Introduction/background: Zolpidem is a sedative-hypnotic widely prescribed in the United States. Recently, the US Food and Drug Administration (FDA) issued a drug safety communication regarding its dosing in women.
Objective: To compare compliance with FDA-approved dosing for zolpidem in women before and after a drug safety communication, and to evaluate compliance based on pharmacy location and prescriber type.
Method: This was a retrospective, observational cohort study. New prescriptions for Ambien, Ambien CR, Edluar, or Zolpimist or their respective generics dispensed from Kerr Drug pharmacies in North Carolina to women 18-64 years of age between April and September of 2012 ("before" cohort) or April and September of 2013 ("after" cohort) were included. χ2 tests were conducted to assess overall compliance, as well as compliance based on location (urban or rural) and prescriber type (physician or midlevel), with FDA-approved dosing for zolpidem. Trends in total prescription volume and total zolpidem prescription volume for all Kerr Drug pharmacies over the study period were also described.
Results: A total of 14,156 prescriptions for zolpidem were included in the primary analysis. Sixteen percent of prescriptions dispensed were in compliance with FDA recommendations following the FDA alert. A statistically significant increase was observed in compliance with FDA-approved dosing for zolpidem (odds ratio = 1.49; 95% CI, 1.35-1.65; P < .0001) postdrug safety communication. Significant increases in compliance were also observed in the post-FDA communication subgroups based on location and prescriber type, though no subgroup was found to be significantly more compliant than another.
Conclusions: The release of a drug safety communication by the FDA resulted in a statistically significant increase in proper dosing of zolpidem in women. Further research is needed in order to determine the impact of FDA alerts on prescribing patterns and the reasons for therapeutic substitution after such alerts.
This CME activity is expired. For more CME activities, visit CMEInstitute.com.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
CME Background
Articles are selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME articles on a variety of topics throughout each volume. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the article, correctly answer at least 70% of the questions in the Posttest, and complete the Evaluation.
CME Objective
After studying this article, you should be able to:
‘ ¢ Follow dosing recommendations for zolpidem
Accreditation Statement
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Creditâ„¢. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Note: The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Creditâ„¢ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 1.0 hour of Category I credit for completing this program.
Date of Original Release/Review
This educational activity is eligible for AMA PRA Category 1 Creditâ„¢ through April 30, 2017. The latest review of this material was February 2015.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Larry Culpepper, MD, MPH, Editor in Chief, has been a member of the speakers/advisory boards for AstraZeneca, Forest, Jazz, Lundbeck, Merck, Shire, Sunovion, and Takeda; has been a stock shareholder of M3 My Mood Monitor; and has received royalties from UpToDate and other financial or material support from Oxford University Press. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears with the article.
ABSTRACT
Introduction/background: Zolpidem is a sedative-hypnotic widely prescribed in the United States. Recently, the US Food and Drug Administration (FDA) issued a drug safety communication regarding its dosing in women.
Objective: To compare compliance with FDA-approved dosing for zolpidem in women before and after a drug safety communication, and to evaluate compliance based on pharmacy location and prescriber type.
Method: This was a retrospective, observational cohort study. New prescriptions for Ambien, Ambien CR, Edluar, or Zolpimist or their respective generics dispensed from Kerr Drug pharmacies in North Carolina to women 18-64 years of age between April and September of 2012 ("before" cohort) or April and September of 2013 ("after" cohort) were included. χ2 tests were conducted to assess overall compliance, as well as compliance based on location (urban or rural) and prescriber type (physician or midlevel), with FDA-approved dosing for zolpidem. Trends in total prescription volume and total zolpidem prescription volume for all Kerr Drug pharmacies over the study period were also described.
Results: A total of 14,156 prescriptions for zolpidem were included in the primary analysis. Sixteen percent of prescriptions dispensed were in compliance with FDA recommendations following the FDA alert. A statistically significant increase was observed in compliance with FDA-approved dosing for zolpidem (odds ratio = 1.49; 95% CI, 1.35-1.65; P < .0001) postdrug safety communication. Significant increases in compliance were also observed in the post-FDA communication subgroups based on location and prescriber type, though no subgroup was found to be significantly more compliant than another.
Conclusions: The release of a drug safety communication by the FDA resulted in a statistically significant increase in proper dosing of zolpidem in women. Further research is needed in order to determine the impact of FDA alerts on prescribing patterns and the reasons for therapeutic substitution after such alerts.
