Lessons Learned at the Interface of Medicine and Psychiatry
The Psychiatric Consultation Service at Massachusetts General Hospital sees medical and surgical inpatients with comorbid psychiatric symptoms and conditions. During their twice-weekly rounds, Dr Stern and other members of the Consultation Service discuss diagnosis and management of hospitalized patients with complex medical or surgical problems who also demonstrate psychiatric symptoms or conditions. These discussions have given rise to rounds reports that will prove useful for clinicians practicing at the interface of medicine and psychiatry.
Prim Care Companion CNS Disord 2026;28(2):25f04072
Author affiliations are listed at the end of this article.
Have you ever wondered whether (and when) you could use electroconvulsive therapy (ECT) in an intensive care unit (ICU) to treat a seriously ill individual with a psychiatric and/or neuropsychiatric disorder? Have you been uncertain about whether you need to be credentialed to perform this treatment and what types of support and training (eg, medical staff, anesthesiologists, nursing staff) you would need to ensure your patient’s safety? Have you puzzled over what type of medical clearance might be required before beginning ECT in the ICU? If you have, this article should prove to be useful.
CASE VIGNETTE
Mr A, a 37-year-old house painter, sustained a spinal cord transection and multiple orthopedic injuries in a 3-story fall while intoxicated. His postoperative course was complicated by respiratory failure, bilateral pulmonary emboli, and pneumonia. During his recovery, he became agitated and delirious, which required use of antipsychotics (haloperidol and quetiapine). His behavioral dysregulation led to dislodgement of his percutaneous endoscopic gastrostomy tube, and he required an emergency laparotomy. In the days that followed, dopamine antagonists were gradually tapered and discontinued, and he was transferred to the medical floor. However, Mr A developed intense fear, rigidity, mutism, staring, catalepsy, stereotypic movements, and profound withdrawal, which were suggestive of catatonia. He was transferred back to the ICU, and high-dose intravenous (IV) lorazepam was administered to permit adequate ventilatory support. Despite use of lorazepam, his symptoms escalated. He became febrile (temperature=39°C [102.2°F]), tachycardic (heart rate [HR]=130 beats/minute), diaphoretic, and rigid, each a hallmark of life-threatening malignant catatonia. Since IV lorazepam failed to solve his symptoms, and motor and autonomic features worsened, he was at high risk of cardiovascular collapse, rhabdomyolysis, and death; a course of ECT was initiated.
DISCUSSION
Which Conditions Can Be Treated by ECT?
ECT is a highly effective treatment for several severe psychiatric conditions (eg, refractory depression, catatonia, mania, and psychosis), and it has been useful for the management of persistent delirium and refractory status epilepticus (RSE).1 While its use in outpatient settings has been established, administering ECT in ICUs presents myriad challenges, as critically ill individuals typically have multiple organ dysfunction, are hemodynamically unstable, and rely on life-support measures that require meticulous and coordinated care.2,3 Central to performing ECT in the ICU is the collaboration among psychiatrists, intensivists, anesthesiologists, and nursing staff to address psychiatric and critical care needs as part of a comprehensive treatment plan that is aligned with ongoing medical management.2,4
Despite its potential benefits, use of ECT in ICUs remains limited by barriers due to a multitude of institutional protocols (eg, with guidelines for patient selection, informed consent, and crisis management, which are essential to the provision of ethical, equitable, and safe care for patients in the ICUs),5,6 a lack of trained teams, and inadequate resources. Enhancing access requires planning, interdisciplinary simulations, and institutional policies that streamline decision-making and facilitate timely care.
When Should ECT Be Considered for Critically Ill Individuals?
ECT can be lifesaving for critically ill individuals with several psychiatric and neuropsychiatric conditions, and it should be considered when life-threatening neuropsychiatric syndromes are refractory to standard medical treatment. Although its use in critical care environments remains uncommon, evidence supporting ICU-based ECT continues to grow, particularly for malignant catatonia, neuroleptic malignant syndrome (NMS), RSE, refractory neuropsychiatric symptoms in autoimmune encephalitis (AE), and prolonged delirium with catatonic features.
Catatonia is the most common indication for ICU-based ECT. It has been recognized increasingly in ICUs, with rates ranging from 3.8% to 23%, depending on the population studied and the diagnostic criteria used.7,8 When catatonia has a medical/neurological etiology, it is often resistant to benzodiazepines (BZDs) and other adjunctive agents (eg, amantadine, zolpidem).
