Using Stimulants for Attention-Deficit/Hyperactivity Disorder: Clinical Approaches and Challenges
Jonathan R. Stevens, MD, MPH; Timothy E. Wilens, MD; and Theodore A. Stern, MD
Have you prescribed a stimulant and been concerned about the risk of substance misuse or diversion, the emergence of tics, or even sudden death? Have you been reluctant to prescribe stimulants for patients with attention-deficit/hyperactivity disorder (ADHD) and comorbid autism, bipolar disorder, or seizures for fear of exacerbating the underlying disorder? Have you felt confused or bewildered as the number of approved stimulant preparations has tripled over the past 15 years? In this article, we explore prescribing practices regarding stimulants for patients with ADHD and examine clinical concerns and challenges to safe and effective prescribing.
WHAT IS ADHD AND HOW IS IT DIAGNOSED?
ADHD is a cognitive and behavioral syndrome characterized by varying levels and expressions of deficits in attention and problem-solving, along with hyperactivity and impulsiveness. ADHD is a common childhood disorder; however, it often persists through adolescence into adulthood. Stimulants, a first-line treatment for this condition, are among the most effective and most studied psychotropic medications. As the diagnosis of ADHD in children and adults has increased over the past decade, the use of stimulants has also increased.
As DSM-IV-TR1 criteria now stand, at least 6 of 9 symptoms of inattention must be present for the inattentive subtype diagnosis, at least 6 of 9 hyperactive/impulsive symptoms must be present for the hyperactive/impulsive subtype diagnosis, and at least 6 of 9 symptoms for both of the first 2 subtypes must be present for a combined subtype diagnosis. These symptoms must begin in childhood and be present in at least 2 settings (eg, school, work, or home).
Establishing a diagnosis of ADHD requires the synthesis of multiple sources of information. Children with behavior disorders (especially under the age of 12 years) notoriously underreport their disruptiveness compared to the adults around them. While parents and teachers tend to agree when identifying a child with a behavioral disorder, the correlation between their reports is often low.2 In contrast, adult patients with ADHD (or their significant other) will often describe their symptoms in terms of work-related performance (eg, frequent job changes; poor organizational, prioritization, and time management skills; or excessive absences) and functional impairment (eg, effort required and coping strategies).3 Establishing a family history of ADHD is important for both children and adults given the high heritability of the condition.
Several rating scales are available to determine the presence of ADHD symptoms; these tests are most useful when combined with other information to provide a fuller picture of a patient, family, and educational/occupational functioning. Rating scales may have advantages to supplement, but not supplant, the clinical interview. Similarly, neuropsychological testing can provide objective measures of sustained attention, impulsivity, and frontal lobe functioning. An individual neuropsychological test cannot confirm symptom criteria for ADHD as much as reflect a person’s cognitive functioning at a given moment in time.4
Recent advances in genetic, neuroanatomical, and neuroimaging approaches have begun to yield insights into the pathogenetic and pathophysiologic basis of ADHD. This emerging picture suggests dysfunction of the prefrontal cortex and subcortical structures with which it is connected. However, at present, these advanced technologies cannot be used in the diagnosis of ADHD and do not replace clinical judgment on the basis of thorough information gathering. Another common pitfall in ADHD diagnosis involves equating a positive response to ADHD medicine (stimulants or otherwise) with confirmation that ADHD is present.
HOW COMMON IS ADHD AND WHO HAS IT?
ADHD affects an estimated 7% to 8% of school-aged children in the United States.5 Recent prevalence estimates indicate that boys (aged 8 to 15 years) have a 2.1 times greater prevalence of ADHD than girls (whereas girls have 2-fold higher rates of mood disorders).6 In clinical samples of children with ADHD, there may be up to a 10:1 overrepresentation of boys to girls, conceivably because disruptive behavior is often a primary reason for an initial referral (thus underrepresenting girls). Girls with predominantly inattentive symptoms are particularly prone to being underrecognized or referred for treatment.
In adult clinical samples, the male:female ratio of patients with ADHD is closer to 3:2.7 The National Comorbidity Survey Replication screened over 3,000 adults and found an estimated 4.4% prevalence of ADHD in the United States, suggesting that 9 million adults are affected nationwide.8 This prevalence rate is similar to the 3.4% estimated prevalence of ADHD in adults based on a subsequent survey of more than 11,000 adults in 10 countries in the Americas, Middle East, and Europe.9
HOW CAN ADHD BE TREATED?
As the understanding of ADHD has grown, so has the appreciation of the need to successfully diagnose and treat ADHD as early as possible in order to avoid risk of complications later in life. A range of successful treatment options exists. Of these, the stimulants are considered first-line treatments for ADHD, supported by decades of research and a history of robust response, good tolerability, and safety across the lifespan. Stimulants are classified by the US Food and Drug Administration (FDA) and the Drug Enforcement Agency as schedule 2 agents. Nonstimulant treatments (eg, atomoxetine and α2 agonists, both schedule 4 agents, respectively) are also available among current medical treatment options. Lastly, nonpharmacologic approaches—including cognitive-behavioral therapies (eg, tutoring, coaching, and traditional behavioral methods)—serve as important adjuncts to medical treatment.
WHY SHOULD ADHD BE TREATED
WHEN IT IS DIAGNOSED?
Untreated ADHD has a significant negative impact on society, resulting in stress upon families and relationships, impact on schools, decreased productivity, and increased health costs. Children with ADHD who remain untreated are at elevated risk for new psychiatric disorders later in life (eg, mood disorders, antisocial behaviors, and substance use disorders).10 Children with ADHD entering adulthood without adequate treatment demonstrate poorer driving abilities and greater traffic-related adverse outcomes,11 including risk of citations for speeding, suspended driving licenses, crashes, and crashes causing bodily injury in unmedicated patients.12 Untreated ADHD significantly increases the risk of early initiation into smoking13 and addictive behaviors overall.14
The suggestion that ADHD is overdiagnosed and overtreated—an argument often popularized in the media—is not supported by scientific literature. Prevalence rates by the National Institute of Mental Health indicate that less than a third of children with ADHD in the United States were diagnosed; moreover, in the preceding 12 months, only 13% of children diagnosed with ADHD were treated for their symptoms.15 More recently, data from the National Comorbidity Survey Adolescent Supplement showed that only 14.2% of youth with a DSM-IV disorder received psychotropic medication in the preceding year. Of those adolescents with ADHD, 20.4% reported stimulant use in the past 12 months, whereas only 0.8% of those without a DSM-IV disorder endorsed stimulant use.16
WHICH STIMULANTS ARE CURRENTLY AVAILABLE AND HOW DO THEY DIFFER FROM ONE ANOTHER?
Two groups of stimulants (methylphenidates [MPH] and amphetamines [AMPH]) have been approved by the FDA for the treatment of ADHD in the pediatric population. These agents are available in both branded and generic formulations. Table 1 lists the names, preparations, strengths, and duration of behavioral effects of the commonly used stimulants.
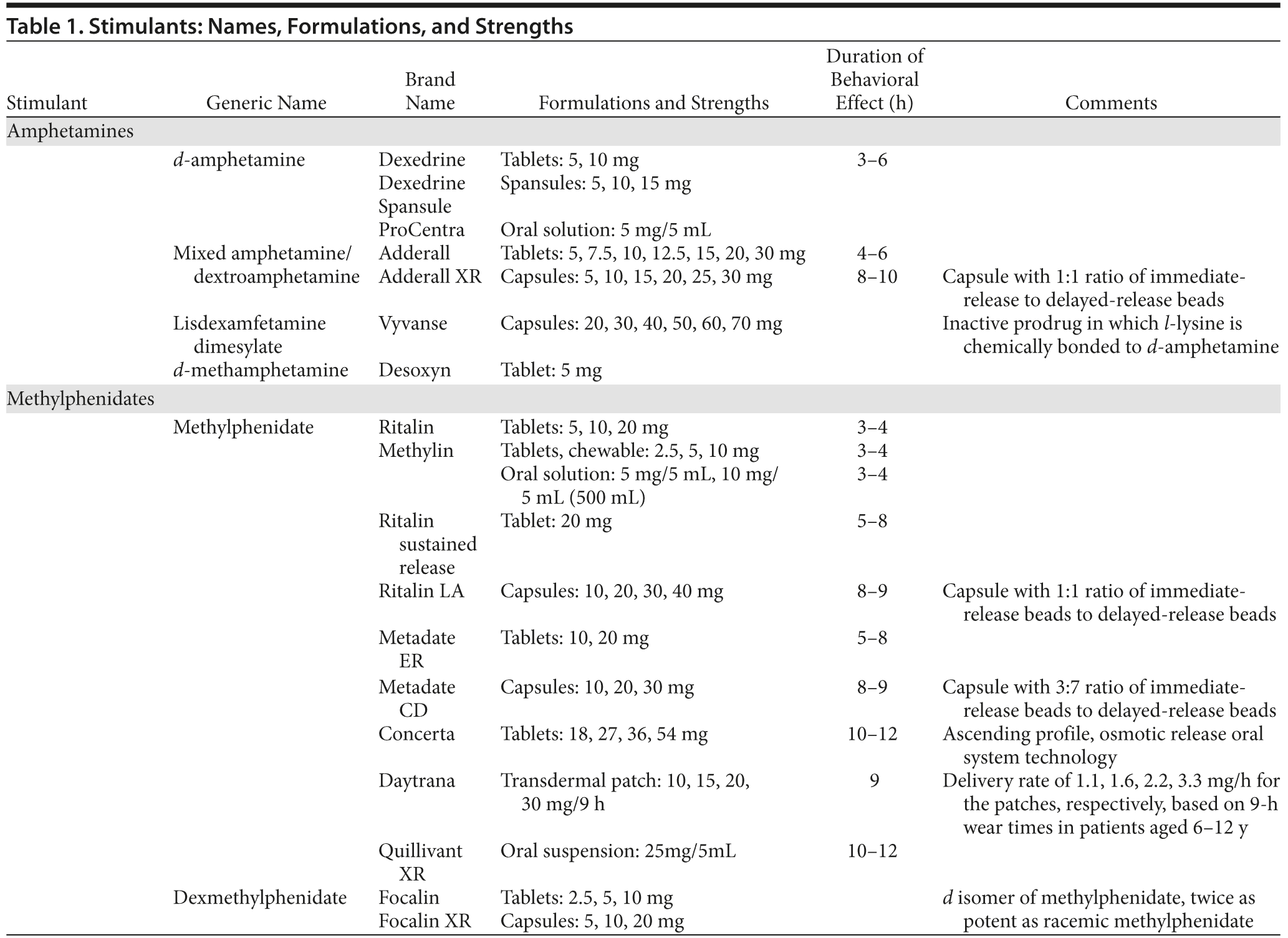
Several different stimulant formulations have been approved by the FDA since April 2000. These formulations include extended-release preparations of oral MPH, transdermal MPH, extended-release dexmethylphenidate (d-MPH), lisdexamfetamine, and an oral solution of dextroamphetamine sulfate. By and large, these longer-acting forms of MPH and AMPH circumvent the short-acting effects (eg, 3–5 hours of efficacy for ADHD) of the immediate-release stimulants.