Prim Care Companion CNS Disord 2015;17(2):doi:10.4088/PCC.14m01728
© Copyright 2015 Physicians Postgraduate Press, Inc.
Submitted: October 2, 2014; accepted December 9, 2014.
Published online: March 5, 2015.
Corresponding author: Andrew J. Muzyk, PharmD, PO Box 3089-Pharmacy, Durham, NC 27710 ([email protected]).
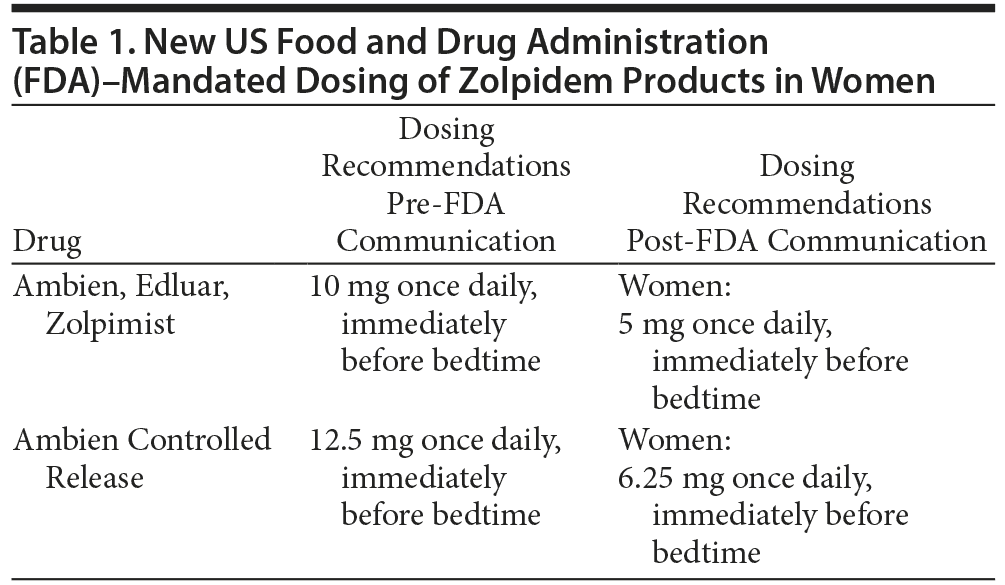
Immediate-release zolpidem was approved in 1992 and is now one of the most popular sleep medications in the United States, totaling 39.4 million prescriptions dispensed in 2011 alone.1 Originally, immediate-release zolpidem was dosed starting at 10 mg (12.5 mg controlled release) immediately before bedtime in adults. On January 10, 2013, the US Food and Drug Administration (FDA) released a drug safety communication regarding new data of elevated serum concentrations of zolpidem in nonelderly women taking doses equal to or greater than 10 mg.2 According to several pharmacokinetic studies cited in the report, women had peak serum concentration and area under the time-concentration curve that were 45% higher than men.2 In addition, women tended to eliminate zolpidem more slowly, resulting in an increased risk of next-day impairment when performing morning tasks.2 Studies evaluating driving performance after taking zolpidem products found that subjects with zolpidem serum concentrations > 50 ng/mL had significant impairment to increase the risk of a motor vehicle accident.2 Eight hours after a dose of immediate-release zolpidem, 15% of women and 3% of men had blood levels > 50 ng/mL. Such blood levels were observed at an even higher rate after administration of controlled-release zolpidem (33% of women and 25% of men, respectively).2
In response to these new data, the FDA warned patients and providers against the use of zolpidem doses exceeding 5 mg for immediate-release products or 6.25 mg for the extended-release product in female patients2 (Table 1). Providers were also advised to caution women about the risks of next-morning impairment in activities such as driving (despite perhaps feeling fully awake). Intermezzo, a zolpidem product, already contained different dosing recommendations for women than for men. The FDA approved a change in the package inserts for other manufacturers of zolpidem products (specifically Ambien, Edluar, Zolpimist, and Ambien Controlled Release) to change the dosing recommendations in their respective package inserts for women in May 2013 (see Table 1).3
While FDA communications are vital to health care practitioners, the effectiveness of these communications on prescriber behavior is not well studied. In particular, the impact of the zolpidem warning on dosing in the affected population has not been published. This study was designed to measure the impact of the original FDA drug safety communication regarding zolpidem in January 2013.
