Olanzapine, an atypical antipsychotic with a good safety profile among the antipsychotic medications, is commonly prescribed in major psychiatric illnesses.1 Although olanzapine is mostly associated with adverse effects related to metabolism, we present the case of a patient with urinary incontinence attributable to olanzapine.
Case Report
A 60-year-old retired, married man of middle socioeconomic status and with an average build was admitted to the psychiatry ward after transfer from the emergency department with a 2-year history of progressively increasing complaints of decreased sleep, odd talk, irritability, disorganized behavior, poor self-care, and assaultive behavior toward family members. There was no history suggestive of diabetes mellitus, hypertension, or complaints pertaining to the urinary tract or bladder. He was well oriented with a Hindi Mental State Examination score of 28/31.2 The neurologic examination at that time was grossly normal with no focal deficits. There was no evidence of infection, and his blood sugar levels were normal.
With a working diagnosis of other nonorganic psychotic disorder (ICD-10 code F28), the patient was started on tablet olanzapine 5 mg/d and uptitrated to 10 mg within 3 days. Within 24 hours of starting the medication, the patient was stressed with loss of bladder control and soiling of his clothes with urine throughout the day along with increased frequency of micturition and urgency. The urine routine microscopy and culture results were within normal limits. Ultrasound of the kidney, ureters, and bladder revealed bilateral pelvicalyceal system distention due to overdistention of the bladder and a single anechoic cystic lesion measuring 4 x 4.3 cm at the interpolar region of the left kidney. The patient’s prostatic-specific antigen was 0.72 ng/mL (normal for his age). Nonspecific changes were reported on brain magnetic resonance imaging. Urology consultation ruled out any urological cause for the incontinence.
To decrease the patient’s distress, urinary catheterization was done 5 days after starting tablet olanzapine. In the following week, the dose of olanzapine was decreased to 7.5 mg/d, given the possibility of drug-related phenomenon as etiology for his urinary incontinence. No other side effects of olanzapine were observed.
By the end of 2 weeks, olanzapine was withheld, and his urinary catheter was clamped and finally removed 24 hours later. The patient reported no urinary incontinence on removal of the catheter. After 2 days of observation, he was started on tablet aripiprazole 2.5 mg/d, which was uptitrated to 5 mg/d. After discontinuation of tablet olanzapine, the patient’s symptoms of urinary incontinence completely resolved. In this patient, urinary incontinence appeared after commencement of olanzapine, and symptoms resolved immediately after stopping the drug. Additionally, we did not prescribe any other drug that could precipitate similar symptoms in our patient. Using the Naranjo Adverse Drug Reaction Probability Scale,3 a score of 9 was obtained, which is suggestive of olanzapine being a definite cause of urinary incontinence in this patient.
Discussion
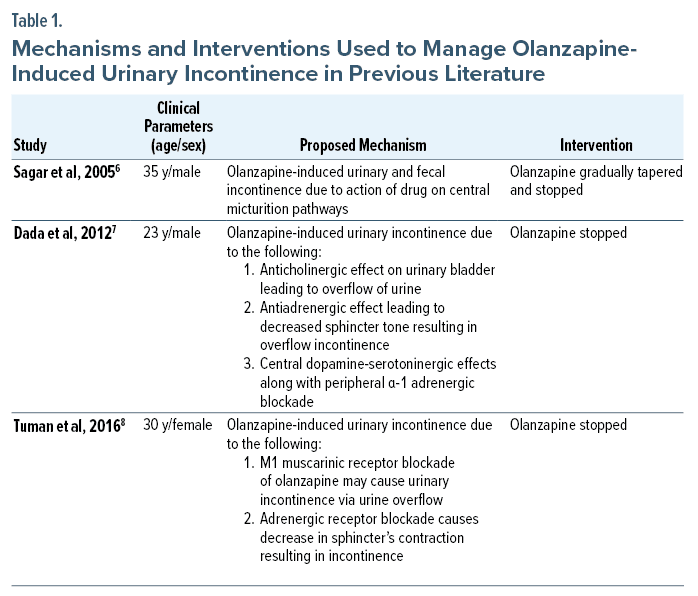
Only a handful of cases of olanzapine-induced bowel/bladder incontinence have been reported. As per a recent systematic review, the rate of occurrence of urinary incontinence is highest with clozapine (20.7%), followed by olanzapine (9.6%), quetiapine (6.7%), and risperidone (6.2%).4 As shown in Table 1, the mechanisms underlying olanzapine-induced urinary incontinence are hypothesized as antiadrenergic effect on the sphincter, blockage of the pudendal reflexes, effect on central micturition pathways, and anticholinergic effect on urinary bladder leading to overflow of urine. In our patient, the complaints of urgency and increased frequency with loss of bladder control point toward urge incontinence or overactive bladder and sphincter involvement. Hence, we propose the antiadrenergic(b3) and M1 muscarinic receptor blockade of olanzapine as the cause of incontinence and the prominent mechanism.
