectrum disorder. Iran J Psychiatry Behav Sci. 2023;18(2).
- Prillinger K, De Lara GA, Klöbl M, et al. Multisession tDCS combined with intrastimulation training improves emotion recognition in adolescents with autism spectrum disorder. Neurotherapeutics. 2024;21(6):e00460. PubMed
- Wang Y, Wang F, Kong Y, et al. High definition transcranial direct current stimulation of the Cz improves social dysfunction in children with autism spectrum disorder: a randomized, sham, controlled study. Autism Res. 2023;16(10):2035–2048. PubMed CrossRef
- Wang Y, Li Z, Ye Y, et al. HD-tDCS effects on social impairment in autism spectrum disorder with sensory processing abnormalities: a randomized controlled trial. Sci Rep. 2025;15(1):9772. PubMed CrossRef
- Salehinejad MA, Paknia N, Hosseinpour AH, et al. Contribution of the right temporoparietal junction and ventromedial prefrontal cortex to theory of mind in autism: a randomized, sham-controlled tDCS study. Autism Res. 2021;14(8):1572–1584. PubMed CrossRef
- Zemestani M, Hoseinpanahi O, Salehinejad MA, et al. The impact of prefrontal transcranial direct current stimulation (tDCS) on theory of mind, emotion regulation and emotional-behavioral functions in children with autism disorder: a randomized, sham-controlled, and parallel-group study. Autism Res. 2022;15(10):1985–2003. PubMed CrossRef
- De Oliveira PV, De Castro ML, Collange-Grecco LA, et al. Transcranial direct current stimulation and cognitive stimulation therapy in children with autism spectrum disorder: randomized, sham-controlled, double-blind, crossover, clinical trial. Brain Imaging Stimul. 2023;2:e4954. CrossRef
- Prillinger K, Radev ST, De Lara GA, et al. Effects of transcranial direct current stimulation on social attention patterns and emotion recognition ability in male adolescents with autism spectrum disorder. J Clin Med. 2023;12(17):5570. PubMed
- Amiri MA, Esmaili SK, Saei S, et al. Effectiveness of transcranial direct current stimulation in behavioral and cognitive aspects of executive function in children with autism spectrum disorder. Iran J Psychiatry Behav Sci. 2022;16(4).
- Schneider HD, Hopp JP. The use of the Bilingual Aphasia Test for assessment and transcranial direct current stimulation to modulate language acquisition in minimally verbal children with autism. Clin Linguistics Phonetics. 2011;25(6-7):640–654. PubMed CrossRef
- Hupfeld K, Ketcham C. Behavioral effects of transcranial direct current stimulation on motor and language planning in minimally verbal children with autism spectrum disorder (ASD): feasibility, limitations and future directions. J Child Develop Disord. 2016;2(3).
- Mahmoodifar E, Sotoodeh MS. Combined transcranial direct current stimulation and selective motor training enhances balance in children with autism spectrum disorder. Percept Mot Ski. 2019;127(1):113–125. CrossRef
- Toscano E, Sanges V, Riccio MP, et al. Fronto-cerebellar tDCS in children with autism spectrum disorder. L Encéphale. 2019;45:S79–S80. CrossRef
- D’Urso G, Toscano E, Sanges V, et al. Cerebellar transcranial direct current stimulation in children with autism spectrum disorder: a pilot study on efficacy, feasibility, safety, and unexpected outcomes in tic disorder and epilepsy. J Clin Med. 2021;11(1):143. PubMed
- Auvichayapat N, Patjanasoontorn N, Phuttharak W, et al. Brain metabolite changes after anodal transcranial direct current stimulation in autism spectrum disorder. Front Mol Neurosci. 2020;13:70. PubMed CrossRef
- Kang J, Fan X, Zhong Y, et al. Transcranial direct current stimulation modulates EEG microstates in Low-Functioning autism: a pilot study. Bioengineering. 2023;10(1):98. PubMed CrossRef
- Kang J, Wu J, Huang X, et al. Differential effects of left DLPFC anodal and cathodal tDCS interventions on the brain in children with autism: a randomized controlled trial. IBRO Neurosci Rep. 2025;18:171–179. PubMed CrossRef
- Kang J, Mao W, Wu J, et al. TDCS modulates brain functional networks in children with autism spectrum disorder: a resting-state EEG study. J Integr Neurosci. 2025;24(3):27314. PubMed CrossRef
- Aydin A, Yildirim A. Assessing the impact of Transcranial Direct Current Stimulation (tDCS) over the dorsolateral prefrontal cortex on social communication in children and adolescents with Autism Spectrum Disorder (ASD). Res Develop Disabil. 2025;161:104958. PubMed CrossRef
- Gómez L, Vidal B, Maragoto C, et al. Non-invasive brain stimulation for children with autism spectrum disorders: a short-term outcome study. Behav Sci. 2017;7(3):63. PubMed CrossRef
- Chen L, Du B, Li K, et al. The effect of tDCS on inhibitory control and its transfer effect on sustained attention in children with autism spectrum disorder: an fNIRS study. Brain Stimul. 2024;17(3):594–606. PubMed CrossRef
- Costanzo F, Menghini D, Casula L, et al. Transcranial direct current stimulation treatment in an adolescent with autism and drug-resistant catatonia. Brain Stimul. 2015;8(6):1233–1235. PubMed CrossRef
- Wilson JE, Quinn DK, Wilson JK, et al. Transcranial direct current stimulation to the right temporoparietal junction for social functioning in autism spectrum disorder. J Ect. 2017;34(1):e10–e13.