METHOD
This was an observational, retrospective cohort study. The primary objective was to compare compliance with FDA-approved dosing of zolpidem in women before and after the original FDA drug safety communication. Secondary objectives were to compare compliance with FDA-approved dosing of zolpidem in women before and after the original FDA drug safety communication based on whether the prescription was dispensed in an urban or rural area or whether the prescriber was a midlevel or physician-level practitioner. New prescriptions for zolpidem dispensed to women at Kerr Drug pharmacies in North Carolina from April 1, 2012, to September 30, 2012 ("before" cohort) and from April 1, 2013, to September 30, 2013 ("after" cohort) were included if they met the following criteria for the FDA drug safety communication: prescriptions for a zolpidem product dispensed to women aged 18-64 years of age. Prescriptions were excluded if the patient received Intermezzo at any time during the study period or if medical records were missing. In order to minimize confounding related to patients simply being dispensed a refill for an existing prescription, only new prescriptions were included. A prescription was identified as "new" if it was associated with a new prescription number. All duplicate prescription numbers correlated with a zolpidem prescription were removed.

- Dosing recommendations for zolpidem products in women have changed due to potential safety risks.
- Even after a drug safety communication released by the US Food and Drug Administration, most prescriptions for zolpidem dispensed to women were above the recommended dose.
- Clinicians can improve patient and public safety by educating patients on the risks associated with the use of zolpidem at doses higher than recommended.
All data were obtained from a central database at Kerr Drug corporate headquarters and blinded prior to being obtained by the investigators. Data obtained from each eligible prescription included the agent prescribed (including strength), the location at which it was dispensed, the date it was prescribed, and the Drug Enforcement Administration (DEA) number of the prescribing provider. City or town population data were obtained through the United States Census Bureau4 and were classified based on the 2010 United States Census Bureau definition for an urban versus rural area. Cities or towns were classified as "urban" if the population was ≥ 2,500 people or "rural" if the population was < 2,500 people.5 Prescribers were identified as midlevel if the DEA number obtained started with "M" or doctor level if the DEA number started with "A, B, F, or X."
In September 2013, Kerr Drug was acquired by Walgreens Pharmacy. As a result, percent change in total prescription volume and total zolpidem prescription volume were described in order to observe whether any changes in zolpidem dispensing may be a result of changing ownership. Each eligible prescription was categorized by time period and by compliance. The time periods were defined as predrug or postdrug safety communication. Subgroups included prescriptions dispensed in a rural or urban area and prescriptions prescribed by midlevel or physician prescribers. Criteria for compliance were prescriptions written for 5 mg of immediate-release zolpidem or 6.25 mg of extended-release zolpidem. Those prescriptions not meeting this definition of compliant were categorized as noncompliant.
The primary endpoint, percent compliance with FDA-approved dosing for new zolpidem prescriptions, was compared between the time periods using a χ2 test of homogeneity. For the secondary endpoints, compliance with FDA-approved dosing for new zolpidem prescriptions was described as percentage compliant for each of the subgroups: location (urban and rural) and prescriber type (midlevel prescriber or physician). Mantel-Haenszel χ2 tests were conducted to evaluate the association between time point (pre-FDA or post-FDA communication) and compliance, adjusting for location or prescriber type. SAS version 9.3 (SAS Institute, Cary, North Carolina) was used to perform all statistical inference. This study was approved as exempt by the Campbell University College of Pharmacy and Health Sciences Institutional Review Board, Buies Creek, North Carolina.
RESULTS
A total of 14,161 new prescriptions for zolpidem were dispensed to women at Kerr Drug pharmacies during the study time period. Five prescriptions were excluded from all analyses due to the patient also being dispensed a prescription for Intermezzo. Twelve prescriptions were excluded from the secondary analysis for prescriber due to missing prescriber records. Therefore, 14,156 prescriptions were available for the primary analysis and secondary analysis of location, while 14,144 prescriptions were included in the secondary analysis of prescriber (Figure 1).