Malignant catatonia, the life-threatening subtype of catatonia marked by autonomic instability (eg, hyperthermia, tachycardia, and labile blood pressure [BP]), can induce myocardial infarction, multiorgan failure, and cardiopulmonary arrest.8,9 It carries exceptionally high morbidity and mortality; when left untreated, fatality rates are as high as 75%–100%. Therefore, malignant catatonia is a medical emergency, for which ICU-based ECT is indicated.9,10
Since the efficacy of ECT diminishes when its initiation has been delayed, timely treatment is essential,11,12 as catatonia becomes increasingly refractory over time. When used for catatonia, ECT should be administered daily. Similarly, ECT schedules in the ICU are more frequent than those used for patients with mood disorders, which reinforces the importance of rapid identification and early treatment.
NMS is an indication for ECT, especially when it has been unresponsive to standard measures (eg, when withholding a dopamine-blocking agent, use of supportive care, and administration of dopamine agonists [eg, bromocriptine, amantadine] have failed).13 ECT likely exerts its therapeutic effect through downregulation of the D2 receptors;14,15 accordingly, reports suggest that ECT reduces symptoms rapidly (ie, within several sessions).16,17 A systematic review of over 400 cases noted that ECT was associated with the lowest mortality among treatment modalities, especially in severe cases.18 Moreover, the benefit of ECT was most pronounced in patients with severe presentations, whereas in those with mild or moderate cases, specific therapies failed to show a significant advantage over supportive care.18
Status epilepticus that is refractory to both first- and second-line treatments, and even anesthetic agents, represents another condition for which ECT may be lifesaving. Case series suggest that it leads to seizure cessation or reduction in 58%–80% of patients treated with ECT.19 Although evidence of its utility comes from low-quality studies, ECT’s effect on γ-aminobutyric acid (GABA)ergic and glutamatergic neurotransmission as well as neuroplastic remodeling is thought to raise the seizure threshold and terminate seizures.20,21 Although more research is needed to establish ECT’s efficiency, it appears to be an important salvage therapy for treatment-refractory seizures.21
In cases of AE that have been refractory to use of immunotherapy and psychotropics, ECT is not a first-line treatment; however, it is increasingly used as an adjunctive intervention for those with catatonia and psychosis. Systematic reviews and case series report response rates between 65% and 77% among patients with AE who received ECT for treatment-resistant symptoms, with catatonia being the most common indication for ECT.22,23
ECT has been administered across various AE subtypes, including anti-N-methyl-D-aspartate receptor and antirecovery in antibody-positive encephalitis. Typically, ECT has been initiated after standard immunosuppressive therapies (eg, corticosteroids, intravenous immunoglobulin, plasma exchange, rituximab, or cyclophosphamide) have been exhausted or when symptom control is required urgently due to life-threatening psychiatric presentations or catatonia. In cases in which BZDs or antipsychotics have failed, or are contraindicated, due to the risk of worsening catatonia, ECT may offer relief and speed the time to recovery. Although scheduling protocols specific to AE are lacking, ECT for this indication is typically delivered using standard catatonia protocols.
The use of ECT in prolonged or refractory delirium remains limited, and it should be reserved for refractory cases. Evidence of its use comes largely from case reports and small case series; benefits have been found in those with severe delirium that has failed to respond to standard pharmacologic and nonpharmacologic strategies.24,25
Delirium and catatonia often resemble one another and frequently co-occur, especially in critically ill individuals. Both syndromes can present with mutism, withdrawal, rigidity, or agitation, and their severity may fluctuate over time.8,26 This overlap creates diagnostic uncertainty. An important clinical clue is symptomatic worsening of symptoms following antipsychotic administration, as this may unearth catatonia.27,28 While the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, Text Revision, discourages diagnosing catatonia in the context of delirium, mounting evidence suggests that this exclusion is too rigid and may lead to missed opportunities for effective treatment.11
In those with an altered mental status and ambiguous features, a lorazepam challenge may clarify the diagnosis and offer a therapeutic benefit. A positive response generally supports the decision to treat catatonia, even in the context of comorbid delirium. When BZDs are ineffective or contraindicated, ECT becomes a meaningful next step—both to manage symptoms and, in some cases, to distinguish between the 2 syndromes.11
When Should ECT Be Tried?
