Background: Self-compassion is a psychological skill associated with good mental health and adjustment to illness in the second half of life, but to date, few self-compassion-based interventions have been developed specifically for use in midlife and older adult cohorts. The purpose of this study was to develop and test the feasibility of a 4-week group self-compassion-based intervention designed to improve self-report and biological markers of well-being in midlife and older adult patients living with chronic illness.
Methods: Treatment development drew on existing literature, expert input, and qualitative interview data. Eight patients in outpatient treatment for a chronic illness were recruited from a rehabilitation hospital (during September and October 2017 and again during February and March 2018) to test feasibility. Participants attended a 1-hour group self-compassion-based intervention once per week for 4 weeks. Feasibility was assessed on 6 domains. Measures of well-being and heart rate variability (HRV), an index of nervous system functioning, were also collected.
Results: Recruitment was feasible and occurred within the expected time frame. Attendance at sessions was high (84.4%), with no dropouts. Participants found that the intervention was acceptable, rating sessions as enjoyable (6.8/7) and relevant to daily life (6.6/7). There were no adverse events. Secondary analysis revealed pre-post improvements for some well-being outcomes, such as a significant reduction in depressive symptoms (Hedges’ g = -1.18, 95% CI, -0.18 to -2.16).
Conclusions: A 4-session group self-compassion-based intervention was found to be feasible and acceptable to midlife and older adult patients in treatment for a chronic illness. A larger, randomized pilot trial is needed to explore the efficacy of this intervention.
Trial Registration: Australian New Zealand Clinical Trials Registry (ANZCTR) identifier: ACTRN12619000709145‘ ‹
ABSTRACT
Background: Self-compassion is a psychological skill associated with good mental health and adjustment to illness in the second half of life, but to date, few self-compassion-based interventions have been developed specifically for use in midlife and older adult cohorts. The purpose of this study was todevelop and test the feasibility of a 4-week group self-compassion-based intervention designed to improve self-report and biological markers of well-being in midlife and older adult patients living with chronic illness.
Methods: Treatment development drew on existing literature, expert input, and qualitative interview data. Eight patients in outpatient treatment for a chronic illness were recruited from a rehabilitation hospital (during September and October 2017and again during February and March 2018) to testfeasibility. Participants attended a 1-hour group self-compassion-based intervention once per week for 4 weeks. Feasibility was assessed on 6 domains. Measures of well-being and heart rate variability (HRV), an index of nervous systemfunctioning, were also collected.
Results: Recruitment was feasible and occurred within the expected time frame. Attendance at sessions was high (84.4%), with no dropouts. Participants found that the intervention was acceptable, rating sessions as enjoyable (6.8/7) and relevant to daily life (6.6/7). There were no adverse events. Secondary analysis revealed pre-post improvements for some well-being outcomes, such as a significant reduction in depressive symptoms (Hedges’ g = -1.18, 95% CI, -0.18 to -2.16).
Conclusions: A 4-session group self-compassion-based intervention was found to be feasible and acceptable to midlife and older adult patients in treatment for a chronic illness. A larger, randomized pilot trial is needed to explore the efficacy of this intervention.
Trial Registration: Australian New Zealand Clinical Trials Registry (ANZCTR) identifier: ACTRN12619000709145
Prim Care Companion CNS Disord 2019;21(5):19m02470
To cite: Brown L, Karmakar C, Flynn M, et al. A self-compassion group intervention for patients living with chronic medical illness: treatment development and feasibility study. Prim Care Companion CNS Disord. 2019;21(5):19m02470.
To share: https://doi.org/10.4088/PCC.19m02470
© Copyright 2019 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, Massachusetts General Hospital, Boston, Massachusetts
bDepartment of Psychiatry, Harvard Medical School, Boston, Massachusetts
cMelbourne School of Psychological Sciences, University of Melbourne, Melbourne, Victoria, Australia
dHealthscope Hospitals, Melbourne, Victoria, Australia
eMelbourne School of Engineering, University of Melbourne, Victoria, Australia
fSchool of Information Technology, Deakin University, Victoria, Australia
*Corresponding author: Lydia Brown, PhD, Melbourne School of Psychological Sciences, Redmond Barry Bldg, University of Melbourne, Victoria, 3010, Australia ([email protected]).
By the time individuals reach age 65 years, they will live with more than 2 chronic medical conditions on average,1 and this prevalence continues to increase with age. While chronic health issues are common, they are also a substantial risk factor for poor mental health and reduced quality of life.2,3 In turn, reductions in mental health in the context of illness can increase the risk of disability, poor treatment compliance, and mortality,3-5 which can further diminish the experience of aging. Given that chronic illness is often an inevitable part of growing older, development of interventions to support midlife and older adults to cultivate emotional health despite illness could have profound benefits for society.
Self-compassion, defined as being kind to oneself during moments of pain or suffering rather than being harshly self-critical,6 is a promising psychological skill that is relevant to midlife and older adult mental health.7-9 Individuals with high self-compassion typically have good mental health and are able to adjust to physical, emotional, and social changes and challenges, such that their mental health is relatively spared despite difficulties such as chronic illness.10 Unlike self-esteem, which can be threatened by stressors and typically declines in later adulthood,11 self-compassion becomes salient during difficulties12 and may potentially be increasingly relevant to well-being with age, enabling individuals to respond to the challenges of aging with kindness.13 Importantly, self-compassion is also a psychological skill that can be enhanced through training.14
Self-compassion-based interventions are a relatively new class of psychological intervention that train people how to be kinder toward themselves in their thinking, emotions, and daily life engagement with the world.14 Specifically, self-compassion-based interventions teach participants a healthy way to relate to hardship that is grounded in compassion, as opposed to self-criticism, nonacceptance, and frustration. Self-compassion-based interventions typically include formal mindfulness-based meditation practices wherein participants learn to self-generate positive emotional states directed toward the self, including loving-kindness, compassion, and gratitude. Concurrently, the interventions include practical self-compassion-based exercises that participants apply to their lives.
A growing body of evidence indicates that self-compassion-based interventions are effective at increasing self-compassion and related positive emotional states, while also reducing symptoms of psychopathology including depression, anxiety, and stress.14-16 Self-compassion is a skill that is relevant to populations both with and without mental illness.8,14 Thus, unlike traditional psychotherapies that are limited in scope to those with psychological symptoms, self-compassion-based interventions may make a unique contribution to enhance resilience in wide populations of aging adults.
A recent systematic review8 of self-compassionate aging identified that surprisingly little work has investigated self-compassion-based interventions in older adult groups. This review8 indicated that just 1 intervention,17 blending self-compassion and mindfulness, has been applied to older adults (mean age of 64 years). The authors17 found that the intervention was effective at improving resilience and reducing avoidance-related coping. This finding indicates that self-compassion training may enable aging adults to take a more proactive role in caring for their health. Indeed, another study15 of patients with diabetes (mean age of 44 years) found that an 8-week self-compassion intervention was associated with a significant reduction in mean blood glucose levels (HbA1C), as well as reduced depression and diabetes distress. Thus, self-compassion interventions may have the potential to simultaneously improve both mental and physical health, but work in this area is quite preliminary.