A management algorithm for antipsychotic-induced incontinence suggested by Arasteh et al9 involves discontinuing the antipsychotic drug if feasible or modifying the dose/dose timings. If these steps are not possible, changing the antipsychotic to a drug with less risk of enuresis is suggested. However, if it is not possible to modify or change the same drug, and the patient suffers from medical comorbidities, adding drugs like desmopressin, imipramine, amitriptyline, solifenacin, oxybutynin, trihexyphenidyl, bethanechol, verapamil, reboxetine, and pseudoephedrine is suggested as an alternative.9 Our case report aims to postulate urinary incontinence as a rare but distressing adverse effect of olanzapine, which needs to be recognized in clinical practice.
Article Information
Published Online: March 14, 2024.
https://doi.org/10.4088/PCC.23cr03631
© 2024 Physicians Postgraduate Press, Inc.
Submitted: August 24, 2023; accepted: November 15, 2023.
To Cite: Dewan G, Mehta S, Domun I. Urinary incontinence: a rare adverse effect of olanzapine. Prim Care Companion CNS Disord. 2024;26(2):23cr03631.
Author Affiliations: Department of Psychiatry, Government Medical College and Hospital, Chandigarh, India (all authors).
Corresponding Author: Shivangi Mehta, MD, Department of Psychiatry, Government Medical College and Hospital, Chandigarh, India ([email protected]).
Relevant Financial Relationships: None.
Funding/Support: None.
Patient Consent: The patient consented to publish the report, and information has been de-identified to protect anonymity.
References (9)

- Gómez JC, Sacristán JA, Hernández J, et al; Pharmacoepidemiologic Study of Olanzapine in Schizophrenia. The safety of olanzapine compared with other antipsychotic drugs: results of an observational prospective study in patients with schizophrenia (EFESO Study). J Clin Psychiatry. 2000;61(5):335–343. PubMed CrossRef
- Ganguli M, Ratcliff G, Chandra V, et al. A Hindi version of the MMSE: the development of a cognitive screening instrument for a largely illiterate rural elderly population in India. Int J Geriatr Psychiatry. 1995;10(5):367–377. CrossRef
- Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30(2):239–245. PubMed CrossRef
- Harrison-Woolrych M, Skegg K, Ashton J, et al. Nocturnal enuresis in patients taking clozapine, risperidone, olanzapine and quetiapine: comparative cohort study. Br J Psychiatry. 2011;199(2):140–144. PubMed CrossRef
- Vernon LT, Fuller MA, Hattab H, et al. Olanzapine-induced urinary incontinence: treatment with ephedrine. J Clin Psychiatry. 2000;61(8):601–602. PubMed CrossRef
- Sagar R, Varghese ST, Balhara YP. Olanzapine-induced double incontinence. Indian J Med Sci. 2005;59(4):163–164. PubMed CrossRef
- Dada MU, Oluwole LO, Adegun PT, et al. Olanzapine as a cause of urinary incontinence: a case report. Iran J Psychiatry. 2012;7(3):146–148. PubMed
- Tuman TC, Cakir U, Yildirim O. Olanzapine-induced urinary incontinence. Anadolu Psikiyatri Derg. 2016;17:15.
- Arasteh A, Mostafavi S, Zununi Vahed S, et al. An association between incontinence and antipsychotic drugs: a systematic review. Biomed Pharmacother. 2021;142:112027. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!