Determining when to initiate ECT in critically ill individuals requires balancing diagnostic, therapeutic, and logistical considerations, with the overarching goal to rapidly address life-threatening neuropsychiatric syndromes when other treatments have failed or are acting too slowly. Clinical urgency is most often defined by the presence of life-threatening features and failure of first-line therapies. In malignant catatonia, for example, ECT should be initiated urgently, when there is no response to administration of high-dose IV BZDs or when symptoms (eg, autonomic instability, hyperthermia, or profound psychomotor disturbance) are present. Similar urgency applies when psychiatric symptoms interfere with necessary critical care (eg, extubation, nutrition) or when NMS or refractory delirium fails to respond to standard pharmacologic approaches.29
While ECT is considered after failed medication trials in critically ill individuals, prolonged delays are not always appropriate. In a patient who is deteriorating or who has a life-threatening psychiatric syndrome, ECT may be initiated before full pharmacologic trials have been conducted. In these cases, a coordinated interdisciplinary approach—engaging psychiatry, anesthesiology, critical care, and nursing—can mobilize care quickly, assess capacity, and streamline legal processes. When patients lack decision-making capacity, consent discussions, court petitions, or surrogate authorizations should be initiated in parallel with efforts at medical stabilization to prevent avoidable delays in potentially life-saving treatment.30
What Is the Role of Prognostication Regarding Medical and Psychiatric Conditions?
Prognostication plays a central role in guiding the use of ECT in palliative contexts. Patients in the ICU or with serious illness carry significant medical comorbidities that complicate ECT administration. Common scenarios include brain metastases, a recent stroke, or assessing the anesthetic risks associated with advanced organ dysfunction. These risks should be carefully considered. In some cases, adjustments can be made to mitigate risk. For example, electrode placement can be modified to avoid tumor-involved areas and burr holes from biopsies or tumor resection.31 However, treatment decisions must account for the patient’s broader clinical trajectory and values.11,29,32
When catatonia has been identified early, most patients respond to treatments (eg, with lorazepam or ECT), with response rates typically between 70% and 100%.9,11,27–29,32–34 On the other hand, delays in diagnosis or treatment are associated with worse outcomes and treatment responses.27–29,32–34 Catatonia may also reflect broader clinical deterioration (eg, a new stroke, systemic infection, psychiatric decompensation, or cancer progression).7,32,35 Like delirium, it may coexist with serious medical illness and can complicate prognostic assessment. Catatonic symptoms—such as withdrawal or immobility—can also independently contribute to medical complications.10 These factors should be considered carefully in discussions about the role of ECT, and evaluations should be individualized. Some patients may opt to proceed with ECT despite the risks of acute and prolonged complications (eg, aspiration, stroke, or extended hospitalization), which may reflect sequelae of the underlying conditions. In practice, the risks of ECT can be anticipated and avoided. Others, especially those nearing the end of life, may decline further interventions once the potential burdens outweigh the expected benefit. For those individuals, clinical judgment should guide decisions about diagnostic testing and treatment trials. In either case, shared decision-making that is informed by the prognosis, patient goals, and the likely therapeutic impact is essential.
Who Should Decide Whether ECT Should Be Initiated and From Whom Should Informed Consent Be Obtained?
In ICUs, the process for obtaining informed consent for ECT begins with a psychiatric consultation to determine its indication, appropriateness, relevant comorbidities, and potential person-specific adverse effects of ECT. Ideally, the recommendation for ECT is a collaborative one among the ICU team and the psychiatric consultants. An important factor to acknowledge is the patient’s medical acuity, which amplifies the implications of choosing one treatment over another.
The informed consent process for ECT proceeds as it would for any other medical procedure as determined by local, state, and federal law. Whenever possible, the patient’s autonomy is respected by allowing them to choose the nature of treatment. However, if a patient’s abnormal mental status precludes full participation in the informed consent process and they are deemed to lack the capacity to make this treatment decision, autonomy can still be honored by referencing advance care planning documents or wishes. If the patient does not have an advanced directive regarding ECT, then a surrogate decision-maker should be contacted. Surrogate decision-makers are selected in several ways, depending on the patient’s situation and local statutes.
As with most medical procedures, written consent should be obtained from the patient or a surrogate decision-maker. Informed consent for ECT requires strategic and effective collaboration among ICU team members, the anesthesiology team, and the psychiatrist who will be performing ECT. In some situations, consents for ECT and for anesthesia are considered as separate processes. The decision-maker must be provided with adequate information about the potential benefits, risks, and alternatives for the intervention. What needs to be covered in the informed consent process has been reasonably well standardized for ECT, and the normal standards apply to patients undergoing ECT in the ICU.36 This process also allows the patient or their surrogate to ask questions to further augment their understanding of the procedure. This collaborative approach to shared decision-making allows the patient, the surrogate, and the treatment teams to have a full understanding of the patient-specific risks and the benefits of the procedure.
