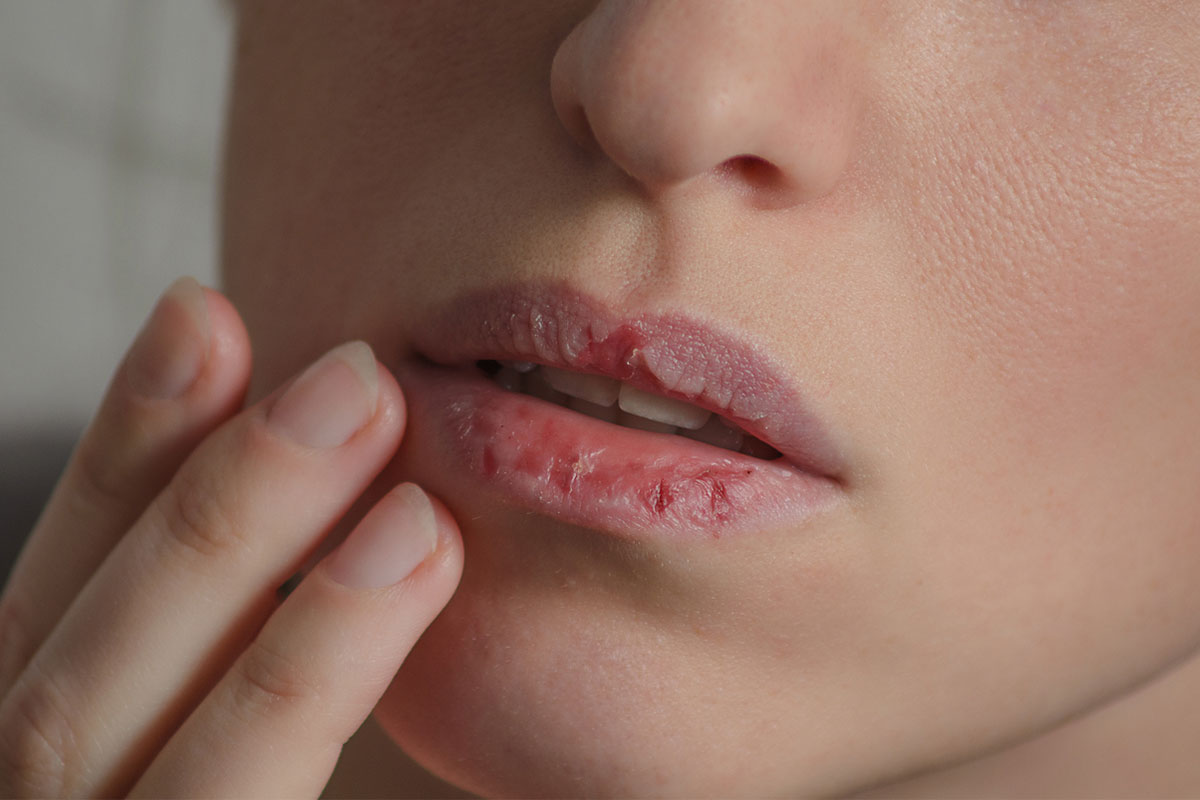
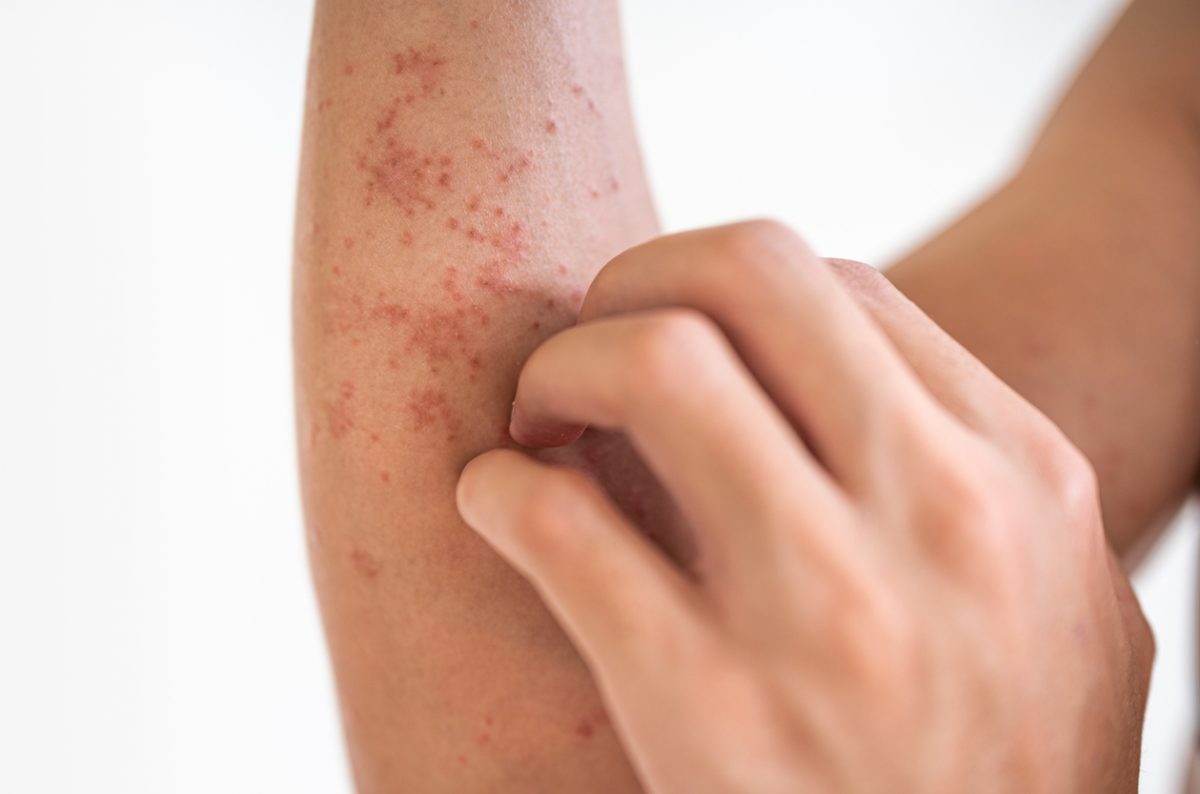
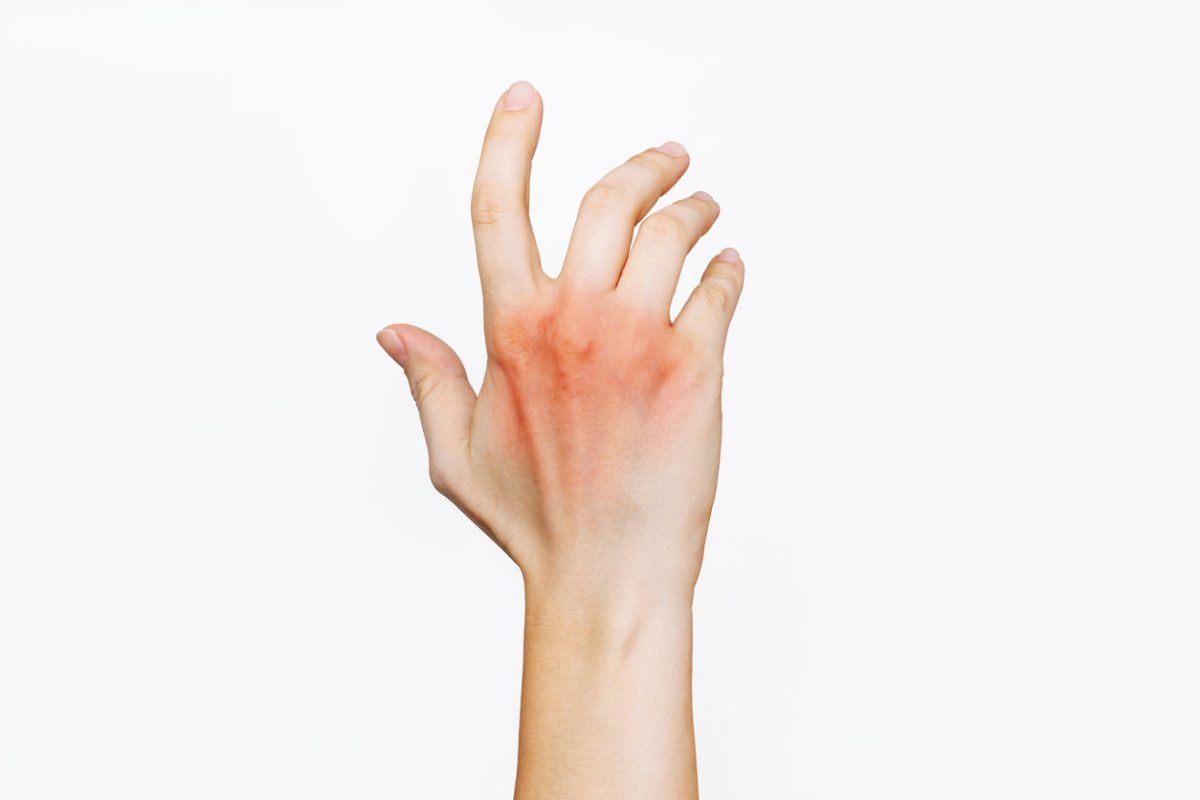
Dermatology, the study of skin and its disorders, has deep connections with mental health. Conditions like psoriasis, eczema, or acne can significantly impact an individual’s psychological well-being, leading to stress, anxiety, or depression. The Journal of Clinical Psychiatry explores this relationship, offering insights into the psychological effects of skin disorders and the role of stress in dermatological conditions. Our expertly curated content ensures that psychiatrists understand the bidirectional relationship between skin health and mental well-being, promoting comprehensive patient care.