Methylphenidate Preparations
Immediate-acting MPH comes in 3 preparations: a tablet, a chewable tablet, and an oral solution. A sustained-release form of MPH (MPH sustained release) and its branded generic (MPH extended release) are essentially identical in that they are both MPH molecules mixed into a wax matrix. A chewable, extended-release MPH preparation is also available as a branded generic, offering MPH in a hydrophilic polymer.
d-MPH is the dextroisomer of MPH. MPH, as a secondary amine, gives rise to 4 optical isomers: d-threo, l-threo, d-erythro, and l-erythro. The standard preparation of racemic MPH comprises d, l-threo-MPH. Data suggest that the d-threo-MPH (d-MPH) isomer is the active form. A head-to-head study showed d-MPH to be similar in efficacy to immediate-release, racemic MPH when used in children with ADHD, although its duration of action was longer.17 A longer-acting version of dexmethylphenidate has been approved for pediatric and adult patients with ADHD.
Of the long-acting versions of MPH, osmotic release oral system (OROS) MPH uses an osmotic pump mechanism that creates an ascending profile of MPH in the blood, providing effective treatment for up to 10 to 12 hours. OROS MPH tablets cannot be crushed without losing their prolonged action. Unlike OROS MPH, several extended-release MPH preparations—MPH controlled delivery, MPH long acting, and d-MPH extended release—utilize a beaded technology that contains spheres of active medication containing anywhere from 30% (MPH controlled delivery) to 50% (MPH long acting and d-MPH extended release) of immediate-release MPH (providing a stimulant effect during the morning hours). These beaded preparations are useful for children who cannot take or who dislike taking pills, as parents can open the capsules and sprinkle the medicine on their child’s food. These preparations are also easier-to-ingest versions of immediate-release MPH, including a chewable tablet or liquid form. These latter 2 MPH forms have not yet been subject to large-scale clinical trials.
The MPH transdermal system was approved in 2006, with a recommended “wear time” of 9 hours.18 This patch can be applied to the skin (eg, on the hip) prior to or upon awakening,19 and it is removed approximately 3 hours before the effect would be prudent to conclude.20 The MPH transdermal system is advantageous for patients (or their parents) who desire having an “off switch” for controlling the delivery of active drug. The latest MPH preparation is a once-daily liquid form of racemic MPH. Initially supplied as a powder, after reconstitution with water, it forms an extended-release oral suspension of MPH. The manufacturer recommends shaking the medication bottle vigorously to ensure that the proper dose is administered.21
Amphetamine Preparations
AMPH is manufactured in the dextro isomer, such as dextroamphetamine (d-AMPH), or in racemic forms with mixtures of d– and l-amphetamine (mixed amphetamine salts). An extended-release mixed amphetamine salt formulation is a dual-pulse capsule preparation that includes both immediate- and extended-release beads.
Lisdexamfetamine is an inactive prodrug that converts to d-AMPH upon cleavage of the lysine portion of the molecule; it was developed with the intention of creating a longer-lasting and more difficult to misuse version of d-AMPH. Lisdexamfetamine has been approved for use in both pediatric and adult patients with ADHD. Intravenously and intranasally administered lisdexamfetamine produces effects comparable to the orally administered form, thereby reducing the likelihood of abuse by these routes of administration.22
HOW ARE STIMULANTS
ABSORBED AND METABOLIZED?
Both MPH and AMPH are almost completely absorbed after oral administration23; food in the stomach has little impact on absorption.24 Immediate-release MPH reaches a peak concentration after 1.5 to 2.5 hours, and it has an elimination half-life that is independent of the preparation of MPH (2.5 to 3.5 hours after oral administration). MPH undergoes extensive presystemic metabolism through hydrolysis or de-esterification with limited oxidation.23,25 Carboxylesterase-1A1 (CES-1), located in the stomach and liver, is the primary enzyme involved with first-pass MPH metabolism. Differences in an individual’s hydrolyzing enzyme activity that are linked to variants in the human CES-1 gene26 can lead to wide variations in MPH metabolism and to corresponding MPH blood concentrations in certain individuals. MTS avoids much of the first-pass metabolism through CES-1,27 thereby producing potentially higher plasma MPH levels.
AMPH absorption is typically rapid, with peak plasma levels of AMPH generally observed 3 hours after oral ingestion.23 The half-life of AMPH is considerably longer than that of MPH (appropriately 7 hours). All types of AMPH are metabolized in the liver by side-chain oxidative deamination and by ring hydroxylation. Because AMPH formulations are basic compounds, urinary excretion is highly dependent on urinary pH. Acidification of the urine increases urinary output of AMPH.28 Therefore, taking the medicine with ascorbic acid or fruit juice may decrease its absorption, while use of alkalinizing agents (eg, sodium bicarbonate) may increase AMPH absorption29 (although the clinical correlates of these alterations remain unclear).
Peak plasma concentrations for both MPH and AMPH vary by a factor of 4 or 5 in children and adults, most likely a result of interindividual variability in metabolism and plasma clearance.25 Interindividual pharmacokinetic differences may be less dramatic when stimulant doses are adjusted for body weight (using mg/kg as a general guide).30 Interindividual variability may also argue for examining an individual’s pharmacokinetics when a person fails to respond to conventional dosing strategies.30 Laboratory testing for plasma levels of stimulants (particularly MPH) is increasingly available; however, the levels are uncommonly assessed in clinical practice.
WHEN AND HOW ARE STIMULANTS
TYPICALLY DOSED?
When starting a psychostimulant, it is wise to start by choosing a stimulant class (eg, MPH or AMPH) and then the desired duration of action of the preparation chosen (eg, longer-acting versus short-acting). There is no evidence of preferential response to one of the stimulant classes. Contemporary guidelines suggest that long-acting, once-daily preparations are preferred for most patients.31 The dose for each patient should be individually optimized on the basis of the drug’s therapeutic efficacy and side effect profile. Steady titration of treatment is advisable until an acceptable response is noted, with the addition of a similar (or sculpted) afternoon dose that is dependent on the presence of breakthrough symptoms.
The starting dose for many preparations of MPH, AMPH compounds, and d-AMPH in most children is typically 2.5 to 5 mg, with a suggested target daily dose ranging between 0.3 to 1 mg/kg for AMPH and 0.6 to 2 mg/kg for MPH. Once pharmacotherapy is initiated, frequent contact with the patient and family is necessary during the initial phase of treatment to carefully monitor the response and any adverse effects. Apparent stimulant “ineffectiveness” may stem from excessively deliberate dose titration or medication underdosing. This underdosing may occur early in the course of a stimulant trial or months (or even years) later in treatment when a previously successful stimulant regimen loses its efficacy because the child has grown and requires a higher dosage to compensate for improved stimulant metabolism and/or clearance. For instance, data suggest that over the first 6 months of treatment, MPH treatment is associated with mild tolerance that may necessitate a 20% to 30% increase in the dose to maintain efficacy.32
Stimulant dosing often varies widely depending on the treatment setting. In the National Institute of Mental Health (NIMH) Multimodal Treatment Study of ADHD,33 mean daily MPH doses for children were significantly lower for those receiving community care (18.7 mg/d) than for investigator-treated subjects (32.8 mg/d); the latter group had superior outcomes.
HOW HIGH CAN STIMULANT DOSES GO?
Absolute dose limits (in mg) of stimulants do not adequately consider use in refractory cases or adult-sized adolescents or adults. For example, the FDA recommends maximum daily MPH doses of up to 60 mg/d for short-acting forms and 72 mg/d for extended-release preparations. Others have proposed prescribing 1 mg/kg/d34 to 2 mg/kg/d of racemic MPH.35
Stevens and associates36 examined serum levels of MPH in patients receiving relatively higher doses of OROS MPH (mean of 169 mg, 3 mg/kg/d) and found acceptable levels of blood pressure and heart rates; serum levels were also within the accepted levels of therapeutic (MPH levels < 50 ng/mL). In fact, no one in that study developed toxic levels.36 While MPH levels can be helpful for patients receiving higher than FDA-approved stimulant doses, results from several studies that have examined the association between plasma levels of stimulants and the improvement of ADHD symptoms have generally been equivocal. Monitoring of serum drug levels may be of some value for confirming compliance and in patients receiving higher than FDA-approved doses of stimulants. These recommendations are tentative, and further clinical research in this area is warranted.
CAN STIMULANT THERAPY BE WITHHELD ON WEEKENDS, OVER THE SUMMER, OR ON HOLIDAYS?