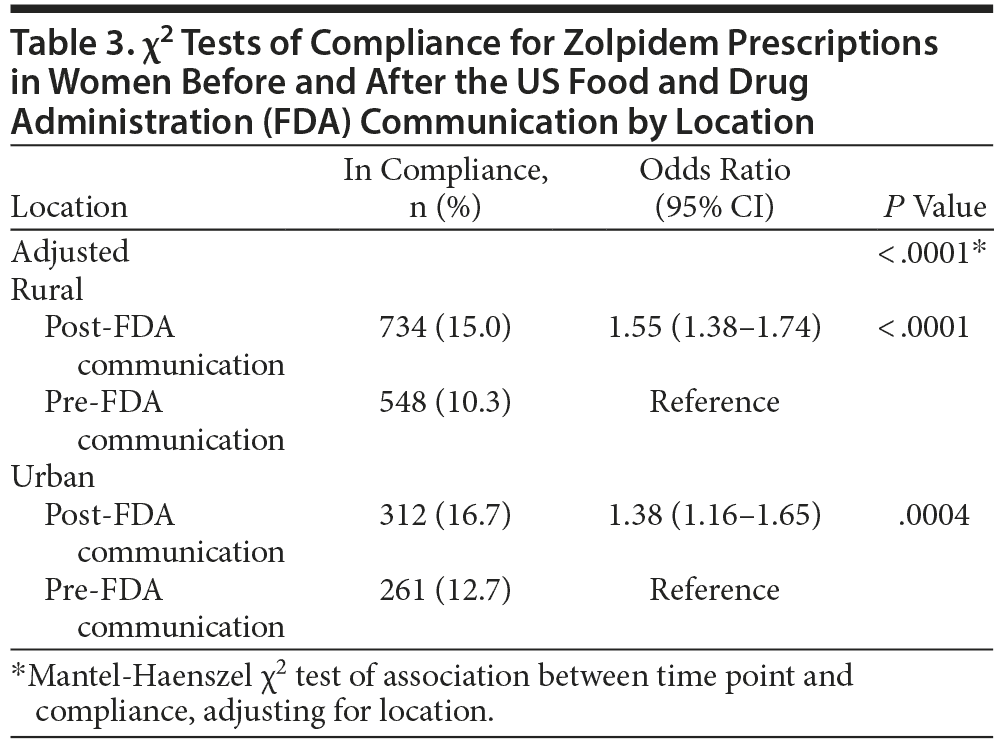
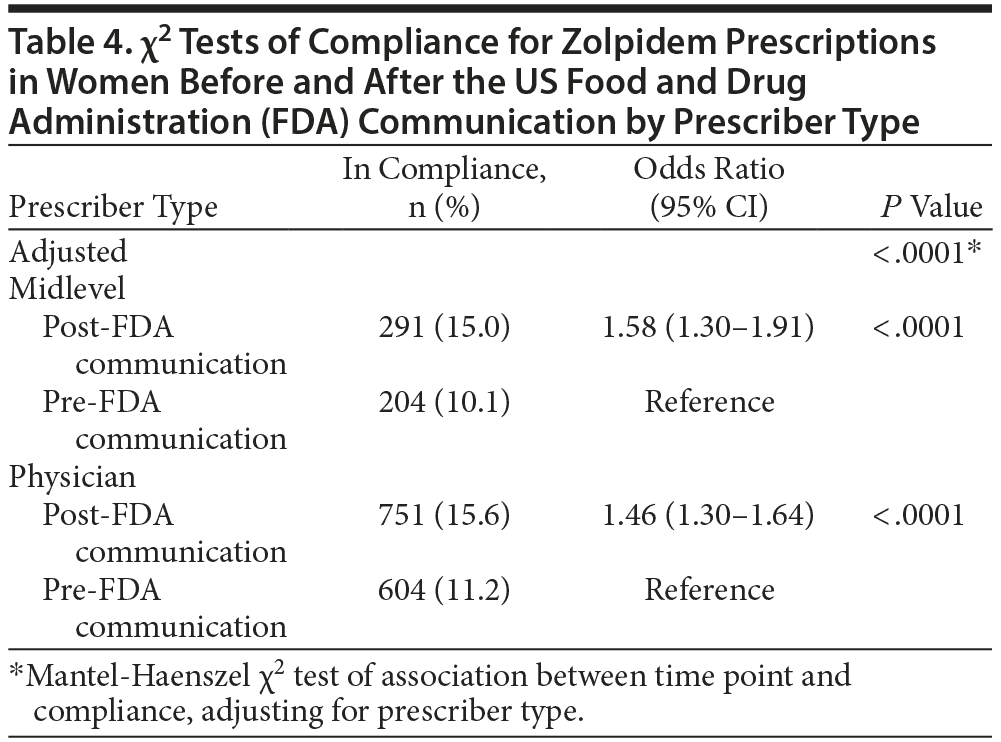
For the primary analysis, prescriptions dispensed to women after the drug safety communication were significantly more likely to be in compliance with FDA-approved dosing for zolpidem (15.5%) compared to those dispensed prescriptions prior to the communication (10.9%; odds ratio [OR] = 1.49; 95% CI, 1.35-1.65; P < .0001). Results for the primary endpoint are summarized in Table 2. When prescriptions were adjusted for location or prescriber type, compliance remained significantly higher in the post-FDA communication group. Tables 3 and 4 illustrate the change in the percent of prescriptions compliant with FDA-approved dosing for zolpidem based on location and prescriber type. In the location subgroups, compliance increased significantly in the post-FDA communication group for both urban (OR = 1.38; 95% CI, 1.16-1.65; P = .0004) and rural areas (OR = 1.55; 95% CI, 1.38-1.74; P < .0001) despite the compliance rates remaining < 17% in all groups. The overlap of the confidence intervals for urban and rural areas suggests that there was no statistically significant difference between the 2 groups with regard to compliance. Similar findings were observed in the prescriber subgroups. Compliance with FDA recommendations increased significantly among both midlevel prescribers (OR = 1.58; 95% CI, 1.30-1.91; P < .0001) and physicians (OR = 1.46; 95% CI, 1.30-1.64; P < .0001) in the post-FDA communication group compared to the pre-FDA communication group despite the compliance rates remaining less than 16% in all groups. As was found with the location groups, no statistically significant difference was observed between the 2 prescriber groups with regard to compliance. Total prescription volume for all prescriptions dispensed to all patients in Kerr Drug pharmacies decreased 3.77%, while total prescription volume for all zolpidem products dispensed to all patients in Kerr Drug pharmacies decreased 9.08%.
DISCUSSION
Prescriptions dispensed to nonelderly women were 49% more likely to be in compliance with FDA recommendations as a result of a drug safety communication. Research is lacking on how the response to FDA communications may vary among different groups of prescribers or how the location of the prescriber may affect his or her response. Results remained statistically significant, even after