The studies by Perez-Blasco et al17 and Friis et al15 described previously were long interventions including 20 hours of contact time spread over at least 2 months. Patients in treatment for chronic illness typically have substantial stressors and life demands that make intensive interventions less feasible.18 Therefore, we aimed to develop a briefer intervention that could be broadly applicable to medical patients with competing demands.
Accordingly, we developed a brief, self-compassion-based intervention for midlife and older adults in outpatient treatment for a chronic physical illness. Drawing on preliminary work in older adults17 and a larger body of work in younger adults,14,15 we hypothesize that the intervention will be feasible and acceptable in the outpatient hospital setting. While this trial is not powered to test efficacy, our secondary aim was to explore effects of the intervention on mental health outcomes such as depressive symptoms, as well as an index of physiologic functioning: heart rate variability (HRV).
METHODS
Treatment Development
Intervention. The goal of the intervention was to teach participants skills to self-generate positive emotional states of compassion and kindness toward the self (primary) and others (secondary). Following the model developed by earlier self-compassion-based interventions,14,15,19 the course integrated formal meditation practice with group discussion on relevant themes and prescription of exercises to enable participants to apply skills discussed during the sessions to their daily lives between sessions.
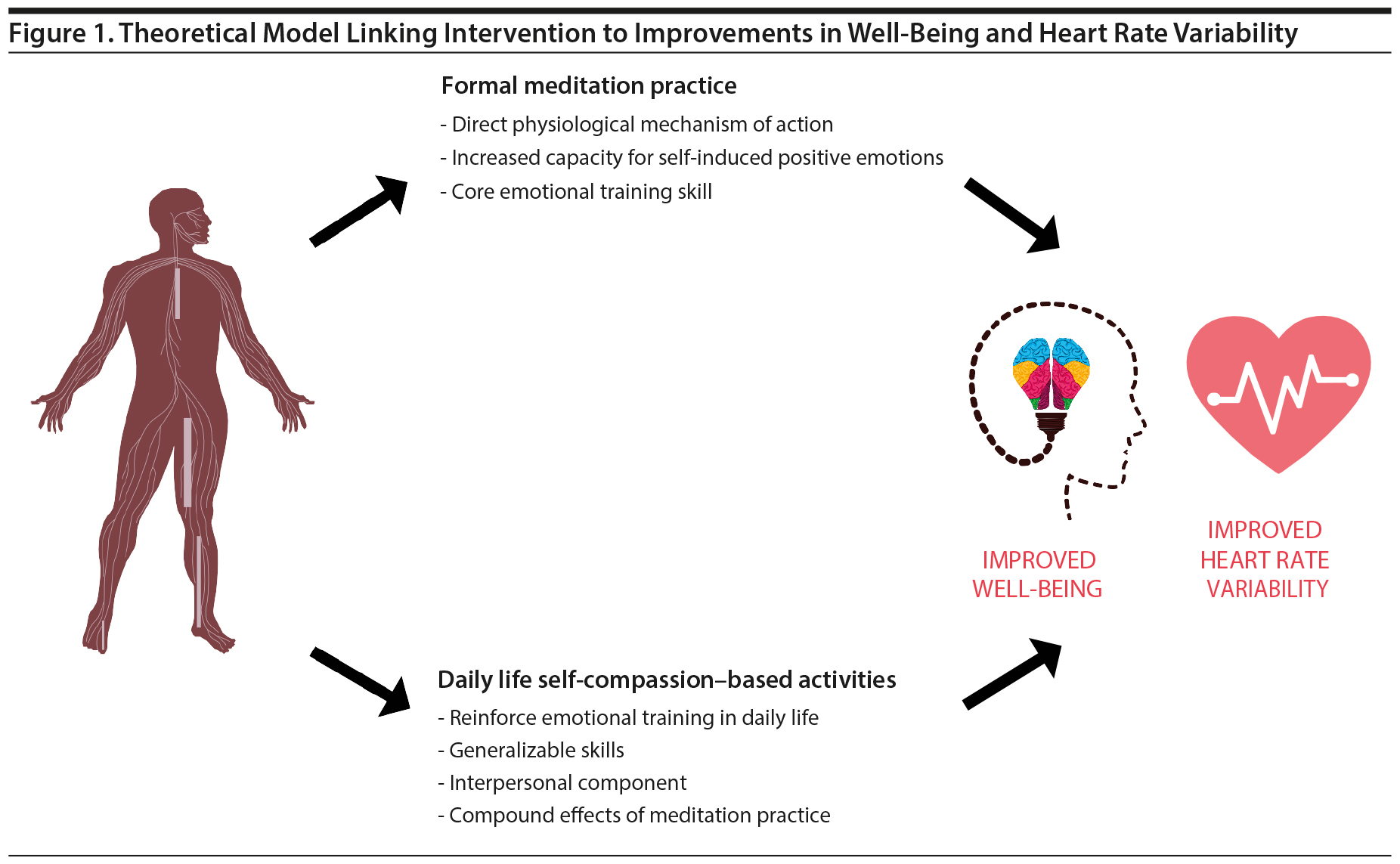
The concurrent focus on meditation practice and the prescription of daily life activities may enrich well-being outcomes more than the focus on either meditation or daily life activities alone. On the one hand, a growing body of evidence20 shows that meditation practice appears to have a direct, protective physiologic mechanism of action, downregulating sympathetic nervous system activity and facilitating physiologic balance. Meditation also enables participants to self-induce positive emotional states without the need for external hedonic cues from the environment, facilitating robust positive affective states not contingent on external life circumstances. This ability to induce positive emotional states may be especially relevant in the context of chronic illness, wherein access to pleasant and meaningful life activities may be limited.21
In addition to meditation training, simultaneously asking participants to apply self-compassion-based skills in daily life, including routine activities, self-talk, and altruistic acts, helps participants reinforce self-compassion into their daily life. Participants practice threading self-compassion into various domains of life, which may generalize and reinforce the positive emotional skills cultivated in formal meditation. A simple theoretical model of these pathways by which the intervention may improve well-being and potentially HRV, an index of nervous system functioning, is shown in Figure 1.
The course consisted of four 60-minute group sessions held weekly. This intervention was relatively short because qualitative research22 indicates that patients with medical illness juggle multiple medical appointments, and logistical issues related to engaging with a long intervention can be challenging alongside other medical needs. In addition to the weekly sessions, participants were provided resources to engage with course material between sessions including a website with meditation audio recordings and a resource kit with tangible cues and prompts relating to the course content (eg, a scented candle to practice mindfulness of the senses).

- Self-compassion-based interventions train people to be kind to themselves during life’s difficulties and thus may be useful to help medical patients cope with chronic illness.