The appropriateness or merits of medication holidays remain unresolved. The symptoms of ADHD, although usually more noticeable in the school or work setting, are often disruptive to the patient’s family and social life. In cases in which important adverse effects are present (eg, appetite suppression), it may be necessary to allow for periodic drug holidays (either during weekends or the summer). In patients whose major symptoms occur during school/work and who prefer to be treated during workdays only, weekends or vacations off medication may be appropriate. Conversely, in patients who manifest symptoms predominately in the home, medication-free holidays may be more problematic. In general, children should begin the academic year receiving an appropriate stimulant dose (initiated 1 or 2 weeks before the resumption of classes). Following a sufficient period of clinical stabilization (eg, 6 to 12 months), it is prudent to reevaluate the need for continued pharmacologic intervention. Supervised discontinuation trials in the middle of the school year (as opposed to the summer months) may facilitate close assessment of a child’s behavior and academic performance from multiple viewpoints.
WHAT DRUG-DRUG INTERACTIONS SHOULD BE CONSIDERED WHEN PRESCRIBING STIMULANTS?
Most interactions between stimulants and the vast majority of prescription and nonprescription medications are typically mild. Stimulants create few worrisome interactions when used with commonly prescribed medications (eg, selective serotonin reuptake inhibitors, second-generation antipsychotics, atomoxetine, or α2-adrenergic medications). However, less common interactions with stimulants may involve increased plasma levels of tricyclic antidepressants; increased plasma levels of phenobarbital, primidone, and phenytoin; increased prothrombin times on anticoagulants; attenuation or reversal of the guanethidine antihypertensive effect; and increased pressor responses to vasopressor drugs.37 The relative lack of drug-drug interactions of MPH and other psychostimulants in the context of more complex medical regimens may be due to the low bioavailability (20% to 30%) of orally administered forms of MPH.24
HOW EFFICACIOUS AND SAFE
ARE STIMULANTS FOR ADHD?
The efficacy and safety of stimulants for the treatment of pediatric patients with ADHD are based on a large number of studies of (primarily) latency-age children wherein the average response rate is 70%.38–40 When clinical response is assessed quantitatively via rating scales, the effect size of stimulant treatment relative to placebo is robust, averaging about 1.0, one of the largest effects for any psychotropic medication.31,41
Treatment data for preschoolers (aged 3 to 5 years) are less robust, although studies suggest that this group may have a lower response rate to stimulants and may be more treatment refractory or be diagnostically heterogenous.42 Results from the largest preschool stimulant treatment study to date, a NIMH-sponsored multisite study of 165 children who tolerated study medication, showed that 85% of patients were MPH responders (versus 10% of placebo responders).43 The effect sizes were smaller than in school-aged youth (with improvement noted in both school and home settings). Findings from the NIMH multisite Preschool ADHD Treatment Study study support the results of earlier, smaller, controlled stimulant studies in preschoolers, showing mostly a modest-to-robust response and improvements in mother-child interactions, behavior, and structured tasks.44
As with preschoolers, there is a smaller body of research of stimulants in adolescents.45,46 The majority of existing studies report at least a moderate response to treatment (without tolerance or evidence of misuse or abuse). For instance, 2 multisite studies demonstrated the efficacy of OROS MPH and mixed amphetamine salts extended release for ADHD. In a controlled study of 177 adolescents with ADHD treated with OROS MPH, over a third (37%) required the highest FDA-approved dose (72 mg) for outcome.45 In another controlled study of 318 adolescents with ADHD, mixed amphetamine salts extended release resulted in significant improvements in mean ADHD measures of inattentiveness and hyperactivity/impulsivity subscales versus placebo.46 In the majority of stimulant studies previously cited, the most common study drug was MPH, followed by AMPH. A review of the existing literature provides little evidence of a differential response for various stimulants.44 Moreover, in many stimulant studies, a crossover design was used and the study lengths were brief (ranging from a few days to a few weeks).45 Most studies have been conducted on white males; there have been fewer data on the safety and efficacy of stimulants in females and those from various minority groups.47
WHICH STIMULANT-RELATED ADVERSE
EFFECTS ARE COMMON AND EXPECTED?
Common adverse effects during stimulant treatment include the delay of sleep onset, headache, appetite suppression, transient headache, transient stomachache, and behavioral rebound (ie, the sudden or pronounced recurrence of ADHD symptoms). Less frequently observed outcomes include mood dysregulation or tics.
Charach et al48 collected side effect data during a 5-year period in children with ADHD who were initially randomized to use of a stimulant or to no medication. The most common sustained side effect reported was loss of appetite. At least 1 physiologic adverse effect (eg, headache, appetite loss, abdominal pain) was reported by half of the children by the end of the fifth year of monitoring. Importantly, children continued to use the medication, suggesting that the adverse events were mild and of minor health concern.48 Although AMPH or MPH were equally likely to produce an improvement in ADHD symptoms,49 the occurrence of stimulant-related side effects may be greater with the use of AMPH (or d-AMPH) compared with MPH (10% versus 6%, P < .05), although idiosyncratic patterns were noted for individual children.50
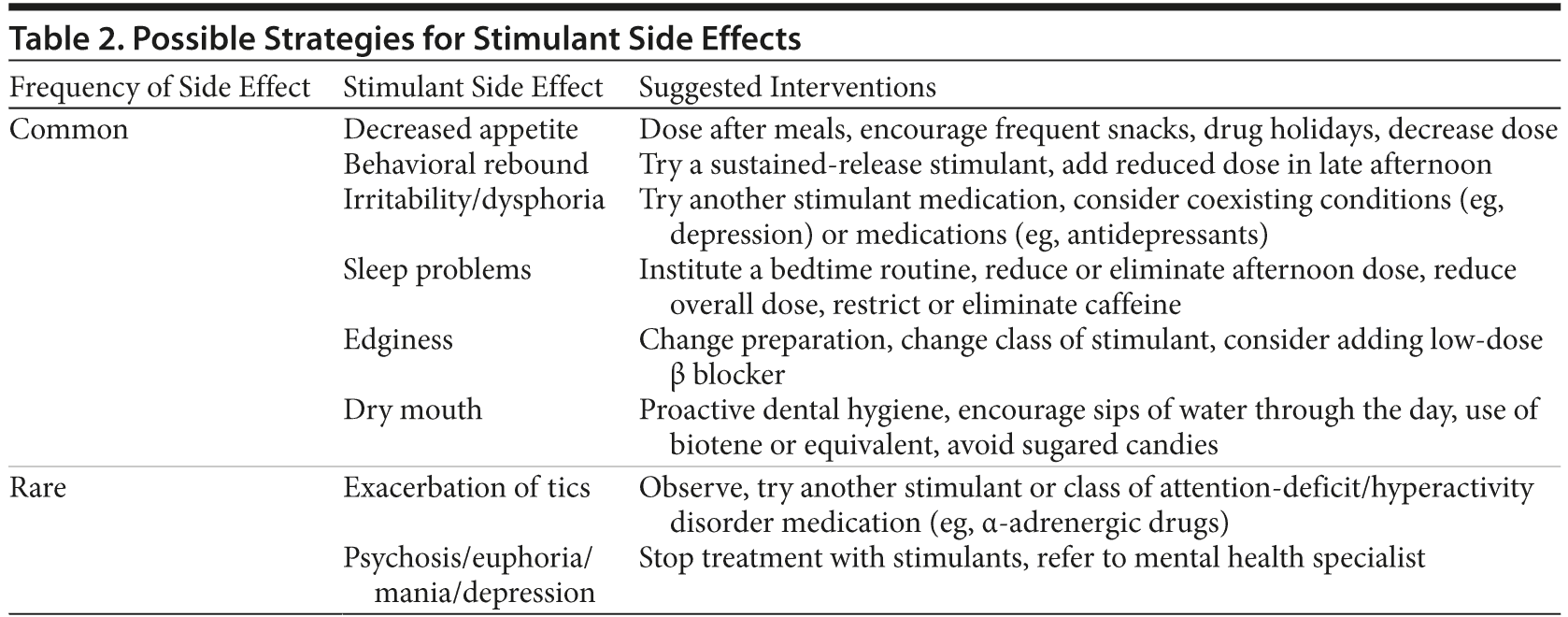
Table 2 delineates suggested strategies for managing common stimulant-related adverse effects. Attempts should be made to manage adverse effects that occur in the context of a satisfactory clinical response to stimulants. In cases of stimulant-induced medical (headaches) or psychiatric symptoms (eg, dysphoria, anxiety), it is necessary to assess whether these symptoms develop 1 to 2 hours postadministration (acute phase) or during the postadministration (wear-off phase). Acute effects generally indicate the need to reduce the maximum concentration in the blood by reducing the dose or altering the release mechanism of the stimulant (eg, changing from immediate-release to extended-release forms), whereas wear-off symptoms necessitate slowing of the stimulant decay curve by adding a stimulant just before symptom onset or changing to a more extended-release agent.
WHAT ARE THE LONG-TERM OR
UNEXPECTED EFFECTS OF STIMULANTS?
Height and Weight Changes
Numerous studies have investigated abnormalities in the growth process related to ADHD, but controversies remain concerning both the direction of the deviation from the norm and the cause of that deviation. Even with the plethora of studies conducted in this area, myriad methodological difficulties interfere with drawing a simple conclusion (eg, the absence of a comparable control group [untreated children with ADHD] or an ADHD group receiving medication treatment with psychotropics other than stimulants).
In the past 10 years, several investigations have confirmed that reduction of growth is stimulant related,50–53 whereas others have failed to show any statistically significant growth delay during treatment. Growth slowdown for height and weight was reported in children aged 7 to 10 years with ADHD who were treated with MPH at a mean dosage of 30 mg/kg/d in the NIMH Multimodal Treatment Study of ADHD.52 School-aged children grew 1.0 cm less and gained 2.5 kg less than predicted by the Centers for Disease Control and Prevention growth charts.52 Similar effects were observed for preschool children who grew 1.5 cm less in height and gained 2.5 kg less weight than predicted while being treated with MPH at a mean dosage of 14 mg/kg/d.54
Spencer and associates55 hypothesized that the disorder-related delay suggested that the observed growth deficit may be connected with ADHD, rather than with stimulant medication. Children with ADHD could develop more slowly than the norm, and the consequence of this would be lower rates of growth in succeeding years than expected and the later achievement of biological maturity than in their healthy peers. Others have reported dissimilar findings50 and have shown that unmedicated children with ADHD were taller than were medicated children.