adjusting for these confounders. Compliance with the drug safety communication increased significantly among all subgroups analyzed and ranged from 38% in the urban group to 58% for midlevel prescribers. No significant difference in compliance was observed between any subgroup, indicating that the location in which a prescriber practiced did not have an effect on the response to this FDA alert, nor did the type of prescriber among midlevel prescribers and physicians overall. Future research may consider evaluating compliance among specific prescriber specialties as opposed to simply dividing all prescribers into the 2 broad categories of midlevel prescribers and physicians.
Following the FDA communication, there was an observed 1.4-fold decrease in the dispensing of zolpidem compared to total prescriptions dispensed from Kerr Drug pharmacies. This relative reduction in zolpidem prescriptions suggests that the decrease is less a result of a decrease in pharmacy business or changing ownership and more likely directly related to the drug safety communication. This finding is consistent with a similar study by Dusetzina et al,6 which reported that FDA communications targeting specific patient populations often spilled over into subpopulations for which the warnings were not intended. The authors noted that drug-specific communications often led to large decreases in utilization.6 A similar study by Hugman and Edwards7 found that, while mass "Dear Healthcare Professional" letters are an essential element of communicating drug safety information to the masses, more individualized communication methods targeted at providers may be more effective in conveying the true message of the drug safety communication without causing a decrease in prescribing of the targeted agent. Postulated reasons for the decrease in dispensing volume observed in our study include patient preference to discontinue the medication due to bad press, increased awareness of serious side effects associated with zolpidem use, or a misunderstanding of the intent of the drug safety communication. Therapeutic substitution could also be a factor contributing to the decrease in zolpidem dispensing observed in this study. However, our study was not designed to identify if the general decrease in zolpidem dispensing observed resulted in an increase in the dispensing of other agents used in the treatment of insomnia.