- A brief, 4-session self-compassion-based intervention was feasible and acceptable to midlife and older adults in treatment for a chronic medical condition.
- The self-compassion-based intervention led to some improvements in mental health, including reduced depressive symptoms and increased gratitude.
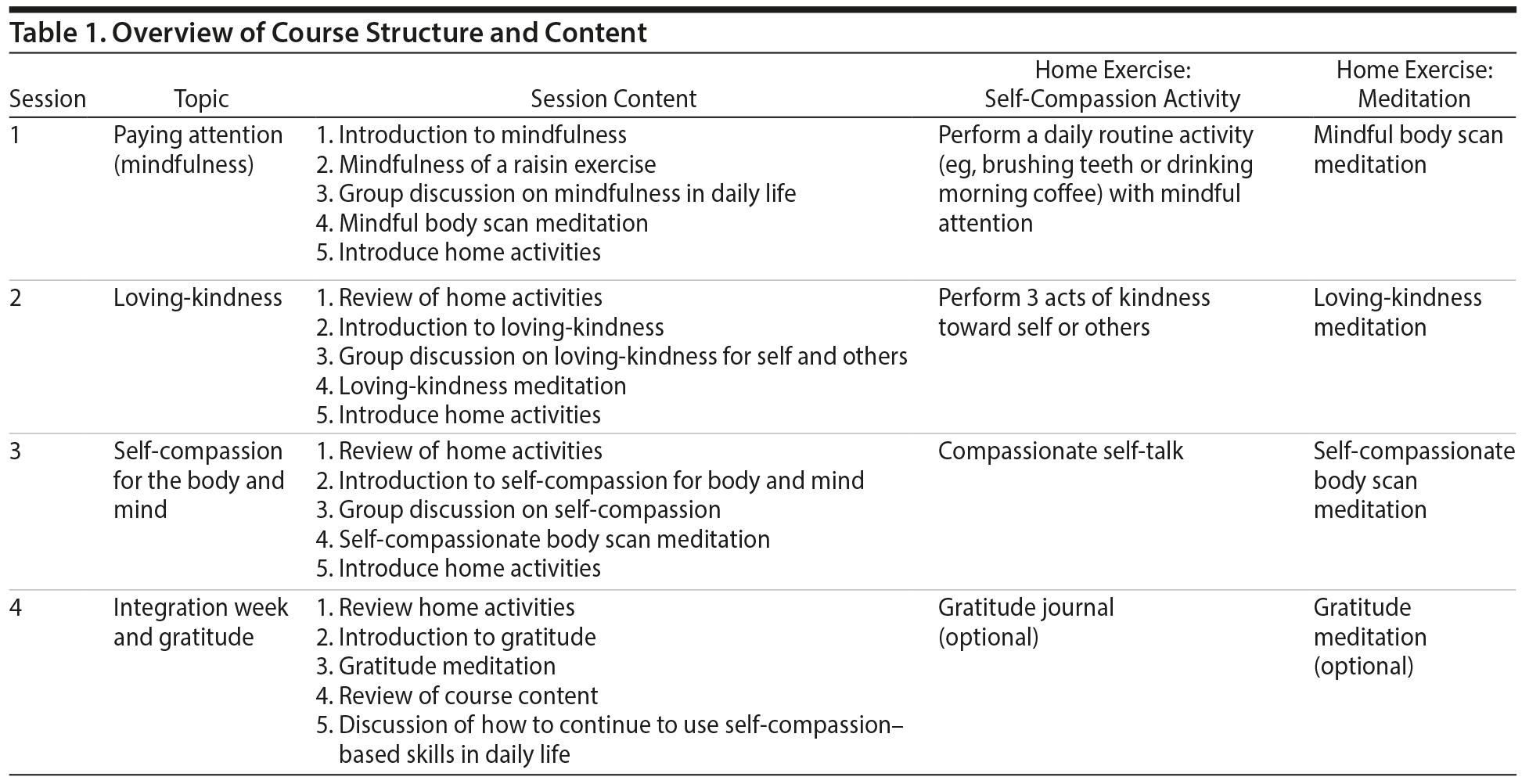
The overarching theme of the intervention was self-compassion. Each week explored a different facet of self-compassion including mindfulness (week 1), loving-kindness (week 2), self-compassion for the body and mind (week 3), and a concluding integration week + gratitude (week 4). Course facilitators (L.B. and M.F.) followed a study manual written by the authors to ensure that the intervention was delivered in a standardized manner. Mindfulness, defined as paying attention on purpose and nonjudgmentally to the present moment,23was chosen as a starting point for the intervention because it is a core component of self-compassion.6 Mindfulness is a foundation for self-compassion because it is necessary to be able to attend to suffering clearly (as opposed to avoiding or overidentifying with it) to be in a position to cultivate a kinder and healthier relationship to it. In this session, participants were instructed in paying attention to the 5 senses through a “mindfulness of a raisin” exercise, in which participants pay attention to the sight, touch, sound, smell, and taste of a raisin slowly and with present-moment awareness. Participants were briefly introduced to the science of mindfulness, and there was group discussion on mindfulness as it relates to both mental and physical health. Participants were also introduced to a 15-minute body scan mindfulness meditation and were asked to practice it daily over the next week using the course website. Between sessions, participants were also asked to bring mindfulness to a daily routine activity such as brushing teeth or drinking morning coffee.
Session 2 introduced loving-kindness. Loving-kindness is a core component of Neff and Germer’s Mindful Self-Compassion Program14 that involves self-generating a loving emotional state toward self and others, but it does not have an explicit focus on paying attention to suffering. To self-generate feelings of loving-kindness, participants were introduced to a loving-kindness meditation practice. There was also group discussion on benefits and barriers to loving-kindness and strategies to cultivate loving-kindness in daily life. Between sessions, participants were asked to complete a daily loving-kindness meditation practice and also to perform 3 acts of kindness toward self or others during the week.
Session 3 emphasized self-compassion for the body and mind. Self-compassion involves paying attention to symptoms of pain and responding with kindness and care. Participants were introduced to the self-compassionate body scan and were asked to notice symptoms of pain or discomfort with a mental attitude of compassion. There was also group discussion around self-compassionate thinking. Participants were invited to compare their inner dialogue with the way they treat others (to whom are they kinder and why). Discussions included curbing the inner critic, cultivating a more compassionate inner dialogue, and the importance of self-compassionate action for physical health management (ie, prioritizing one’s own needs, such as the need for regular exercise, alongside potentially competing demands from family or others). Between sessions, participants were asked to continue with a daily self-compassionate body scan meditation and to pay more attention to their inner dialogue (choosing more self-compassionate self-talk in daily life).
The fourth and final session involved a summary and integration of the content previously described. Participants were invited to reflect on the self-compassion-related skills they had acquired during the course and explore how those skills applied to their daily life. Participants were also invited to reflect on gratitude (such as past personal successes for which they are grateful). In group discussion, participants were also invited to explore how they might continue to apply self-compassion and associated positive emotions to their daily lives following completion of the intervention. A summary of the intervention structure and content is presented in Table 1.