The aggregate literature seems to suggest small but clinically insignificant reductions in weight over time. Height may be negatively influenced over the first 1 to 2 years of treatment; however, catch-up or rebound in height to expected values emerges with chronic treatment.56 Monitoring of growth (in height and weight) should be part of the management of ADHD-affected youth receiving stimulants.
Tics
Stimulant use in patients with mild-to-moderate tics remains of concern. Most studies have failed to confirm the hypothesis that stimulants exacerbate tic severity in youth with ADHD.57,58 For example, Palumbo and colleagues59 pooled data from 3 placebo-controlled trials (total N = 416) and 2 open-label studies involving patients receiving OROS MPH, MPH, or placebo. Although approximately 13% of patients in each of the 3 groups had a history of tics, there was no significant difference between the 3 groups in the number of children who experienced tics.59 Double-blind clinical trials of both immediate-release and long-acting stimulants have not found that stimulants increase the rate of tics relative to placebo.60,61 Children with comorbid ADHD and tic disorders, on average, show a decline in tics that persists even after a year of treatment when treated with a stimulant.62
If a patient has treatment-emergent tics during a trial of a given stimulant, an alternative stimulant or a nonstimulant should be tried. If the patient’s ADHD symptoms respond adequately only to a stimulant that induces tics, then combined pharmacotherapy of the stimulant and an α-agonist (eg, clonidine or guanfacine) is recommended.63 Withdrawal of stimulants in a placebo-controlled double-blind fashion did not change the frequency or severity of tics in a series of 19 children with ADHD, and the frequency and severity of vocal or motor tics did not change when stimulants were used for an extended time.64
Sudden Death and Cardiac Complications
A disputed cardiovascular effect of stimulants is sudden death; this has been a source of great concern and publicity in recent years. Wilens and coworkers65 summarized the literature, and a commentary was provided by the FDA in 2009.66 Wilens and colleagues65 cited more than 300 controlled trials of stimulant medication involving more than 5,000 subjects; no cases of sudden death were observed. Moreover, the autopsy-assessed anatomic characteristics of subjects who had sudden death during stimulant treatment were found to be similar to those of individuals who had sudden death in the general population. However, Gould and associates67 found an association between stimulant use and sudden death in a matched case-control study of 564 cases of sudden unexplained death in youth and a comparison group (who died as passengers in motor vehicle traffic accidents). Ten of the subjects (1.8%) in the unexplained sudden death group were taking MPH, whereas only 2 (0.4%) in the comparison group of young people who died in road traffic accidents were taking stimulants (odds ratio of 7.4, 95% CI, 1.4–74.9). As a case-controlled study, this analysis could not determine causality.67 Referring to this study, in 2009 the FDA commented that it was unable to “conclude that the data in the study affect the overall risk-benefit profile of stimulant medications used to treat ADHD in children.” 66
More recently, Hammerness and colleagues68 reviewed relevant clinical literature through 2011 and found that stimulants were associated with small elevations of blood pressure (≤ 5 mm Hg) and heart rate (≤ 10 beats/min) without electrocardiographic changes (eg, QTc prolongation). However, it was extremely rare for a child or adolescent receiving stimulants to have a serious cardiovascular event during treatment; in fact, the overall cardiovascular risk was the same as that of groups of youth not receiving stimulant medication.68 This last finding was further supported by a large retrospective cohort study69 involving greater than 1 million children and young adults analyzed for potential serious cardiovascular events (eg, sudden death, acute myocardial infarction, or stroke). Users of ADHD medicine were not at an increased risk for serious cardiovascular events compared to nonusers or former users of stimulants.69
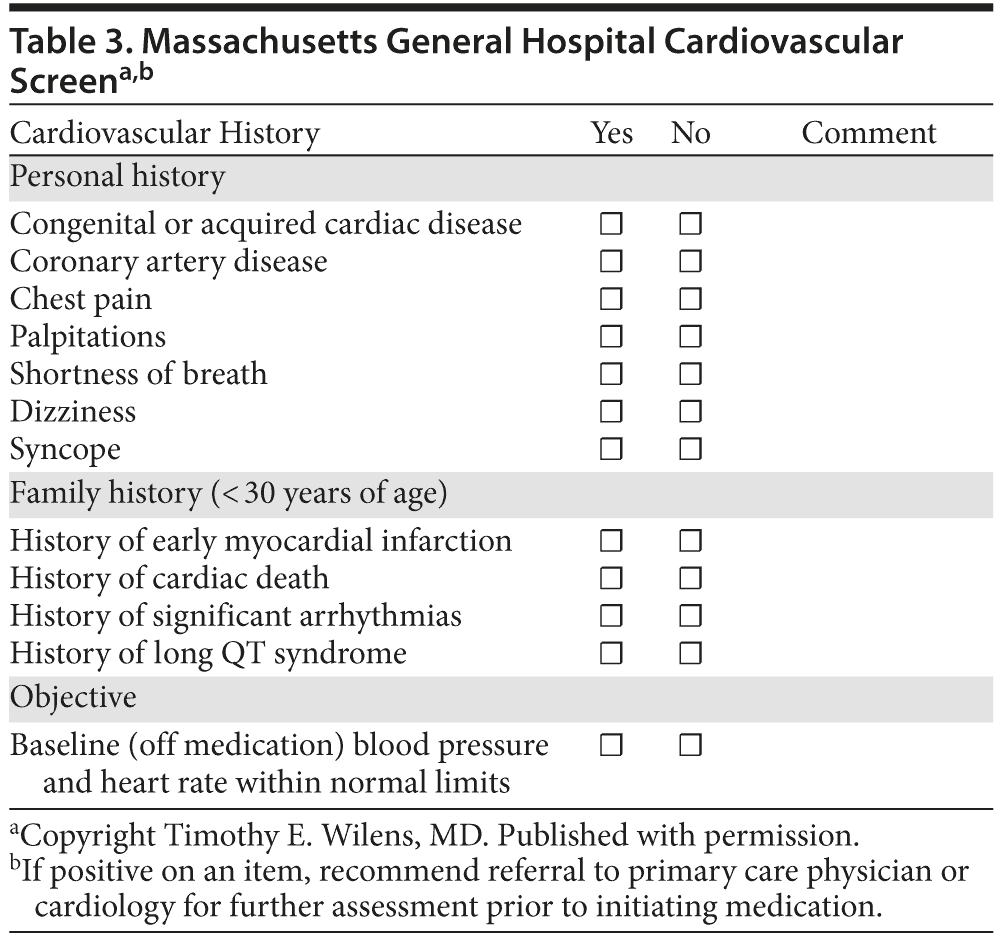
At this stage, it appears that sudden death is a rare event in youth with ADHD, and there are insufficient data to establish a causal link with stimulant medication used to treat this condition.70 Nevertheless, it would be prudent to follow the guidelines of the use of stimulants in youth (monitoring risk factors and measuring blood pressure and pulse), although obtaining an electrocardiogram is not mandatory.71,72 Pertinent questions that should be asked with regard to family history of premature sudden death and a personal history of cardiovascular symptoms are summarized in Table 3. Patients answering any of these questions affirmatively should be examined and investigated more carefully.
Psychosis or Mania
Psychosis or mania may be a rare adverse effect of stimulant (and nonstimulant) therapy for ADHD. A recent pooled analysis73 of 49 randomized trials in children and > 800 reports in adults and children treated for ADHD (some with nonstimulants, including atomoxetine) found events in 1.48 per 100 person-years in the pooled drug group compared to none in the placebo group. In approximately 90% of the cases, there was no history of a similar psychiatric condition prior to ADHD treatment.73 Hallucinations (involving visual and/or tactile sensations of insects, snakes, or worms) were common in cases involving children.73 In reviewing several trials, the FDA found that stimulant-associated psychotic-like and manic-like symptoms occurred rarely (ie, in approximately 0.25% of children treated with stimulants). In 55 of 60 reported cases of psychotic-like or manic-like symptoms in response to stimulants, the symptoms resolved when the stimulant was discontinued.73 In the 5 cases in which symptoms persisted, the patients were rediagnosed with schizophrenia or bipolar disorder. The occurrence of psychosis and other pronounced mood dysregulation with stimulant treatment generally warrants consideration of an alternative agent. In some cases, children may tolerate a carefully monitored rechallenge of a stimulant used at a lower dose.73
Carcinogenic Effects
In 2005, El-Zein and colleagues74 reported “chromosomal breaks” in the peripheral lymphocytes of children taking therapeutic doses of MPH. This study added to a sparse body of animal literature that showed increased hepatic tumors in rodents treated with very high (4 to 47 mg/kg) oral doses of MPH.75 However, the preponderance of data seems to suggest a lack of association between the use of stimulants and the development of cancer. For instance, an older study of pharmacy records suggested that the number of cancers in patients receiving stimulants was actually less than expected.76 Moreover, a large systematic review found either negative or weakly positive results for chromosomal changes in rodent assay systems.77 In what is likely to be one of many follow-up studies addressing this pressing issue, Ponsa and associates78 followed a similar strategy as El-Zein and coworkers74 and found no evidence of an increased frequency of micronuclei (indicative of genomic damage). In the 3 endpoints studied (ie, a cytokinesis-block micronucleus assay, a sister chromatid exchange analysis, and a determination of chromosome aberrations), the results did not support a potential increased risk of cancer after exposure.