What Are the Legal Considerations for Consenting to ECT?
When a patient lacks the capacity to consent to ECT, the process for obtaining informed consent varies across the United States. Variability in regional statutes regarding ECT and medical center policies also affects the ECT consent process.37 While much of the legislation regarding ECT aims to enhance patient safety, regulations often make the process more complex and may erect significant barriers to accessing ECT. Some US states treat ECT like any other medical procedure, while others have introduced limitations for surrogate consent for ECT when the patient is unable to consent for themselves. The process of identifying the surrogate decision-maker can be complicated, depending on state laws. In some states, ECT is considered an “extraordinary treatment,” raising the threshold of decision-making and needing additional legal processes to identify the appropriate person to provide consent, even in an emergency. For example, some states require legal guardianship to provide surrogate consent for ECT, a complex legal process that, in some areas, can take months to accomplish. Others allow surrogate consent only when there is a formal advance directive stating that ECT is allowed. Some jurisdictions have exceptions for emergencies or life-threatening situations, but this is not universal.37,38 Furthermore, statutes often employ vague or unclear language that leaves room for interpretation, in which case, legal counsel may be required to ensure that clinicians interpret regulations properly and act in kind. Similar variability and challenges have also been noted to impact ECT delivery in European countries.39
What Should Be Discussed With Families Regarding ECT?
Interdisciplinary communication among patients, families, and staff is crucial to the successful utilization of ECT in the ICU environment. Consultation-liaison (C-L) psychiatrists are instrumental in facilitating communication with the treatment team and the patient’s family, sharing important logistical considerations while also providing psychoeducation about ECT and its expected risks and benefits. In some states, families of incapacitated patients may be required to provide informed consent for their loved ones for ECT (as opposed to judicial authorization for ECT in other states), in which case, a C-L psychiatrist can provide in-person guidance to the patient’s surrogate decision-maker, improving the family’s understanding of the treatment and helping to combat stigma, which might otherwise limit or delay the family’s willingness to provide consent. A systematic review of relatives’ knowledge, attitudes, and experiences toward ECT found that exposure to ECT and the use of psychoeducational tools improved both knowledge and attitudes in caregivers. However, in general, family members wanted more information about the risks and side effects of ECT before proceeding with treatment.40
When meeting with families before embarking on ECT, C-L psychiatrists may wish to provide materials about the procedure that can be reviewed by family. It is especially helpful for the psychiatrist to discuss the signs and symptoms that the family can look for and that may indicate neuropsychiatric worsening or improvement, so that families may monitor their loved ones closely for clinical indicators. Similarly, teams may benefit from receiving guidance about clinical signs in the posttreatment period. These features can be documented in the psychiatrist’s note, alongside notable information about the procedure. Some institutions create pre- and postprocedure checklists when conducting ECT in ICUs, so that the treatment team has a clear resource that outlines the process for administering, monitoring, and managing the sequelae of adverse events. Such checklists can be created with input from psychiatry, the ICU, and anesthesia services to ensure that appropriate expertise is incorporated.
During the post-ECT period, the psychiatrist should evaluate the patient for evidence of postprocedure worsening or improvement and check with the family and the ICU team. The psychiatrist’s presence may facilitate an opportunity for the family to ask additional questions about the procedure and obtain reassurance that their loved one has tolerated the ECT treatment well. C-L psychiatrists may also ensure that other members of the interdisciplinary treatment group feel comfortable about the procedure moving forward and address remaining concerns. By having a frequent physical presence in the ICU, psychiatrists can establish a strong working relationship with open lines of communication with all stakeholders, thus facilitating the successful administration of ECT in the ICU.
Which Medications Should Be Reviewed in Critically Ill Patients Receiving ECT?
Standing and continuous medications. Critically ill patients in whom ECT is indicated often receive medications that interfere with successful treatment, particularly in those with malignant catatonia (eg, in those receiving high-dose BZDs) or RSE (receiving antiepileptic drugs [AEDs]). Depending on the agent selected, standing doses of sedating agents may also alter the seizure threshold. In outpatient practices, discontinuing or lowering the dosage of these medications is advised, with the dose held the evening before the treatment. Studies provide diverging findings regarding the impact of maintaining BZDs and AEDs on the seizure threshold, its duration, and the treatment response41; studies that have investigated these factors in ICUs are lacking.