Feasibility Study
Design. This study was a single-arm, pre/post intervention design. The intervention was delivered twice over a 6-month period. Ethics approval was obtained from Healthscope Hospital’s Melbourne Clinic Ethics Board (#285). The study was registered in the Australian New Zealand Clinical Trials Registry (identifier: ACTRN12619000709145).
Participants and recruitment. Participants were recruited from a private rehabilitation center in metropolitan Melbourne, Australia (Healthscope North Eastern Rehabilitation Centre [NERC]) during September and October 2017 and again during February and March 2018. We aimed to recruit a total of 8 participants, which is adequate for a feasibility study.24 Eligibility criteria included (1) being a NERC patient currently in treatment for a chronic medical condition, (2) age ≥ 40 years, and (3) permission to participate from the treating physician. Exclusion criteria included (1) a lack of conversational English, (2) presence of psychotic symptoms, and (3) a diagnosis of cognitive decline based on discussion with the treatment team.
A member of the research team attended NERC’s cardiac rehabilitation program to briefly inform cardiac patients about the study on 4 occasions (talking to approximately 24 cardiac patients in total). Presentations were also made at NERC’s joint replacement group (talking to approximately 18 patients). The rehabilitation allied health treatment team was informed of the study and were invited to recommend patients in treatment for a chronic condition who they thought might be interested in participating in the study. Flyers for the study were also placed in the NERC lobby.
Prospective participants were informed about the study and provided a copy of the plain language statement. Those who wished to participate and met the inclusion criteria gave written informed consent. Written consent was also obtained from each participant’s treating physician. Participants were reimbursed travel costs for their participation in the study.
Primary Outcome: Feasibility
Following recommended protocol,25 the feasibility of the intervention was determined on 6 a priori-defined dimensions: recruitment, adherence, acceptability, attrition, safety, and data collection.
Recruitment. Data were collected on the time taken to recruit and the proportion of those interested who met eligibility criteria to participate. A comparable feasibility study of tango dancing for patients with Parkinson disease recruited 3 participants per month over 2 months.26 Given our broader inclusion criteria, we expected a recruitment rate of approximately 4 participants per month over 2 months, resulting in a total sample size of approximately 8 participants.
Adherence. Session attendance rates between 60% and 70% have been recorded in prior health and well-being interventions for midlife and older adults.27,28 A session attendance rate of at least 65% (ie, that participants attend a mean of 2.6 of 4 sessions) was expected to indicate feasibility of adherence.
Acceptability. Participants were asked to indicate their course satisfaction on 4 dimensions relating to enjoyment, relevance to daily life, engagement with home practice, and usefulness of mindfulness in evoking calm on a scale from 1 (least satisfied) to 7 (most satisfied). We determined that a satisfaction rating of at least 5 of 7 would indicate acceptability of the intervention. Written qualitative feedback from participants was also collected at the conclusion of the intervention.
Attrition. We defined an attrition rate ≤ 15% as indicating feasibility, given this rate has been used in comparable research of older adults with physical conditions.26
Safety. Any adverse psychological or physical symptoms were documented at each session, with medical follow-up provided as needed to ensure participant safety.
Data collection. We assessed the practicality of including electrocardiogram (ECG) measurements in the study, alongside conventional self-reported measures of psychological functioning. We expected that at least 80% of data collected would be fit for analysis. A reason to exclude data could be the presence of irregular heartbeats. We also assessed the practicality of asking participants to complete baseline self-reported measures at home, which would be returned prior to starting the intervention.
Secondary Outcomes: Pre-Post Change
Center for Epidemiologic Studies Depression Scale (CES-D). The CES-D29 is a widely used 20-item measure of depression. Depressive symptoms are rated on a severity scale ranging from 0 (rarely or none of the time) to 3 (most or almost all the time). A score ≥ 16 is indicative of high depressive symptoms and the probable presence of a depressive disorder.30 The CES-D has good psychometric properties, and the Cronbach’s α was 0.86 in this study.
Self-Compassion Scale (SCS). The 26-item SCS6 measures self-compassion along 3 interrelated dichotomies: mindfulness versus overidentification, self-kindness versus self-criticism, and common humanity versus a sense of overidentification. Cronbach’s α for the SCS was 0.78.
Positive and Negative Affect Schedule (PANAS). The 10-item positive affect subscale of the PANAS31 was used to measure positive affect. Participants rate the degree to which they experienced positive emotions (such as “interested” and “enthusiastic” during the past week). Items are rated on a 5-point Likert scale ranging from 1 (slightly or not at all) to 5 (extremely). The PANAS had good reliability in this study (α = 0.84).
Satisfaction With Life Scale (SWL). The SWL32 is a popular 5-item measure of global satisfaction with life validated for use in midlife and older adults. The Cronbach’s α in this study was 0.77.
Gratitude Questionnaire (GQ-6). The GQ-633 is a validated 6-item measure of gratitude. Participants rate the degree to which they respond to questions such as “I have so much in life for which to be thankful” on a 7-point scale ranging from 1 (strongly disagree) to 7 (strongly agree). Reliability for the scale was α = 0.76.
HRV. HRV, defined as the beat-to-beat variance in resting heart rate, is a marker for both autonomic nervous system functioning and heart health.34 High HRV is thought to indicate good parasympathetic modulation of stressors, and it is predictive of meaningful physical health outcomes such as reduced risk of cardiac events, all-cause mortality, and depression.35-38 HRV has been recommended for use as a primary outcome measure in compassion-based interventions.39 In this study, HRV was measured via 3-lead ECG at rest for a 10-minute recording using the US Food and Drug Administration-approved BIOPAC MP150 measurement system containing a BioNomadix Logger and transceiver (BIOPAC Systems, Inc, Goleta, California). Data on the standard deviation of NN intervals were extracted, and high-frequency HRV (0.15-0.4 Hz) and low-frequency HRV (0.04-0.15 Hz) were derived using the Fourier transform. Further details on HRV data collection and data cleaning are found in Supplementary Appendix 1.
Procedure
At enrollment, participants were given a hardcopy of self-reported measures to self-complete at home and were instructed to bring them to the first session. An appointment was made to collect baseline HRV data prior to starting the course. HRV was recorded in a relaxed, seated posture. Participants were not instructed to control eye movements or breathing rate during the recording. Participants completed these measurements again at the conclusion of the fourth session.
Statistical Analyses
Feasibility was determined based on the 6 feasibility criteria recommended by Moore and colleagues.25 To assess feasibility, we compared quantitative data obtained from the study (eg, duration of time needed for recruitment, mean enjoyment and usefulness ratings of each session, and attrition rates) to the a priori-determined criteria.