WHAT IS THE RISK OF SUBSEQUENT SUBSTANCE ABUSE AFTER TREATMENT WITH A STIMULANT?
Wilens and colleagues79 performed a meta-analysis and reported that the use of stimulants did not increase the risk for later substance use disorders in either adolescents or adults. Subsequently, Katusic and colleagues80 and Wilens and associates81 reported a protective effect of stimulants into young adulthood, whereas Biederman et al82 and Mannuzza et al83 simultaneously reported that early stimulant treatment neither increased nor decreased the risk for subsequent substance use disorders in young adulthood. The waning of the protective effect in adults may be a reflection of findings in adolescents not spanning the full age of risk of substance use disorders or that most adolescents have stopped their ADHD treatment, thus losing the protective effect of stimulants.
HOW ARE STIMULANTS MISUSED AND DIVERTED?
There has been substantial interest in the misuse and diversion of stimulants prescribed for ADHD (for review see Wilens et al84). While the majority of individuals treated for their ADHD use their medications appropriately,85 some appear to misuse the stimulants. Survey studies have indicated that approximately 5% of college students have misused stimulants86,87 and that it is more common in competitive colleges and that the drugs are more often misused for their procognitive effects than for euphoria87 Data regarding those to whom the stimulants were diverted show that these individuals misused the stimulants in context with other substances of abuse88 and other psychopathology (such as depression89 and conduct disorder87).
CAN STIMULANTS BE USED EFFECTIVELY
IN THOSE WITH COMORBID CONDITIONS?
The use of stimulants in the treatment of ADHD and common comorbid conditions is an important topic. The lifetime prevalence of comorbid psychiatric or learning disorders is estimated to be as high as 80%.35,90 Common comorbid diagnoses with ADHD include mood disorders (eg, major depressive disorder, bipolar disorder, dysthymia), anxiety disorders, and substance use disorders. Some medical conditions (eg, epilepsy) are more likely to be comorbid with ADHD. The presence of comorbidities often worsens the prognosis of these patients and complicates their treatment, thereby warranting special mention.
Bipolar Disorder
The majority of youth with bipolar disorder have co-occurring psychiatric illnesses. Among these, ADHD is the most common comorbidity with pediatric bipolar disorder.91 Clinical studies generally demonstrate high rates of ADHD, ranging from 60% to 90%, in patients who have bipolar disorder.92 Likewise, cross-sectional studies have found rates of bipolar disorder ranging from 11% to 23% in youth who have ADHD.93
Two controlled trials suggest that stimulants are effective in treating comorbid ADHD without precipitating hypomania or mania in mood-stabilized youth with bipolar disorder. Scheffer and colleagues94 performed a double-blind, placebo-controlled trial of pediatric patients diagnosed with bipolar disorder and ADHD. In this study of 31 children, treatment with divalproex sodium (mean dose: 750 mg/d) reduced manic symptoms in 80% of participants, but reduced ADHD symptoms in only 7.5% of participants. With the addition of AMPH, the subsequent improvement in ADHD was significantly greater than with placebo.94 Findling and colleagues95 reported that in youth stabilized with a dose of at least 1 mood stabilizer, concomitant treatment with MPH improved ADHD in a dose-dependent manner (without destabilization of mood). Therefore, for bipolar youth with co-occurring ADHD, mood stabilization with a traditional mood stabilizer or an atypical antipsychotic medication is recommended before starting stimulant therapy.96 Clinicians must take into account the risk of adverse effects or potential mood destabilization from stimulants and discuss this risk with families; however, they should not overvalue potential risks when making a recommendation.
Developmental Disorders
The recent rise in the diagnosis of autistic disorder and other pervasive developmental disorders has refocused attention on stimulants as a possible therapeutic option for patients with functionally impairing hyperactivity, distractibility, and impulsiveness.
Several randomized, controlled studies have supported the appropriateness of stimulant use in pervasive developmental disorders in children.97–100 These studies contradict the results of a retrospective review of stimulant use in 195 patients with pervasive developmental disorders,101 which suggested a low rate of treatment success and frequent side effects (eg, irritability and increased stereotypic movements). A preponderance of the evidence suggests that symptoms of ADHD are common in pervasive developmental disorders and that MPH is an empirically supported treatment to target ADHD symptoms in pervasive developmental disorders. However, tolerability remains a problem, and caregivers should be cautioned to be watchful for potential adverse effects. With the increasing recognition of autism spectrum disorders in young children, further studies are needed to clarify the roles of AMPH and nonstimulant medications in the treatment of ADHD symptoms in children with pervasive developmental disorders.
Seizure Disorders
Studies in pediatric epilepsy have found a 2.5-fold to 5.5-fold increased risk of ADHD compared with healthy controls.102,103 Although not a contraindication, the Physicians’ Desk Reference104 discourages the use of stimulants in children with seizure disorders because stimulants lower the seizure threshold. Baptista-Neto and colleagues105 have reviewed this topic, citing retrospective chart reviews, open-label trials, and controlled trials of MPH in patients with epilepsy and ADHD showing significant improvements in ADHD symptoms without an exacerbation of seizures or an adverse effect on antiepileptic drug serum levels. Moreover, a large retrospective cohort study of > 30,000 pediatric patients found no statistically significant association between the use of stimulants and seizure risk in children with ADHD and without a prior seizure disorder.106 Recently, a pilot randomized controlled trial of OROS MPH to treat ADHD plus epilepsy (N = 31) found that stimulant treatment reduced ADHD symptoms more than placebo treatment.107 However, there were too few seizures during the active (5 seizures) and placebo (3 seizures) arms to assess seizure risk.107 These recent works suggest that stimulants may be a safe and effective treatment in certain children with seizure disorders.
CAN STIMULANTS PROTECT AGAINST THE DEVELOPMENT OF PSYCHIATRIC DISORDERS?
One of the newest areas of interest discussed in the stimulant literature concerns whether treatment with stimulants modifies long-term outcomes. Daviss and colleagues108 examined the association between stimulant treatment for ADHD and the risk for subsequent major depressive disorder by comparing the rates of pharmacotherapy in teenagers with ADHD and major depressive disorder (history of major depressive disorder: n = 36) to those without a lifetime history of major depressive disorder (never depressed: n = 39). The investigators found that stimulants protected youth against subsequent development of major depressive disorder. More recently, a case-control, 10-year prospective follow-up study of white males with (n = 140) and without (n = 120) ADHD was conducted.109 At the 10-year follow-up, participants with ADHD who were treated with stimulants were significantly less likely to develop depressive and anxiety disorders and disruptive behavior and were less likely to repeat a grade compared with participants with ADHD who were not treated. This study highlights additional protective effects of stimulants (eg, lower risk for the subsequent development of psychopathology and grade retention).109
WHEN TO REFER TO A SPECIALIST?
Primary care physicians may consider referring a difficult or unclear case to a psychiatrist, psychologist, or developmental pediatrician, especially if the physician suspects comorbid conditions (eg, depression, anxiety disorders, or bipolar disorder). If the symptom presentation cannot be differentiated from neurologic disorders such as seizures, referral to a neurologist is appropriate. Patients who are refractory to treatment (ie, fail 2 classes of stimulants) or who experience an atypical response are also ideal candidates for referral.2,4 Ultimately, a clinician’s referral offers the opportunity to confirm or refine a diagnosis, optimize treatment, and achieve greater adherence with treatment regimens such as pharmacotherapy.
CONCLUSION
Pharmacotherapy with stimulants is a mainstay of evidence-based treatment for pediatric and adult populations with ADHD and comorbid conditions. The use of stimulants should follow a careful evaluation of the child and his or her family, including psychiatric, social, cognitive, and educational evaluations. Early therapeutic intervention is important before complications, chronicity, and social incapacitation occur. Otherwise, the challenge of treatment and restabilization of functional life habits becomes more difficult. The patient and family of the patient need to be made familiar with the risks and benefits of such intervention, the availability of alternative treatments, and the likely short-term and long-term adverse effects. Certain adverse effects can be anticipated on the basis of known pharmacologic properties of the drug (eg, decreased appetite, insomnia), whereas others are idiosyncratic and are difficult to anticipate on the basis of the drug properties. Short-term adverse effects can be minimized by introducing the medication at low initial doses and titrating upward steadily. Long-term side effects require monitoring of potential adverse effects (such as growth impairment). Idiosyncratic adverse effects generally require drug discontinuation and selection of alternative treatment modalities. Special attention must be given to issues of comorbidity with other psychiatric and medical disorders.
REFERENCES
1. American Psychiatric Association. Diagnostic and Statistical Manuel of Mental Disorders, Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
2. Baumgaertel A, Wolraich M. Practice guidelines for the diagnosis and management of attention deficit hyperactivity disorder. Ambul Child Health. 1998;4:45–48.
3. Biederman J, Faraone SV, Spencer T, et al. Patterns of psychiatric comorbidity, cognition, and psychosocial functioning in adults with attention deficit hyperactivity disorder. Am J Psychiatry. 1993;150(12):1792–1798. PubMed
4. Manos MJ. Diagnosing ADHD in children and adolescents: a guide for the practicing clinician. In: Biederman JB, ed. ADHD Across the Lifespan: An Evidence-Based Understanding From Research to Clinical Practice. Hasbrouck Heights, NJ: Veritas Institute for Medical Education; 2006:111–141.