Pharmacologic management of critically ill patients often leads to difficulty in predicting changes in pharmacodynamics and pharmacokinetics (eg, with impaired enteral absorption, reduced hepatic blood flow, changes in glymphatic flow, inflammation-mediated changes in P450/p-glycoprotein functioning, changes in serum protein-binding capacity, volume shifts) and changes in pharmacokinetics due to interventions (eg, dialysis and extracorporeal membrane oxygenation).42 Moreover, it can be difficult to anticipate the degree and duration of central nervous system drug penetration. In-depth discussion and joint treatment planning among the treatment teams is essential before initiating ECT. Given the variability in medication regimens and the clinical status of patients considered for ECT in critical care units, it is impractical to propose a standardized protocol for individual agents ahead of treatment. However, several principles can guide medication management.
For drug classes that are expected to impact the seizure threshold, it is reasonable to use drugs with a lower volume of distribution and a shorter half-life. Redundant or ineffective medications should be discontinued, if possible, and necessary medications should be maintained at the lowest effective dose. It is also reasonable to hold relevant medications at least one half-life ahead of ECT, although for those drugs administered in high dosages or for a protracted period, successful seizure induction may still be problematic. It is often helpful to consider that most centrally active medications are lipid soluble and build up significant reservoirs of drugs in the brain due to their high lipid concentration. This is likely reflected in the higher stimulus parameters needed for adequate treatment in ICU patients. Augmentation methods, discussed below, can be helpful in these cases.
Regarding standing sedation, dexmedetomidine at 1 mcg/kg can provide BP control during treatment without affecting seizure duration. Ketamine at 1.5–2 mg/kg can provide sedation with minimal impact on seizure threshold or duration.43 When propofol is being used, it is best to taper the dose several hours before beginning treatment and to hold it for 30–60 minutes ahead of treatment. Midazolam, when used, should be held at least 3–4 hours ahead of the treatment, with consideration about when to administer flumazenil.
Pretreatment medication considerations. The use of nondepolarizing neuromuscular-blocking agents (eg, rocuronium or atracurium) should be considered, as these patients have often experienced prolonged periods of immobilization that increase the risk of hyperkalemia, which can be compounded by depolarizing agents (eg, succinylcholine) due to upregulated extrajunctional acetylcholine receptors.
Similar to the use of ECT in non-ICU settings, pretreatment with caffeine or theophylline can prolong the seizure. Although caffeine can be associated with tachycardia44 in animal models, caffeine damages hippocampal neurons,45 and theophylline is associated with an increased risk of status epilepticus46; therefore, it may not be suitable for individuals with significant cardiac risk factors. Volitional or ventilator-assisted hyperventilation to an ETCO2 of 30 mm Hg can reduce the seizure threshold.47 There is conflicting evidence regarding the potential impact of flumazenil on seizure threshold and duration; however, this agent is, in general, considered safe and effective for those who are taking high doses of GABAA agonists prior to treatment and may reduce the stimulation that would otherwise be required in this population.48 It is prudent to wait long enough after the administration of flumazenil, as some patients show clinical effects shortly after injection, but the peak effect does not occur until 6–10 minutes after the dose. Also, if flumazenil is used, it is important to give additional BZD doses after the treatment to compete with flumazenil at BZD receptors.
Common hypnotic agents—propofol, etomidate, thiopental, and methohexital—exhibit anticonvulsant properties. High induction doses may impair seizure initiation and duration, while subanesthetic doses risk intraoperative awareness if not carefully titrated. Opioids are sometimes coadministered to reduce hypnotic dose and preserve seizure quality, although current evidence does not demonstrate a clear benefit of opioid-hypnotic combinations over hypnotic monotherapy.49
Dexmedetomidine, as a centrally acting α2-adrenergic agonist, has been evaluated as an adjunct to traditional anesthetic agents or beta-blockers.50,51 Small studies suggest that it may reduce postictal agitation, improve patient satisfaction, and attenuate sympathetic responses, including elevations in HR and mean arterial pressure.51 Propofol might also be considered for use as a general anesthetic, as it lowers BP and HR that may reduce some of the physiologic stress associated with the seizure. Fentanyl and its derivatives may prolong the seizure duration, but they have been associated with greater HR and BP variability during ECT.52
Etomidate remains a favorable option for hemodynamic stability, particularly in the critically ill. However, its use is tempered by its well-documented suppression of adrenal steroidogenesis—even after a single dose.53 This risk is especially concerning in patients with sepsis or high illness severity, where impaired adrenal function may contribute to worse outcomes and has been associated with increased mortality in some studies.54 Risk-benefit assessment is therefore essential when considering etomidate in this population.