- Thair H, Holloway AL, Newport R, et al. Transcranial Direct Current Stimulation (TDCS): a beginner’s guide for design and implementation. Front Neurosci. 2017;11:641. PubMed CrossRef
- Stagg CJ, Bachtiar V, Amadi U, et al. Local GABA concentration is related to network-level resting functional connectivity. eLife. 2014;3:e01465. PubMed CrossRef
- Puts NAJ, Wodka EL, Harris AD, et al. Reduced GABA and altered somatosensory function in children with autism spectrum disorder. Autism Res. 2016;10(4):608–619. PubMed CrossRef
- Andrade AC, Magnavita GM, Allegro JVBN, et al. Feasibility of transcranial direct current stimulation use in children aged 5 to 12 years. J Child Neurology. 2013;29(10):1360–1365. CrossRef
- Akremi H, Hamel R, Dumas A, et al. Cerebellar transcranial direct current stimulation in children with developmental coordination disorder: a randomized, double-blind, sham-controlled pilot study. J Autism Develop Disord. 2021;52(7):3202–3213. CrossRef
- Arturi L, Scoppola C, Riccioni A, et al. Application of transcranial direct current stimulation combined with cognitive-behavioral therapy for communication and pragmatic skills’ improvement in autism spectrum disorder. Neurosci Appl. 2023;2:102574. CrossRef
- Chan MMY, Choi CXT, Tsoi TCW, et al. Effects of multisession cathodal transcranial direct current stimulation with cognitive training on sociocognitive functioning and brain dynamics in autism: a double-blind, sham-controlled, randomized EEG study. Brain Stimul. 2023;16(6):1604–1616. PubMed CrossRef
- Catoira B, Lombardo D, De SS, et al. Exploring the effects of cerebellar TDCS on brain connectivity using Resting-State FMRI. Brain Behav. 2025;15(2).
- D’Urso G, Bruzzese D, Ferrucci R, et al. Transcranial direct current stimulation for hyperactivity and noncompliance in autistic disorder. World J Biol Psychiatry. 2015;16(5):361–366. PubMed
- Donaldson PH, Kirkovski M, Rinehart NJ, et al. Autism-relevant traits interact with temporoparietal junction stimulation effects on social cognition: a high-definition transcranial direct current stimulation and electroencephalography study. Eur J Neurosci. 2017;47(6):669–681. PubMed CrossRef
- D’Urso G, Toscano E, Sanges V. Long-term home based Fronto-cerebellar transcranial direct current stimulation for Autism Spectrum Disorder: a case series. Psychiatr Danub. 2022;34(suppl. 3):10–57.
- Han YMY, Chan MMY, Shea CKS, et al. Neurophysiological and behavioral effects of multisession prefrontal tDCS and concurrent cognitive remediation training in patients with autism spectrum disorder (ASD): a double-blind, randomized controlled fNIRS study. Brain Stimul. 2022;15(2):414–425. PubMed CrossRef
- Han YM, Chan MM, Shea CK, et al. Effects of prefrontal transcranial direct current stimulation on social functioning in autism spectrum disorder: a randomized clinical trial. Autism. 2023;27(8):2465–2482. PubMed CrossRef
- Hadoush H, Hadoush A. Modulation of resting-state brain complexity after bilateral cerebellar anodal transcranial direct current stimulation in children with autism spectrum disorders: a randomized controlled trial study. The Cerebellum. 2022;22(6):1109–1117. PubMed CrossRef
- Kang J, Cai E, Han J, et al. Transcranial direct current stimulation (TDCS) can modulate EEG complexity of children with autism spectrum disorder. Front Neurosci. 2018;12:201. PubMed CrossRef
- Kang J, Li Y, Lv S, et al. Effects of transcranial direct current stimulation on brain activity and cortical functional connectivity in children with autism spectrum disorders. Front Psychiatry. 2024;15:1407267. PubMed CrossRef
- Parmar D, Enticott PG, Albein-Urios N. Anodal HD-tDCS for cognitive inflexibility in autism spectrum disorder: a pilot study. Brain Stimul. 2021;14(5):1298–1300. PubMed CrossRef
- Qiao Y, Hu Q, Xuan R, et al. High-definition transcranial direct current stimulation facilitates emotional face processing in individuals with high autistic traits: a sham-controlled study. Neurosci Lett. 2020;738:135396. PubMed CrossRef
- Rothärmel M, Moulier V, Vasse M, et al. A prospective open-label pilot study of transcranial direct current stimulation in high-functioning autistic patients with a dysexecutive syndrome. Neuropsychobiology. 2019;78(4):189–199. PubMed CrossRef
- Ratsapbhayakul T, Keeratitanont K, Chonprai C, et al. Anodal transcranial direct-current stimulation and non-verbal intelligence in autism spectrum disorder: a randomized controlled trial. Develop Med Child Neurology. 2024;66(9):1244–1254. PubMed CrossRef
- Van Steenburgh JJ, Varvaris M, Schretlen DJ, et al. Balanced bifrontal transcranial direct current stimulation enhances working memory in adults with high-functioning autism: a sham-controlled crossover study. Mol Autism. 2017;8(1):40. PubMed CrossRef
- Wilson J, Trumbo M, Tesche C. Transcranial direct current stimulation (tDCS) improves empathy and recognition of facial emotions conveying threat in adults with autism spectrum disorder (ASD): a randomized controlled pilot study. NeuroRegulation. 2021;8(2):87–95. CrossRef
- Ye H, Chen S, Huang D, et al. Modulation of neural activity in the temporoparietal junction with transcranial direct current stimulation changes the role of beliefs in moral judgment. Front Hum Neurosci. 2015;9:659. PubMed CrossRef
- Zhou T, Kang J, Li Z, et al. Transcranial direct current stimulation modulates brain functional connectivity in autism. NeuroImage Clin. 2020;28:102500. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!