The Reliable Change Index40 was used to calculate significant and reliable improvement, nonimprovement, or deterioration in depressive symptoms during the intervention. We also investigated clinically meaningful change in depressive symptoms using the CES-D clinical cutoff of 16 to determine if postintervention depression scores reduced to the subclinical range.
To estimate effect size of pre-post changes in secondary outcomes, Hedges’ g and its 95% confidence interval (CI),was considered for each outcome. Hedges’ g is a variation of Cohen d that corrects for upwards bias.41 Hedges’ g estimates the standardized change (in standard deviation units) in scores between timepoints. Following standard protocol, we took Hedges’ g values of 0.2, 0.5, and 0.8 to indicate a small, medium, and large effect, respectively.42 Effect size analyses were conducted using SPSS (IBM Corporation, Armonk, New York) and XECI software (Dr Paul Dudgeon, Melbourne School of Psychological Sciences, Parkville, Victoria, Australia) packages. Reported P values are derived from paired-sample t tests.
Feasibility studies are typically underpowered to detect statistically significant changes in outcomes.25 Using Hedges’ g results, a power analysis was conducted to determine the sample size that would be needed to detect a significant effect with 80% statistical power in a larger follow-up trial. Power analyses were conducted using GPower. Test-retest correlation coefficients for well-being outcome variables can be found in Supplementary Appendix 2.
RESULTS
Demographics
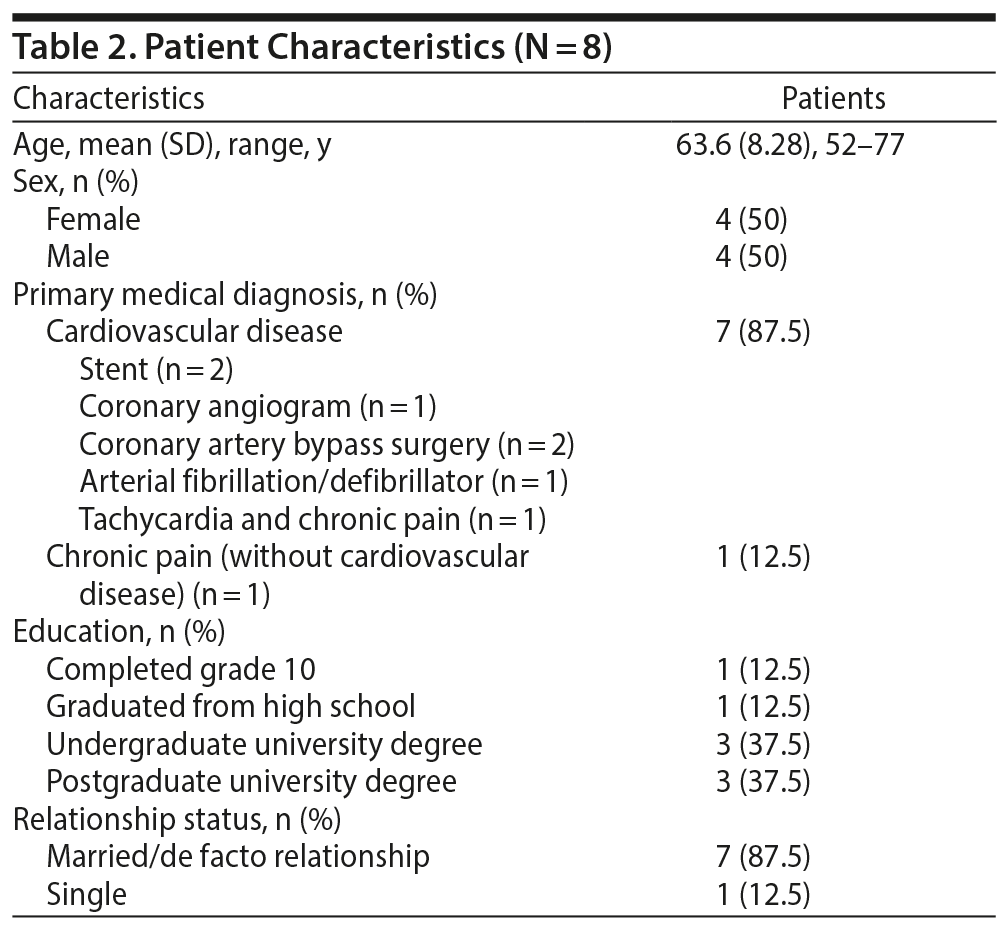
Patient characteristics are presented in Table 2. This study included an equal number of male (n = 4) and female (n = 4) participants, and the majority (75%) of the sample was university educated.
Primary Outcome: Feasibility
Recruitment. A total of 11 participants expressed interest in the study, and all met study inclusion criteria. Eight were recruited and enrolled in this study. Reasons for not participating were travel commitments (n = 2) and declining health (n = 1). The majority (n = 6) of participants were recruited based on the presentations made at the hospital’s cardiac rehabilitation program, resulting in a response rate from this source of 25%. No participants responded to presentations made in the joint replacement group. The remaining 2 participants were recruited via referral from their allied health treatment team. No participants self-referred through responding to the study flyers.
Recruitment commenced in September 2017. Over a period of 6 weeks, 5 participants were recruited for the study, and these participants formed the first group (run in October-November 2017). Recruitment recommenced in late February 2018. Three participants were recruited over a period of 4 weeks, forming the second group (run in March-April 2018). A total of 10 weeks of recruitment was required to enroll 8 participants, which is only slightly longer than anticipated.
Adherence. The adherence rate was 84.4%, and participants attended a mean of 3.4 of the 4 sessions. The reasons that participants missed sessions included illness (1 session), work commitments (2 sessions), a planned vacation (1 session), and forgetting (1 session). In addition to good attendance of group sessions, participants also verbally reported that they regularly practiced assigned home activities between sessions.
Acceptability. The intervention was well received by the participants. They rated the sessions as highly enjoyable (mean rating: 6.8/7). The meditation-based practices were found to be calming (mean rating: 6.6/7), and participants felt that the sessions were relevant to their everyday lives (mean rating: 6.6/7). Motivation to practice home-based activities had a mean rating of 5.4/7.
In the open-ended question for feedback on the course, 3 participants suggested that sessions should be longer to allow more time to cover the course content. One participant suggested including a list of practical strategies to improve mood in daily life, and 1 participant suggested more clarity about the likely time commitment required for between-session home-based activities. Participants reported enjoying the opportunities for group discussion, to share experiences of how they were applying topics discussed in the sessions to their daily lives, and to support health behaviors and symptom management of chronic illness (eg, mindful preparation of a healthy breakfast, using mindfulness to manage sleep disturbance associated with chronic pain).
Attrition. No participants dropped out of the study after enrolling.
Safety. No adverse psychological or physical events occurred during the study.
Data collection. HRV data from 1 participant were excluded due to an insufficient number (< 50%) of regular heart beats. One participant failed to return baseline self-report measures, thus self-reported data are excluded for this case.