While ECT’s autonomic effects are typically short-lived, they may be attenuated with short-acting agents, such as beta-blockers (eg, esmolol, labetalol) and calcium channel blockers (eg, nicardipine). Anticholinergic agents may also be used to blunt the initial parasympathetic surge and reduce bradyarrhythmia or asystole risk. Proactive pharmacologic planning is especially important in patients with limited physiologic reserve.
Esmolol, a short-acting selective β1-antagonist, can reduce the acute rise in HR and BP following ECT.55 Labetalol, a combined α- and β-adrenergic antagonist, exerts similar effects with a longer duration of action. Its α1-blocking properties may reduce reflex vasoconstriction, and it is advantageous in patients vulnerable to increased systemic or pulmonary vascular resistance.56 Moreover, labetalol causes greater reductions in middle cerebral artery blood flow velocity compared to esmolol, suggesting a potential neuroprotective benefit in patients with a history of cerebrovascular disease.57
What Should Medical Workup Consist of for ECT in the ICU?
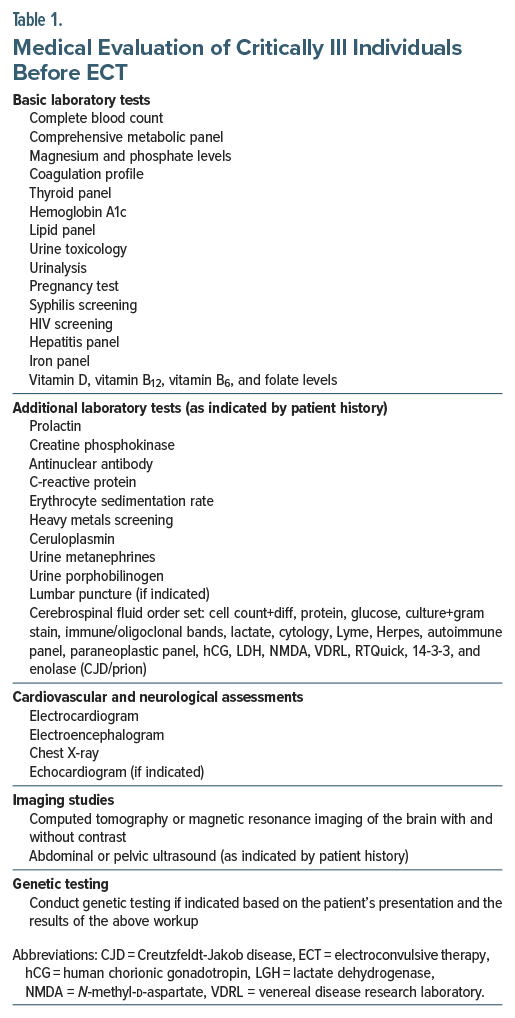
Before administering ECT in the ICU, a thorough medical workup is necessary. Essential components of the medical workup for ECT in the ICU are displayed in Table 1.58,59 These tests should be considered based on the appropriate indications and to assist if the diagnosis has not yet been established.
What Are the Relative and Absolute Contraindications for Performing ECT?
An elevated intracranial pressure has been cited as an absolute contraindication to ECT, primarily due to the risk of exacerbating cerebral edema or precipitating herniation.60 The presence of signs or symptoms of an intracranial lesion (eg, papilledema, focal neurological deficits, mass effect, or brain edema on imaging) or a recent acute hemorrhagic stroke (particularly within 30 days and with poorly controlled BP) is considered a high-risk indicator.29,61
Individuals with intracranial space-occupying lesions report complications (eg, Todd’s paralysis, ping-pong gaze, coma, delirium, myoclonic seizures) following ECT, although evidence of this is minimal. Brain imaging may be warranted in higher-risk individuals (eg, heritable disorders with intracranial aneurysms, prior aneurysms, age older than 60 years, history of smoking, history of hypertension). Intra-arterial BP monitoring may be necessary while administering esmolol to determine the real-time variation of BP during each phase (eg, preictal, ictal, and postictal) to determine the dosing of esmolol in future sessions. Administering dexamethasone in individuals with intracranial tumors may reduce the risk of edema surrounding the tumor and administering phenytoin may reduce the risk of recurrent seizures.62 Recent reports no longer support listing space-occupying lesions as an absolute contraindication for ECT. Each case should be reviewed individually in consultation with specialists.61
Other conditions are considered relative contraindications that significantly elevate risk and necessitate careful management, and this includes a recent hemorrhagic stroke (within 30 days) or poorly controlled cerebrovascular disease, and uncontrolled hypertension or severe cardiovascular instability (eg, recent myocardial infarction, unstable angina, decompensated heart failure, severe valvular disease, or significant arrhythmias). Additional considerations include the presence of an unrepaired large abdominal aortic aneurysm and pheochromocytoma without adequate adrenergic blockade. Each of these conditions requires thorough evaluation and tailored management to mitigate potential risks associated with ECT.29,63
Can ECT Be Performed Safely in the Context of Cardiac and Neurologic Conditions?