5. Centers for Disease Control and Prevention (CDC). Mental health in the United States: prevalence of diagnosis and medication treatment for attention-deficit/hyperactivity disorder—United States, 2003. MMWR Morb Mortal Wkly Rep. 2005;54(34):842–847. PubMed
6. Merikangas KR, He JP, Brody D, et al. Prevalence and treatment of mental disorders among US children in the 2001–2004 NHANES. Pediatrics. 2010;125(1):75–81. doi:10.1542/peds.2008-2598 PubMed
7. Biederman J, Faraone SV, Spencer T, et al. Gender differences in a sample of adults with attention deficit hyperactivity disorder. Psychiatry Res. 1994;53(1):13–29. doi:10.1016/0165-1781(94)90092-2 PubMed
8. Kessler RC, Adler L, Barkley R, et al. The prevalence and correlates of adult ADHD in the United States: results from the National Comorbidity Survey Replication. Am J Psychiatry. 2006;163(4):716–723. doi:10.1176/appi.ajp.163.4.716 PubMed
9. Fayyad J, De Graaf R, Kessler R, et al. Cross-national prevalence and correlates of adult attention-deficit hyperactivity disorder. Br J Psychiatry. 2007;190(5):402–409. doi:10.1192/bjp.bp.106.034389 PubMed
10. Biederman J. Impact of comorbidity in adults with attention-deficit/hyperactivity disorder. J Clin Psychiatry. 2004;65(suppl 3):3–7. PubMed
11. Fischer M, Barkley RA, Smallish L, et al. Hyperactive children as young adults: driving abilities, safe driving behavior, and adverse driving outcomes. Accid Anal Prev. 2007;39(1):94–105. doi:10.1016/j.aap.2006.06.008 PubMed
12. Barkley RA, Murphy KR, Kwasnik D. Motor vehicle driving competencies and risks in teens and young adults with attention deficit hyperactivity disorder. Pediatrics. 1996;98(6, pt 1):1089–1095. PubMed
13. Milberger S, Biederman J, Faraone SV, et al. ADHD is associated with early initiation of cigarette smoking in children and adolescents. J Am Acad Child Adolesc Psychiatry. 1997;36(1):37–44. doi:10.1097/00004583-199701000-00015 PubMed
14. Biederman J, Faraone SV, Spencer TJ, et al. Functional impairments in adults with self-reports of diagnosed ADHD: a controlled study of 1001 adults in the community. J Clin Psychiatry. 2006;67(4):524–540. doi:10.4088/JCP.v67n0403 PubMed
15. Jensen PS, Kettle L, Roper MT, et al. Are stimulants overprescribed? treatment of ADHD in four US communities. J Am Acad Child Adolesc Psychiatry. 1999;38(7):797–804. doi:10.1097/00004583-199907000-00008 PubMed
16. Merikangas KR, He J, Rapoport J, et al. Medication use in US youth with mental disorders. JAMA Pediatr. 2013;167(2):141–148. doi:10.1001/jamapediatrics.2013.431 PubMed
17. Wigal S, Swanson JM, Feifel D, et al. A double-blind, placebo-controlled trial of dexmethylphenidate hydrochloride and d,l-threo-methylphenidate hydrochloride in children with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2004;43(11):1406–1414. doi:10.1097/01.chi.0000138351.98604.92 PubMed
18. Findling RL, Lopez FA. Efficacy of transdermal methylphenidate with reference to Concerta in ADHD. 25th Annual Meeting of the American Academy of Child and Adolescent Psychiatry; October 18–23, 2005; Toronto, Canada.
19. Wilens TE, Hammerness P, Martelon M, et al. A controlled trial of the methylphenidate transdermal system on before-school functioning in children with attention-deficit/hyperactivity disorder. J Clin Psychiatry. 2010;71(5):548–556. doi:10.4088/JCP.09m05779pur PubMed
20. Wilens TE, Boellner SW, López FA, et al. Varying the wear time of the methylphenidate transdermal system in children with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2008;47(6):700–708. doi:10.1097/CHI.0b013e31816bffdf PubMed
21. Medication Guide. Quillivant XR. http://www.fda.gov/downloads/Drugs/DrugSafety/UCM322209.pdf.
22. Jasinski DR, Krishnan S. Human pharmacology of intravenous lisdexamfetamine dimesylate: abuse liability in adult stimulant abusers. J Psychopharmacol. 2009;23(4):410–418. doi:10.1177/0269881108093841 PubMed
23. Patrick KS, Markowitz JS. Pharmacology of methylphenidate, amphetamine enantiomers and pemoline in attention-deficit hyperactivity disorder. Hum Psychopharmacol. 1997;12(6):527–546. doi:10.1002/(SICI)1099-1077(199711/12)12:6<527::AID-HUP932>3.0.CO;2-U
24. Gualtieri CT, Wargin W, Kanoy R, et al. Clinical studies of methylphenidate serum levels in children and adults. J Am Acad Child Psychiatry. 1982;21(1):19–26. doi:10.1097/00004583-198201000-00005 PubMed
25. Markowitz JS, Patrick KS. Differential pharmacokinetics and pharmacodynamics of methylphenidate enantiomers: does chirality matter? J Clin Psychopharmacol. 2008;28(suppl 2):S54–S61. doi:10.1097/JCP.0b013e3181733560 PubMed
26. Zhu HJ, Patrick KS, Yuan HJ, et al. Two CES1 gene mutations lead to dysfunctional carboxylesterase 1 activity in man: clinical significance and molecular basis. Am J Hum Genet. 2008;82(6):1241–1248. doi:10.1016/j.ajhg.2008.04.015 PubMed
27. Zhu HJ, Wang JS, Donovan JL, et al. Interactions of attention-deficit/hyperactivity disorder therapeutic agents with the efflux transporter P-glycoprotein. Eur J Pharmacol. 2008;578(2–3):148–158. doi:10.1016/j.ejphar.2007.09.035 PubMed
28. Greenhill LL, Pliszka S, Dulcan MK, et al; American Academy of Child and Adolescent Psychiatry. Practice parameter for the use of stimulant medications in the treatment of children, adolescents, and adults. J Am Acad Child Adolesc Psychiatry. 2002;41(suppl 2):26S–49S. doi:10.1097/00004583-200202001-00003 PubMed
29. Vitiello B. Research in child and adolescent psychopharmacology: recent accomplishments and new challenges. Psychopharmacology (Berl). 2007;191(1):5–13. doi:10.1007/s00213-006-0414-3 PubMed
30. Angrist B, Corwin J, Bartlik B, et al. Early pharmacokinetics and clinical effects of oral D-amphetamine in normal subjects. Biol Psychiatry. 1987;22(11):1357–1368. doi:10.1016/0006-3223(87)90070-9 PubMed
31. Pliszka S; AACAP Work Group on Quality Issues. Practice parameter for the assessment and treatment of children and adolescents with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(7):894–921. doi:10.1097/chi.0b013e318054e724 PubMed
32. Wilens T, Pelham W, Stein M, et al. ADHD treatment with a once-daily formulation of methylphenidate hydrochloride: a two-year study. Presented at: 156th Annual Meeting of the American Psychiatric Association; May 17–22, 2003; San Francisco, CA.
33. Jensen PS, Hinshaw SP, Swanson JM, et al. Findings from the NIMH Multimodal Treatment Study of ADHD (MTA): implications and applications for primary care providers. J Dev Behav Pediatr. 2001;22(1):60–73. doi:10.1097/00004703-200102000-00008 PubMed
34. Sachdev PS, Trollor JN. How high a dose of stimulant medication in adult attention deficit hyperactivity disorder? Aust N Z J Psychiatry. 2000;34(4):645–650. doi:10.1080/j.1440-1614.2000.00732.x PubMed
35. Biederman J, Mick E, Surman C, et al. A randomized, placebo-controlled trial of OROS methylphenidate in adults with attention-deficit/hyperactivity disorder. Biol Psychiatry. 2006;59(9):829–835. doi:10.1016/j.biopsych.2005.09.011 PubMed
36. Stevens JR, George RA, Fusillo S, et al. Plasma methylphenidate concentrations in youths treated with high-dose osmotic release oral system formulation. J Child Adolesc Psychopharmacol. 2010;20(1):49–54. doi:10.1089/cap.2008.0128 PubMed
37. Stevens JR, Fava M, Rosenbaum JF, et al. Psychopharmacology in the medical setting. In: Stern TA, Fricchione GL, Cassem NH, et al, eds. Massachusetts General Hospital Handbook of General Hospital Psychiatry. 6th ed, Chapter 34. Philadelphia: Elsevier Science Publishers; 2010: 441–466.
38. The MTA Cooperative Group. Multimodal Treatment Study of Children with ADHD. A 14-month randomized clinical trial of treatment strategies for attention-deficit/hyperactivity disorder. Arch Gen Psychiatry. 1999;56(12):1073–1086. doi:10.1001/archpsyc.56.12.1073 PubMed
39. Spencer T, Biederman J, Wilens T, et al. Pharmacotherapy of attention-deficit hyperactivity disorder across the life cycle. J Am Acad Child Adolesc Psychiatry. 1996;35(4):409–432. doi:10.1097/00004583-199604000-00008 PubMed
40. Spencer T, Biederman J, Wilens T. Pharmacotherapy of ADHD: A life span perspective. In: Oldham J, Riba M, eds. American Psychiatric Press Review of Psychiatry. Washington, DC: American Psychiatric Association; 1997:87–128.
41. Faraone SV, Biederman J, Spencer TJ, et al. Comparing the efficacy of medications for ADHD using meta-analysis. MedGenMed. 2006;8(4):4. PubMed
42. Byrne JM, Bawden HN, DeWolfe NA, et al. Clinical assessment of psychopharmacological treatment of preschoolers with ADHD. J Clin Exp Neuropsychol. 1998;20(5):613–627. doi:10.1076/jcen.20.5.613.1121 PubMed
43. Greenhill LL, Abikoff H, Kollins S, et al. CS Outcome results from the NIMH multi-site Preschool ADHD Treatment Study (PATS). 51st Annual Meeting of the American Academy of Child and Adolescent Psychiatry; October 19–24, 2004; Washington, DC.
44. Wilens TE. Treatment and assessment of ADHD in children and adolescents. In: Biederman JB, ed. ADHD across the Lifespan: An Evidence-Based Understanding from Research to Clinical Practice. Hasbrouck, NJ: Veritas Institute for Medical Education; 2006:176–206.