Secondary Outcome: Pre-Post Change
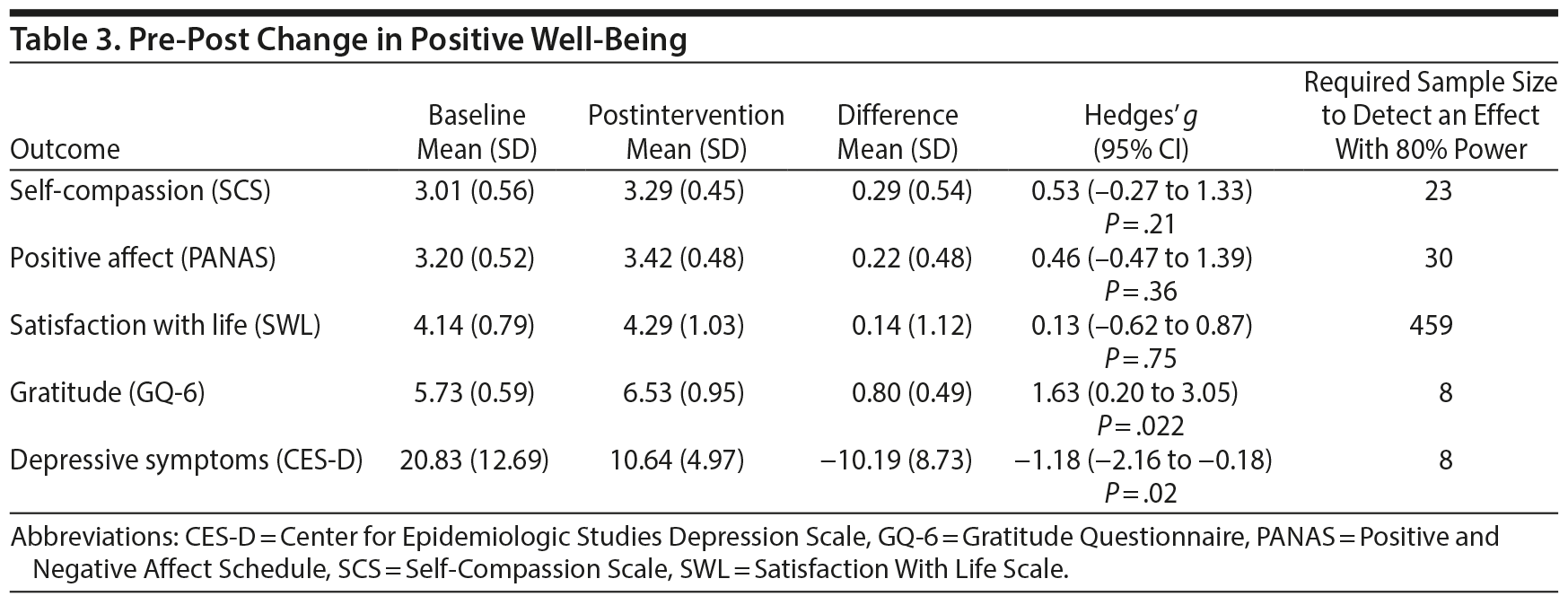
Mental health: depressive symptoms. The intervention was associated with a large and significant reduction in symptoms of depression (Hedges’ g = -1.18; 95% CI, -0.18 to -2.16; P = .02; Table 3). Four of 7 participants had clinically significant levels of depression (CES-D score > 16) at baseline, and all 4 of these participants dropped to subclinical levels postintervention. Each of these participants also experienced reliable change, with Reliable Change Index scores all > 2. Other participants who had subclinical depression at baseline experienced no reliable change in depressive symptoms postintervention.
Mental health: positive well-being. The intervention was associated with a statistically significant increase in gratitude (Hedges’ g = 1.63; 95% CI, 0.20-3.05; P = .02; Table 3). There were nonsignificant trends toward moderate increases in self-compassion (g = 0.53) and positive affect (g = 0.46). A power calculation revealed that the required sample sizes to detect significant change in these outcomes were 23 (self-compassion) and 30 (positive affect). The intervention did not appear to have a meaningful impact on the cognitive aspect of hedonic well-being measured by satisfaction with life (Hedges’ g = 0.13, P = .75).
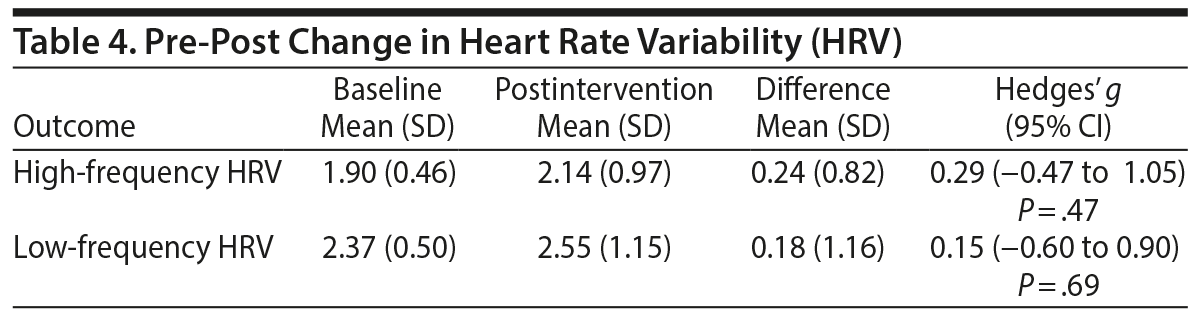
HRV. The intervention was associated with a nonsignificant increase in high-frequency HRV (Hedges’ g = 0.29, small effect, Table 4). The sample size needed to detect significant change in high-frequency HRV with 80% power and a 2-tailed α set at 0.05 is 96. There was only a trivial trend to increased low-frequency HRV (Hedges’ g = 0.15).
DISCUSSION
This self-compassion-based group intervention was found to be feasible as an adjunct treatment for midlife and older outpatients with a chronic medical condition. Participants found the sessions to be enjoyable (6.8/7) and relevant to their daily lives. Adherence to the group was high (84.4%), and there were no dropouts from the study. Recruitment occurred within the expected time frame, and there were no adverse events.
A small but growing number of studies have demonstrated that self-compassion-based interventions are both acceptable and useful to younger adults.14-16 Here, we contribute to the literature on self-compassion-based interventions by demonstrating that such interventions also appear to be acceptable to patients in a health care setting and for midlife and older adults in treatment for chronic illness. To date, only 1 study17 has investigated a self-compassion-based intervention in older adults, but this intervention was long (20 contact hours over 10 weeks), and the authors did not measure self-compassion. While our study was underpowered to detect statistically significant changes in self-compassion, we found that participants increased in self-compassion by a mean of 0.53 standard deviation units. This effect size is smaller than that reported by Neff and Germer14 in an 8-week self-compassion training program delivered to healthy subjects (Hedges’ g = 1.67), indicating a possibility that length of intervention may moderate effect sizes. Interestingly, however, our intervention was associated with larger improvements in depressive symptoms (Hedges’ g = 1.18), adding weight to the idea that self-compassion may help ameliorate distress—perhaps especially in the context of medical illness.