Mortality directly attributed to ECT is exceptionally rare. When complications do occur in patients receiving ECT, the underlying mechanisms are attributed to hemodynamic alterations, the effects of anesthesia, and convulsions. Risks associated with ECT can be mitigated with appropriate collaboration, education, use of safety protocols, and communication.63
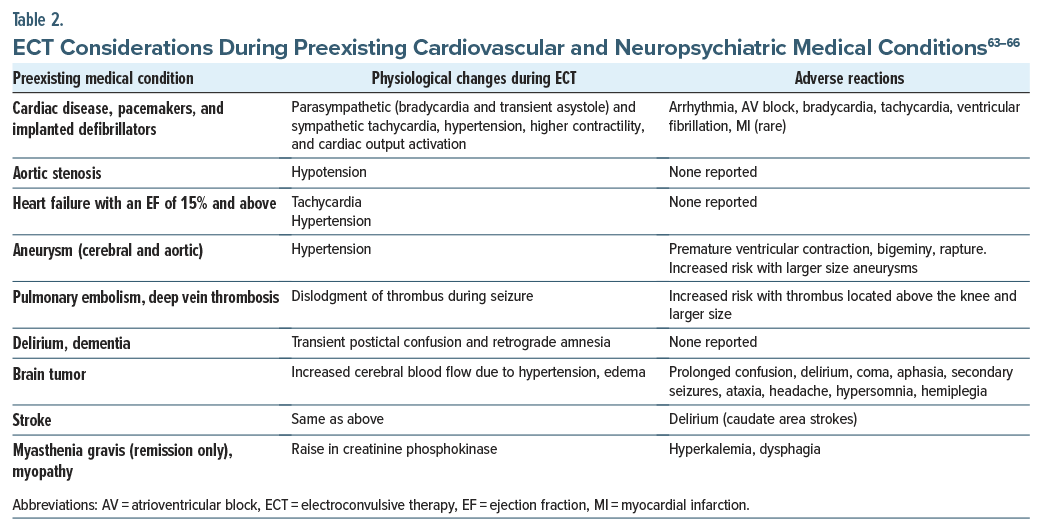
ECT is a relatively safe procedure and can be used in patients with preexisting cardiac and neuropsychiatric conditions (Table 2). A transient asystole and bradycardia may occur initially as the electric stimulus is parasympathetic followed by a catecholamine surge lasting several minutes. During this surge, individuals may experience increased cardiac contractility, hypertension, and tachycardia. Cardiac output may increase to 81%. Changes are often blocked by administering a β-adrenoceptor antagonist.63
Unfortunately, insufficient information is available regarding cardiac failure and ECT. In a case series, 5 patients with congestive heart failure did not experience any sentinel events. Moreover, data support the safety of ECT in patients with a cardiac ejection fraction (EF) as low as 15%. Treatment strategies should focus on administering ECT along with medications that assist in reducing the cardiac workload or beta-blockers that reduce hypertension and tachycardia.63
Cardiac monitoring (eg, electrocardiography, non-invasive BP monitoring, pulse oximetry) in ECT is essential. Detecting rapid changes in patients with high-risk conditions may require continuous monitoring to detect hemodynamic changes. Patients with a low EF may require monitoring with BP cuffs on both arms with continuous cycling and an arterial line.63
There are risks associated with ECT in neurological (eg, aneurysms, strokes) conditions. The risks of ECT in aneurysms appear to increase as the size of the aneurysm increases. Post-ECT delirium during the poststroke period was more commonly observed after caudate strokes.63
Should ECT Be Administered to Patients With Limited Prognosis or for “Palliative” Indications in the ICU?