This study has several limitations worth noting. Although the FDA communication resulted in a 49% increase in compliance with FDA recommendations, only approximately 16% of prescriptions dispensed for zolpidem were in compliance. The primary endpoint measures prescription compliance, which may be influenced by individual prescribers’ biases. Though controlled-release zolpidem has been studied out to 6 months, zolpidem products are generally recommended only for short-term insomnia therapy (2 to 4 weeks) followed by reevaluation of the patient.8,9 In spite of the lack of data, zolpidem therapy is often initiated and never discontinued in patients suffering from insomnia symptoms. We included only new prescriptions in our analysis but could not account for updated orders for patients who had previously failed the indicated dose of 5 mg of zolpidem. Some prescribers instruct patients to split tablets for cost savings. Since directions for use were not obtained for each prescription, some prescriptions for zolpidem 10 mg may have actually been dosed correctly if the patient was pill-splitting. For the secondary endpoint of compliance based on location, location reflected from where the prescription was dispensed, not where the prescriber practiced. It is possible that some prescribers practicing in urban areas were included in our analysis with rural prescriptions due to where the prescription was dispensed and vice versa. Future research may consider analysis of a smaller sample of specific patients and providers, which includes all patient medication data in order to identify therapeutic changes in specific patients, and minimize some of the limitations of this study.
CONCLUSIONS
These prescriptions dispensed for zolpidem in this cohort of women demonstrated a significant increase in compliance with FDA dosing recommendations after a drug safety communication and was consistent among subgroups based on location and prescriber type. Further research is needed to better understand the clinical significance of such results.
Drug names: zolpidem (Ambien, Edluar, and others).
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this article.
Author affiliations: Department of Pharmacy Practice (Drs Clinard, Jiroutek, and Muzyk), Campbell University College of Pharmacy and Health Sciences (Dr Harward), Buies Creek, and Kerr Drug/Walgreens (Drs Lingerfeldt and Harward), Fuquay-Varina, North Carolina.
Financial disclosure: Drs Harward, Clinard, Jiroutek, Lingerfeldt, and Muzyk have no personal affiliations or financial relationships with any commercial interest to disclose relative to the article.
Funding/support: None reported.
Previous presentation: This research was presented at the American Pharmacists Association (APhA) Annual Meeting (via poster); March 27-31, 2014; Orlando, Florida, and at the 45th Southeastern Residency Conference; May 1-2, 2014; Athens, Georgia.
REFERENCES
1. IMS National Prescription Audit. Top 25 US Pharmaceutical Products by Dispensed Prescriptions. Danbury, CT: IMS Health; 2013.
2. Food and Drug Administration. FDA Drug Safety Communication: risk of next-morning impairment after use of insomnia drugs; FDA requires lower recommended doses for certain drugs containing zolpidem (Ambien, Ambien CR, Edluar, and Zolpimist). http://www.fda.gov/drugs/drugsafety/ucm334033.htm. Accessed September 4, 2013.
3. Food and Drug Administration. FDA Drug Safety Communication: FDA approves new label changes and dosing for zolpidem products and a new recommendation to avoid driving the day after using Ambien CR. http://www.fda.gov/Drugs/DrugSafety/ucm352085.htm. Accessed September 4, 2013.
4. United States Census Bureau. American FactFinder. Community facts. http://www.factfinder.census.gov/faces/nav/jsf/pages/index.xhtml. Accessed February 20, 2014.
5. United States Department of Health and Human Services. Defining the Rural Population. http://www.hrsa.gov/ruralhealth/policy/definition_of_rural.html. Accessed September 8, 2013.
6. Dusetzina SB, Higashi AS, Dorsey ER, et al. Impact of FDA drug risk communications on health care utilization and health behaviors: a systematic review. Med Care. 2012;50(6):466-478. PubMed doi:10.1097/MLR.0b013e318245a160
7. Hugman B, Edwards IR. The challenge of effectively communicating patient safety information. Expert Opin Drug Saf. 2006;5(4):495-499. PubMed doi:10.1517/14740338.5.4.495
8. Krystal AD, Erman M, Zammit GK, et al; ZOLONG Study Group. Long-term efficacy and safety of zolpidem extended-release 12.5 mg, administered 3 to 7 nights per week for 24 weeks, in patients with chronic primary insomnia: a 6-month, randomized, double-blind, placebo-controlled, parallel-group, multicenter study. Sleep. 2008;31(1):79-90. PubMed
9. Schutte-Rodin S, Broch L, Buysse D, et al. Clinical guideline for the evaluation and management of chronic insomnia in adults. J Clin Sleep Med. 2008;4(5):487-504. PubMed