While participants found the sessions to be enjoyable and relevant to their daily lives, motivation to practice exercises between sessions was slightly lower (5.4/7), with participants identifying competing family, occupational, and health care demands as a key barrier to practice. In this intervention, prescribed guided meditations were 15 minutes in length given that this duration has been found to induce meaningful improvements in affect and willingness to tolerate negative experiences.43 Signed self-contracts are often used in traditional psychotherapies such as cognitive-behavioral therapy as a form of motivation to promote behavior change during treatment.44 A larger trial could integrate self-contracts, wherein participants make a commitment to themselves to practice the home activities during the course of the intervention as a means to enhance motivation for home practice during the intervention.
This study has a number of limitations. First, we did not include a control condition. Especially for patients who have recently experienced a health event (eg, surgery prior to cardiac rehabilitation), psychological symptoms may be related to the health event and resolve naturally with the course of time.45,46 Thus, a comparison group is needed in a future trial to control for natural changes in well-being with time. Second, given that one participant failed to return the baseline self-reported measures, scheduling face-to-face or telephone appointments to collect self-reported data could also improve the quality of a future trial, as would inclusion of a quantitative measure of homework completion for participants to complete each week during the intervention. Third, this study includes a small sample size of 8 participants. Effect size estimates are known to be unstable in small samples, and results from small pilot studies are not always replicated in larger controlled trials,47 warranting the need for a larger pilot randomized controlled trial. Fourth, most participants were recruited from a cardiac rehabilitation program, thus the generalizability of the intervention to other clinical groups remains to be tested. Finally, our study participants were highly educated, with 6 of 8 participants being university educated. Application of the intervention in diverse groups with lower levels of education and recruitment from multiple sites would be a helpful extension of this study.
This study demonstrates that a brief, 4-session, self-compassion-based group intervention is acceptable to patients in outpatient treatment for chronic medical conditions. Group-based interventions can be more cost-effective than individual psychological therapy,48 and a group setting has the added advantage of facilitating social support among patients managing chronic illness. The provision of this intervention in a medical health setting may facilitate holistic health care, supporting patients to maintain mental health during illness. Accordingly, this study indicates that self-compassion-based interventions appear to be acceptable to midlife and older patients in medical treatment for a chronic health issue. A randomized, controlled pilot trial would be beneficial to explore the efficacy of this intervention in improving indicators of mental health.
Submitted: April 15, 2019; accepted July 1, 2019.
Published online: September 26, 2019.
Potential conflicts of interest: Dr Celano has received honoraria for talks to Sunovion Pharmaceuticals on topics unrelated to this research. Drs Brown, Karmakar, Palaniswami, Huffman, and Bryant; Ms Flynn; and Mr Motin report no conflicts of interest related to the subject of this article.
Funding/support: This work was supported by an Endeavour postdoctoral fellowship (Dr Brown) and a Hallmark Aging Research Initiative grant awarded by The University of Melbourne (Dr Bryant). Time for editing and analysis was supported by the National Heart, Lung, and Blood Institute through grant K23HL123607 (Dr Celano).
Role of the sponsor: The supporters had no role in the design, analysis, interpretation, or publication of this study.
Acknowledgments: The authors thank Nicholas Blandford (health care consumer, Canberra, Australian Capital Territory, Australia) for his helpful feedback during the development of the study manual.
Supplementary material: See accompanying pages.
REFERENCES
1.Barnett K, Mercer SW, Norbury M, et al. Epidemiology of multimorbidity and implications for health care, research, and medical education: a cross-sectional study. Lancet. 2012;380(9836):37-43. PubMed CrossRef
2.Bair MJ, Robinson RL, Katon W, et al. Depression and pain comorbidity: a literature review. Arch Intern Med. 2003;163(20):2433-2445. PubMed CrossRef
3.Carney RM, Freedland KE. Depression and coronary heart disease. Nat Rev Cardiol. 2017;14(3):145-155. PubMed CrossRef
4.DiMatteo MR, Lepper HS, Croghan TW. Depression is a risk factor for noncompliance with medical treatment: meta-analysis of the effects of anxiety and depression on patient adherence. Arch Intern Med. 2000;160(14):2101-2107. PubMed CrossRef
5.Katon W, Lin EH, Kroenke K. The association of depression and anxiety with medical symptom burden in patients with chronic medical illness. Gen Hosp Psychiatry. 2007;29(2):147-155. PubMed CrossRef
6.Neff KD. The development and validation of a scale to measure self-compassion. Self Ident. 2003;2(3):223-250. CrossRef
7.Brown L, Bryant C, Brown V, et al. Self-compassion, attitudes to ageing and indicators of health and well-being among midlife women. Aging Ment Health. 2016;20(10):1035-1043. PubMed CrossRef
8.Brown L, Huffman JC, Bryant C. Self-compassionate aging: a systematic review. Gerontologist. 2019;59(4):e311-e324. PubMed
9.Phillips WJ, Ferguson SJ. Self-compassion: a resource for positive aging. J Gerontol B Psychol Sci Soc Sci. 2013;68(4):529-539. PubMed CrossRef
10.Sirois FM, Molnar DS, Hirsch JK. Self-compassion, stress, and coping in the context of chronic illness. Self Ident. 2015;14(3):334-347. CrossRef
11.Robins RW, Trzesniewski KH, Tracy JL, et al. Global self-esteem across the life span. Psychol Aging. 2002;17(3):423-434. PubMed CrossRef
12.Neff KD, Vonk R. Self-compassion versus global self-esteem: two different ways of relating to oneself. J Pers. 2009;77(1):23-50. PubMed CrossRef
13.Hwang S, Kim G, Yang J-W, et al. The moderating effects of age on the relationships of self’ compassion, self’ esteem, and mental health. Jpn Psychol Res. 2016;58(2):194-205. CrossRef
14.Neff KD, Germer CK. A pilot study and randomized controlled trial of the Mindful Self-Compassion Program. J Clin Psychol. 2013;69(1):28-44. PubMed CrossRef
15.Friis AM, Johnson MH, Cutfield RG, et al. Kindness matters: a randomized controlled trial of a mindful self-compassion intervention improves depression, distress, and HbA1c among patients with diabetes. Diabetes Care. 2016;39(11):1963-1971. PubMed CrossRef
16.Kirby JN, Tellegen CL, Steindl SR. A meta-analysis of compassion-based interventions: current state of knowledge and future directions. Behav Ther. 2017;48(6):778-792. PubMed CrossRef
17.Perez-Blasco J, et al. The effects of mindfulness and self-compassion on improving the capacity to adapt to stress situations in elderly people living in the community. Clin Gerontol. 2016;39(2):90-103. CrossRef
18.Leventhal H, Halm E, Horowitz, et al. Living with chronic illness: a contextualized, self-regulation approach. In: Sutton S, Baum A, Johnston M, eds. The SAGE Handbook of Health Psychology. London, UK: SAGE Publications; 2004:197-240.