45. Wilens TE, McBurnett K, Bukstein O, et al. Multisite controlled study of OROS methylphenidate in the treatment of adolescents with attention-deficit/hyperactivity disorder. Arch Pediatr Adolesc Med. 2006;160(1):82–90. doi:10.1001/archpedi.160.1.82 PubMed
46. Grcevich S. SLI381: a long-acting psychostimulant preparation for the treatment of attention-deficit hyperactivity disorder. Expert Opin Investig Drugs. 2001;10(11):2003–2011. doi:10.1517/13543784.10.11.2003 PubMed
47. Pelham WE Jr, Walker JL, Sturges J, et al. Comparative effects of methylphenidate on ADD girls and ADD boys. J Am Acad Child Adolesc Psychiatry. 1989;28(5):773–776. doi:10.1097/00004583-198909000-00021 PubMed
48. Charach A, Ickowicz A, Schachar R. Stimulant treatment over five years: adherence, effectiveness, and adverse effects. J Am Acad Child Adolesc Psychiatry. 2004;43(5):559–567. doi:10.1097/00004583-200405000-00009 PubMed
49. Barbaresi WJ, Katusic SK, Colligan RC, et al. Long-term stimulant medication treatment of attention-deficit/hyperactivity disorder: results from a population-based study. J Dev Behav Pediatr. 2006;27(1):1–10. doi:10.1097/00004703-200602000-00001 PubMed
50. Swanson JM, Elliott GR, Greenhill LL, et al. Effects of stimulant medication on growth rates across 3 years in the MTA follow-up. J Am Acad Child Adolesc Psychiatry. 2007;46(8):1015–1027. doi:10.1097/chi.0b013e3180686d7e PubMed
51. Poulton A, Cowell CT. Slowing of growth in height and weight on stimulants: a characteristic pattern. J Paediatr Child Health. 2003;39(3):180–185. doi:10.1046/j.1440-1754.2003.00107.x PubMed
52. MTA Cooperative Group. National Institute of Mental Health Multimodal Treatment Study of ADHD follow-up: changes in effectiveness and growth after the end of treatment. Pediatrics. 2004;113(4):762–769. doi:10.1542/peds.113.4.762 PubMed
53. Lisska MC, Rivkees SA. Daily methylphenidate use slows the growth of children: a community based study. J Pediatr Endocrinol Metab. 2003;16(5):711–718. doi:10.1515/JPEM.2003.16.5.711 PubMed
54. Swanson J, Greenhill L, Wigal T, et al. Stimulant-related reductions of growth rates in the PATS. J Am Acad Child Adolesc Psychiatry. 2006;45(11):1304–1313. doi:10.1097/01.chi.0000235075.25038.5a PubMed
55. Spencer TJ, Biederman J, Harding M, et al. Growth deficits in ADHD children revisited: evidence for disorder-associated growth delays? J Am Acad Child Adolesc Psychiatry. 1996;35(11):1460–1469. doi:10.1097/00004583-199611000-00014 PubMed
56. Faraone SV, Biederman J, Morley CP, et al. Effect of stimulants on height and weight: a review of the literature. J Am Acad Child Adolesc Psychiatry. 2008;47(9):994–1009. PubMed
57. Pidsosny IC, Virani A. Pediatric psychopharmacology update: psychostimulants and tics: past, present and future. J Can Acad Child Adolesc Psychiatry. 2006;15(2):84–86. PubMed
58. Law SF, Schachar RJ. Do typical clinical doses of methylphenidate cause tics in children treated for attention-deficit hyperactivity disorder? J Am Acad Child Adolesc Psychiatry. 1999;38(8):944–951. doi:10.1097/00004583-199908000-00009 PubMed
59. Palumbo D, Spencer T, Lynch J, et al. Emergence of tics in children with ADHD: impact of once-daily OROS methylphenidate therapy. J Child Adolesc Psychopharmacol. 2004;14(2):185–194. doi:10.1089/1044546041649138 PubMed
60. Biederman J, Lopez FA, Boellner SW, et al. A randomized, double-blind, placebo-controlled, parallel-group study of SLI381 (Adderall XR) in children with attention-deficit/hyperactivity disorder. Pediatrics. 2002;110(2, pt 1):258–266. doi:10.1542/peds.110.2.258 PubMed
61. Wolraich ML, Greenhill LL, Pelham W, et al. Randomized, controlled trial of oros methylphenidate once a day in children with attention-deficit/hyperactivity disorder. Pediatrics. 2001;108(4):883–892. doi:10.1542/peds.108.4.883 PubMed
62. Gadow KD, Sverd J, Sprafkin J, et al. Long-term methylphenidate therapy in children with comorbid attention-deficit hyperactivity disorder and chronic multiple tic disorder. Arch Gen Psychiatry. 1999;56(4):330–336. doi:10.1001/archpsyc.56.4.330 PubMed
63. Group TT; Tourette’s Syndrome Study Group. Treatment of ADHD in children with tics: a randomized controlled trial. Neurology. 2002;58(4):527–536. doi:10.1212/WNL.58.4.527 PubMed
64. Nolan EE, Gadow KD, Sprafkin J. Stimulant medication withdrawal during long-term therapy in children with comorbid attention-deficit hyperactivity disorder and chronic multiple tic disorder. Pediatrics. 1999;103(4, pt 1):730–737. doi:10.1542/peds.103.4.730 PubMed
65. Wilens TE, Prince JB, Spencer TJ, et al. Stimulants and sudden death: what is a physician to do? Pediatrics. 2006;118(3):1215–1219. doi:10.1542/peds.2006-0942 PubMed
66. US Food and Drug Administration. FDA issues safety communication about an ongoing review of stimulant medications used in children with ADHD. US Department of Health and Human Services Web site. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2009/ucm166616.htm. Updated January 14, 2010. Accessed January 8, 2013.
67. Gould MS, Walsh BT, Munfakh JL, et al. Sudden death and use of stimulant medications in youths. Am J Psychiatry. 2009;166(9):992–1001. doi: 10.1176/appi.ajp.2009.09040472 .
68. Hammerness PG, Perrin JM, Shelley-Abrahamson R, et al. Cardiovascular risk of stimulant treatment in pediatric attention-deficit/hyperactivity disorder: update and clinical recommendations. J Am Acad Child Adolesc Psychiatry. 2011;50(10):978–990. doi:10.1016/j.jaac.2011.07.018 PubMed
69. Cooper WO, Habel LA, Sox CM, et al. ADHD drugs and serious cardiovascular events in children and young adults. N Engl J Med. 2011;365(20):1896–1904. doi:10.1056/NEJMoa1110212 PubMed
70. Besag FM. Is suicidality serious? Curr Drug Saf. 2009;4(2):95–96. doi:10.2174/157488609788173053 PubMed
71. Perrin JM, Friedman RA, Knilans TK, et al; Section on Cardiology and Cardiac Surgery. Cardiovascular monitoring and stimulant drugs for attention-deficit/hyperactivity disorder. Pediatrics. 2008;122(2):451–453. doi:10.1542/peds.2008-1573 PubMed
72. Vetter VL, Elia J, Erickson C, et al. Cardiovascular monitoring of children and adolescents with heart disease receiving stimulant drugs: a Scientific Statement from the American Heart Association Council on Cardiovascular Disease in the Young Congenital Cardiac Defects Committee and the Council on Cardiovascular Nursing. Circulation. 2008;117(18):2407–2423. doi:10.1161/CIRCULATIONAHA.107.189473 PubMed
73. Mosholder AD, Gelperin K, Hammad TA, et al. Hallucinations and other psychotic symptoms associated with the use of attention-deficit/hyperactivity disorder drugs in children. Pediatrics. 2009;123(2):611–616. doi:10.1542/peds.2008-0185 PubMed
74. El-Zein RA, Abdel-Rahman SZ, Hay MJ, et al. Cytogenetic effects in children treated with methylphenidate. Cancer Lett. 2005;230(2):284–291. doi:10.1016/j.canlet.2005.01.003 PubMed
75. Dunnick JK, Hailey JR. Experimental studies on the long-term effects of methylphenidate hydrochloride. Toxicology. 1995;103(2):77–84. doi:10.1016/0300-483X(95)03109-S PubMed
76. Selby JV, Friedman GD, Fireman BH. Screening prescription drugs for possible carcinogenicity: eleven to fifteen years of follow-up. Cancer Res. 1989;49(20):5736–5747. PubMed
77. National Toxicology Program. US Department of Health and Human Services. Center for the Evaluation of Risks to Human Reproduction NTP-CERHR monograph on the potential human reproductive and developmental effects of methylphenidate. NIH Publication No 05-4473; 2005. http://ntp.niehs.nih.gov/ntp/ohat/stimulants/methylphenidate/MethylphenidateMonograph.pdf. Updated August 2005. Accessed October 29, 2012.