19.Smeets E, Neff K, Alberts H, et al. Meeting suffering with kindness: effects of a brief self-compassion intervention for female college students. J Clin Psychol. 2014;70(9):794-807. PubMed CrossRef
20.Levine GN, Lange RA, Bairey-Merz CN, et al; American Heart Association Council on Clinical Cardiology; Council on Cardiovascular and Stroke Nursing; and Council on Hypertension. Meditation and cardiovascular risk reduction: a scientific statement from the American Heart Association. J Am Heart Assoc. 2017;6(10):e002218. PubMed CrossRef
21.Millstein RA, Huffman JC. Psychosocial management of patients with heart disease. In: Vranceanu AM, Greer JA, Safren SA, eds. The Massachusetts General Hospital Handbook of Behavioral Medicine. New York, NY: Humana Press; 2017:211-229.
22.Lawton J, Parry O, Peel E, et al. Diabetes service provision: a qualitative study of newly diagnosed type 2 diabetes patients’ experiences and views. Diabet Med. 2005;22(9):1246-1251. PubMed CrossRef
23.Kabat’ Zinn J. Mindfulness’ based interventions in context: past, present, and future. Clin Psychol Sci Pract. 2003;10(2):144-156. CrossRef
24.Leon AC, Davis LL, Kraemer HC. The role and interpretation of pilot studies in clinical research. J Psychiatr Res. 2011;45(5):626-629. PubMed CrossRef
25.Moore CG, Carter RE, Nietert PJ, et al. Recommendations for planning pilot studies in clinical and translational research. Clin Transl Sci. 2011;4(5):332-337. PubMed CrossRef
26.Blandy LM, Beevers WA, Fitzmaurice K, et al. Therapeutic argentine tango dancing for people with mild Parkinson’s disease: a feasibility study. Front Neurol. 2015;6:122. PubMed CrossRef
27.Fielding RA, Katula J, Miller ME, et al; Life Study Investigators. Activity adherence and physical function in older adults with functional limitations. Med Sci Sports Exerc. 2007;39(11):1997-2004. PubMed CrossRef
28.Proyer RT, Gander F, Wellenzohn S, et al. Positive psychology interventions in people aged 50-79 years: long-term effects of placebo-controlled online interventions on well-being and depression. Aging Ment Health. 2014;18(8):997-1005. PubMed CrossRef
29.Radloff LS. The CES-D scale: a self-report depression scale for research in the general population. Appl Psychol Meas. 1977;1(3):385-401. CrossRef
30.Beekman AT, Deeg DJ, Van Limbeek J, et al. Criterion validity of the Center for Epidemiologic Studies Depression Scale (CES-D): results from a community-based sample of older subjects in The Netherlands. Psychol Med. 1997;27(1):231-235. PubMed CrossRef
31.Watson D, Clark LA, Tellegen A. Development and validation of brief measures of positive and negative affect: the PANAS scales. J Pers Soc Psychol. 1988;54(6):1063-1070. PubMed CrossRef
32.Diener E, Emmons RA, Larsen RJ, et al. The Satisfaction With Life Scale. J Pers Assess. 1985;49(1):71-75. PubMed CrossRef
33.Mccullough ME, Emmons RA, Tsang JA. The grateful disposition: a conceptual and empirical topography. J Pers Soc Psychol. 2002;82(1):112-127. PubMed CrossRef
34.Shaffer F, McCraty R, Zerr CL. A healthy heart is not a metronome: an integrative review of the heart’s anatomy and heart rate variability. Front Psychol. 2014;5:1040. PubMed CrossRef
35.Brown L, Karmakar C, Gray R, et al. Heart rate variability alterations in late life depression: a meta-analysis. J Affect Disord. 2018;235:456-466. PubMed CrossRef
36.Dekker JM, Crow RS, Folsom AR, et al. Low heart rate variability in a 2-minute rhythm strip predicts risk of coronary heart disease and mortality from several causes: the ARIC Study, Atherosclerosis Risk In Communities. Circulation. 2000;102(11):1239-1244. PubMed CrossRef
37.Tsuji H, Larson MG, Venditti FJ Jr, et al. Impact of reduced heart rate variability on risk for cardiac events: the Framingham Heart Study. Circulation. 1996;94(11):2850-2855. PubMed CrossRef
38.Tsuji H, Venditti FJ Jr, Manders ES, et al. Reduced heart rate variability and mortality risk in an elderly cohort: the Framingham Heart Study. Circulation. 1994;90(2):878-883. PubMed CrossRef
39.Kirby JN, Doty JR, Petrocchi N, et al. The current and future role of heart rate variability for assessing and training compassion. Front Public Health. 2017;5:40. PubMed CrossRef
40.Bauer S, Lambert MJ, Nielsen SL. Clinical significance methods: a comparison of statistical techniques. J Pers Assess. 2004;82(1):60-70. PubMed CrossRef
41.Lakens D. Calculating and reporting effect sizes to facilitate cumulative science: a practical primer for t-tests and ANOVAs. Front Psychol. 2013;4:863. PubMed CrossRef
42.Cohen J. Statistical Power Analysis for the Behavioral Sciences. 2nd Edition. Mahwah, NJ: Lawrence Erlbaum Associates; 1988.
43.Arch JJ, Craske MG. Mechanisms of mindfulness: emotion regulation following a focused breathing induction. Behav Res Ther. 2006;44(12):1849-1858. PubMed CrossRef
44.Sagawa M, Oka M, Chaboyer W. The utility of cognitive behavioural therapy on chronic haemodialysis patients’ fluid intake: a preliminary examination. Int J Nurs Stud. 2003;40(4):367-373. PubMed CrossRef
45.Celano CM, Millstein RA, Bedoya CA, et al. Association between anxiety and mortality in patients with coronary artery disease: a meta-analysis. Am Heart J. 2015;170(6):1105-1115. PubMed CrossRef
46.Murphy BM, Elliott PC, Higgins RO, et al. Anxiety and depression after coronary artery bypass graft surgery: most get better, some get worse. Eur J Cardiovasc Prev Rehabil. 2008;15(4):434-440. PubMed CrossRef
47.Button KS, Ioannidis JP, Mokrysz C, et al. Power failure: why small sample size undermines the reliability of neuroscience. Nat Rev Neurosci. 2013;14(5):365-376. PubMed CrossRef
48.Tucker M, Oei TP. Is group more cost effective than individual cognitive behaviour therapy? The evidence is not solid yet. Behav Cogn Psychother. 2007;35(1):77-91. CrossRef
Enjoy this premium PDF as part of your membership benefits!