78. Ponsa I, Ramos-Quiroga JA, Ribasés M, et al. Absence of cytogenetic effects in children and adults with attention-deficit/hyperactivity disorder treated with methylphenidate. Mutat Res. 2009;666(1–2):44–49. doi:10.1016/j.mrfmmm.2009.03.014 PubMed
79. Wilens T, Faraone S, Biederman J, et al. Does stimulant therapy of ADHD beget later substance abuse: a metanalytic review of the literature. Pediatrics. 2003;111(1):179–185. doi:10.1542/peds.111.1.179 PubMed
80. Katusic SK, Barbaresi WJ, Colligan RC, et al. Psychostimulant treatment and risk for substance abuse among young adults with a history of attention-deficit/hyperactivity disorder: a population-based, birth cohort study. J Child Adolesc Psychopharmacol. 2005;15(5):764–776. doi:10.1089/cap.2005.15.764 PubMed
81. Wilens TE, Adamson J, Monuteaux MC, et al. Effect of prior stimulant treatment for attention-deficit/hyperactivity disorder on subsequent risk for cigarette smoking and alcohol and drug use disorders in adolescents. Arch Pediatr Adolesc Med. 2008;162(10):916–921. doi:10.1001/archpedi.162.10.916 PubMed
82. Biederman J, Monuteaux MC, Spencer T, et al. Stimulant therapy and risk for subsequent substance use disorders in male adults with ADHD: a naturalistic controlled 10-year follow-up study. Am J Psychiatry. 2008;165(5):597–603. doi:10.1176/appi.ajp.2007.07091486 PubMed
83. Mannuzza S, Klein RG, Truong NL, et al. Age of methylphenidate treatment initiation in children with ADHD and later substance abuse: prospective follow-up into adulthood. Am J Psychiatry. 2008;165(5):604–609. doi:10.1176/appi.ajp.2008.07091465 PubMed
84. Wilens TE, Adler LA, Adams J, et al. Misuse and diversion of stimulants prescribed for ADHD: a systematic review of the literature. J Am Acad Child Adolesc Psychiatry. 2008;47(1):21–31. doi:10.1097/chi.0b013e31815a56f1 PubMed
85. Wilens TE, Gignac M, Swezey A, et al. Characteristics of adolescents and young adults with ADHD who divert or misuse their prescribed medications. J Am Acad Child Adolesc Psychiatry. 2006;45(4):408–414. doi:10.1097/01.chi.0000199027.68828.b3 PubMed
86. McCabe SE, Knight JR, Teter CJ, et al. Non-medical use of prescription stimulants among US college students: prevalence and correlates from a national survey. Addiction. 2005;100(1):96–106. doi:10.1111/j.1360-0443.2005.00944.x PubMed
87. Teter CJ, McCabe SE, LaGrange K, et al. Illicit use of specific prescription stimulants among college students: prevalence, motives, and routes of administration. Pharmacotherapy. 2006;26(10):1501–1510. doi:10.1592/phco.26.10.1501 PubMed
88. McCabe SE, Teter CJ, Boyd CJ. The use, misuse and diversion of prescription stimulants among middle and high school students. Subst Use Misuse. 2004;39(7):1095–1116. doi:10.1081/JA-120038031 PubMed
89. Poulin C. From attention-deficit/hyperactivity disorder to medical stimulant use to the diversion of prescribed stimulants to non-medical stimulant use: connecting the dots. Addiction. 2007;102(5):740–751. doi:10.1111/j.1360-0443.2007.01758.x PubMed
90. Willoughby MT, Curran PJ, Costello EJ, et al. Implications of early versus late onset of attention-deficit/hyperactivity disorder symptoms. J Am Acad Child Adolesc Psychiatry. 2000;39(12):1512–1519. doi:10.1097/00004583-200012000-00013 PubMed
91. Nandagopal JJ, DelBello MP, Kowatch R. Pharmacologic treatment of pediatric bipolar disorder. Child Adolesc Psychiatr Clin N Am. 2009;18(2):455–469, x. doi:10.1016/j.chc.2008.11.004 PubMed
92. Joshi G, Wilens T. Comorbidity in pediatric bipolar disorder. Child Adolesc Psychiatr Clin N Am. 2009;18(2):291–319, vii–viii. doi:10.1016/j.chc.2008.12.005 PubMed
93. Wozniak J, Biederman J, Kiely K, et al. Mania-like symptoms suggestive of childhood-onset bipolar disorder in clinically referred children. J Am Acad Child Adolesc Psychiatry. 1995;34(7):867–876. doi:10.1097/00004583-199507000-00010 PubMed
94. Scheffer RE, Kowatch RA, Carmody T, et al. Randomized, placebo-controlled trial of mixed amphetamine salts for symptoms of comorbid ADHD in pediatric bipolar disorder after mood stabilization with divalproex sodium. Am J Psychiatry. 2005;162(1):58–64. doi:10.1176/appi.ajp.162.1.58 PubMed
95. Findling RL, Short EJ, McNamara NK, et al. Methylphenidate in the treatment of children and adolescents with bipolar disorder and attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(11):1445–1453. doi:10.1097/chi.0b013e31814b8d3b PubMed
96. DelBello MP, Soutullo CA, Hendricks W, et al. Prior stimulant treatment in adolescents with bipolar disorder: association with age at onset. Bipolar Disord. 2001;3(2):53–57. doi:10.1034/j.1399-5618.2001.030201.x PubMed
97. Quintana H, Birmaher B, Stedge D, et al. Use of methylphenidate in the treatment of children with autistic disorder. J Autism Dev Disord. 1995 Jun;25(3):283–294. PubMed
98. Handen BL, Johnson CR, Lubetsky M. Efficacy of methylphenidate among
children with autism and symptoms of attention-deficit hyperactivity disorder. J Autism Dev Disord. 2000 Jun;30(3):245–255. PubMed
99. Research Units on Pediatric Psychopharmacology Autism Network. Randomized, controlled, crossover trial of methylphenidate in pervasive developmental disorders with hyperactivity. Arch Gen Psychiatry. 2005 Nov;62(11):1266–1274. doi:10.1001/archpsyc.62.11.1266 PubMed
100. Posey DJ, Aman MG, McCracken JT, et al. Positive effects of methylphenidate on inattention and hyperactivity in pervasive developmental disorders: an analysis of secondary measures. Biol Psychiatry. 2007 Feb 15;61(4):538–544. doi:10.1016/j.biopsych.2006.09.028 PubMed
101. Stigler KA, Desmond LA, Posey DJ, et al. A naturalistic retrospective analysis of psychostimulants in pervasive developmental disorders. J Child Adolesc Psychopharmacol. 2004;14(1):49–56. doi:10.1089/104454604773840481 PubMed
102. Davies S, Heyman I, Goodman R. A population survey of mental health problems in children with epilepsy. Dev Med Child Neurol. 2003;45(5):292–295. doi:10.1111/j.1469-8749.2003.tb00398.x PubMed
103. Hermann B, Jones J, Dabbs K, et al. The frequency, complications and aetiology of ADHD in new onset paediatric epilepsy. Brain. 2007;130(pt 12):3135–3148. doi:10.1093/brain/awm227 PubMed
104. Physicians’ Desk Reference, 2012, 66th Edition. PDR Network: Montvale, NJ; 2011.
105. Baptista-Neto L, Dodds A, Rao S, et al. An expert opinion on methylphenidate treatment for attention deficit hyperactivity disorder in pediatric patients with epilepsy. Expert Opin Investig Drugs. 2008;17(1):77–84. doi:10.1517/13543784.17.1.77 PubMed
106. McAfee AT, Holdridge KC, Johannes CB, et al. The effect of pharmacotherapy for attention deficit hyperactivity disorder on risk of seizures in pediatric patients as assessed in an insurance claims database. Curr Drug Saf. 2008;3(2):123–131. doi:10.2174/157488608784529233 PubMed
107. Gonzalez-Heydrich J, Whitney J, Waber D, et al. Adaptive phase I study of OROS methylphenidate treatment of attention deficit hyperactivity disorder with epilepsy. Epilepsy Behav. 2010;18(3):229–237. doi:10.1016/j.yebeh.2010.02.022 PubMed
108. Daviss WB, Birmaher B, Diler RS, et al. Does pharmacotherapy for attention-deficit/hyperactivity disorder predict risk of later major depression? J Child Adolesc Psychopharmacol. 2008;18(3):257–264. doi:10.1089/cap.2007.0100 PubMed
109. Biederman J, Monuteaux MC, Spencer T, et al. Do stimulants protect against psychiatric disorders in youth with ADHD? a 10-year follow-up study. Pediatrics. 2009;124(1):71–78. doi:10.1542/peds.2008-3347 PubMed
LESSONS LEARNED AT THE INTERFACE OF MEDICINE AND PSYCHIATRY
The Psychiatric Consultation Service at Massachusetts General Hospital (MGH) sees medical and surgical inpatients with comorbid psychiatric symptoms and conditions. Such consultations require the integration of medical and psychiatric knowledge. During their twice-weekly rounds, Dr Stern and other members of the Consultation Service discuss the diagnosis and management of conditions confronted. These discussions have given rise to rounds reports that will prove useful for clinicians practicing at the interface of medicine and psychiatry.
Dr Stevens is an attending psychiatrist in Behavioral Health Services at Henry Ford Health Systems, Dearborn, and clinical assistant professor at Wayne State University, Detroit, Michigan; Dr Wilens is an attending psychiatrist in the Pediatric Psychopharmacology Clinic and codirector of the Center of Addiction Medicine at Massachusetts General Hospital, Boston, and associate professor of psychiatry at Harvard Medical School, Boston, Massachusetts; Dr Stern is chief of the Psychiatric Consultation Service at Massachusetts General Hospital, Boston, and a professor of psychiatry at Harvard Medical School, Boston, Massachusetts.
Dr Wilens has served as a consultant to Abbott, Eli Lilly, Euthymics, McNeil, Merck, National Institutes of Health, Novartis, and Shire; has received grant/research support from Abbott, Eli Lilly, McNeil, Merck, National Institutes of Health, National Institute on Drug Abuse, and Shire; has served as a speaker for Eli Lilly, McNeil, Novartis, and Shire; and has published a book (Straight Talk About Psychiatric Medications for Kids) with Guilford Press. Dr Stern is an employee of the Academy of Psychosomatic Medicine, has served on the speaker’s board of Reed Elsevier, is a stock shareholder in WiFiMD (Tablet PC), and has received royalties from Mosby/Elsevier and McGraw Hill. Dr Stevens reports no conflicts of interest related to the subject of this article.
Prim Care Companion CNS Disord
2013;15(2):doi:10.4088/PCC.12f01472
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: September 25, 2012; accepted October 22, 2012.
Published online: March 28, 2013.
Funding/support: None reported.
Corresponding author: Jonathan R. Stevens, MD, MPH, Henry Ford Health Systems, 5111 Auto Club Rd, Ste 112, Dearborn, MI 48126 ([email protected]).