Palliative care is specialized medical care that is focused on relieving symptoms and the stress of serious illness. It emphasizes multidisciplinary management of physical, psychological, and spiritual suffering while supporting complex decision-making for patients and their families. Strong evidence supports its utility across a wide range of conditions (eg, cancer, heart failure, agitation associated with dementia,31 and chronic obstructive pulmonary disease). Benefits of ECT include an improved quality of life, reduced caregiver burden, and, in some cases, prolonged survival.32,33 Palliative care psychiatry involves managing psychiatric symptoms in medically ill patients, applying palliative principles to treatment-refractory mental illness, and considering interventions like ECT, when appropriate.34
Although ECT in palliative care or for patients at the end of life in ICU settings remains understudied, ECT rapid onset and efficacy in the treatment of catatonia, depression, and psychosis make it a promising option in situations in which time is limited. While no studies have evaluated its effect on quality of life or caregiver distress associated with serious illness, clinical experience suggests that ECT can restore function and communication even when it is used near the end of life. In this context, ECT may serve as a valuable palliative intervention. Moreover, there is growing interest in the use of “palliative ECT.”35,36 A recent cohort study of those referred for palliative care and ECT found that many individuals were treated for catatonia in the setting of cancer, advanced organ disease, or dementia. Outcomes included cognitive improvement, reversal of life-threatening weight loss, and correction of dementia that was misdiagnosed and later attributed to catatonia. Those patients experienced symptom relief and, on occasion, a reorientation of goals of care.36
As with any intervention near the end of life, the decision to pursue ECT must be carefully contextualized within the patient’s prognosis, values, and goals of care. The potential benefits of symptom relief, functional recovery, or reestablished communication should be weighed against the procedural and anesthetic risks, the expected trajectory of the underlying illness, and what outcomes the patient and family consider meaningful. Integrating palliative care early in these discussions supports realistic prognostication, shared decision-making, and alignment of treatment with the patient’s priorities, ensuring that the use of ECT, when chosen, reflects both medical appropriateness and the values that define a good outcome for that individual.
What Is the Recommended Duration of ECT for Medically Ill Patients?
Since there is no current consensus on the exact number of ECT treatments, the duration of treatment should be based on the patient’s clinical symptoms, response, and side effects. The course of ECT should not be prefixed; rather, it should be dynamic and reflect the individual’s needs. Acute courses of ECT may occur approximately 2–3 times per week.67 On average, 8–12 sessions are recommended before deeming ECT ineffective. Changing electrode placement or pulse duration should be tried before discontinuing ECT.59 Some patients may require more sessions (eg, with schizophrenia) compared to those with depression or mania. Cases of malignant catatonia may need more than 20 sessions.68
For malignant catatonia, international treatment guidelines recommend daily ECT at least for the first week. Some patients may need several sessions after partial or full remission of catatonia.2
How Does Stigma Surrounding ECT Impact Its Acceptance and Use as a Treatment Option, Particularly in Critical Care Settings?
Large-scale, epidemiological research on ECT in the United States is limited, especially when it has been restricted to critical care settings. However, stigma surrounding the use of ECT impairs its acceptance and utilization, including in the ICU, despite its demonstrated efficacy in life-threatening conditions like malignant catatonia. This stigma is multifactorial, rooted in outdated perceptions perpetuated by media misrepresentation, public and professional misinformation, and fears of cognitive side effects. This stigma exists in the general population but is also prevalent among health care providers, which contributes to systemic and interpersonal barriers to ECT implementation.69
In critical care settings, this stigmatization can delay or prevent timely ECT administration. In several case series,2,3 patients with malignant catatonia in the ICU showed significant clinical improvement with ECT after the failure of pharmacologic agents. However, delays were introduced when multidisciplinary consensus and family consent were required, especially when negative perceptions of ECT clouded clinical judgment and decision-making. This delay in care is impacted by inadequate institutional ECT infrastructure, staff unfamiliarity with procedural protocols in the ICU, and interdepartmental reluctance to coordinate ECT delivery under emergent conditions.
Geographic, racial, and socioeconomic disparities in ECT acceptance and use are documented. Nearly 90% of US hospitals do not offer ECT,69 and its utilization disproportionately favors white, older, wealthier patients with private insurance.70,71 Moreover, racial and ethnic minorities are significantly less likely to receive ECT even when clinically indicated, suggesting the role of implicit clinician bias and cultural diagnostic discordance.70,72 This biased referral pattern likely compounds stigma among underserved populations and reinforces underdiagnosis and undertreatment.
ECT appears to be a broadly underused modality in psychiatric care, and there are data on the decline of ECT use over time. Data from the National Institute of Mental Health (NIMH) Sample Survey Program from private general hospitals, private psychiatric hospitals, public general hospitals, and state and country mental hospitals demonstrated that in 1975, 58,667 patients received ECT compared with 36,558 in 1986. 73 In the United States, the National Inpatient Sample, a nationally representative survey conducted annually by the Agency for Healthcare Research and Quality, demonstrated that in US community hospitals from 1993 to 2009, the annual number of inpatient psychiatric stays involving ECT fell from 12.6 to 7.2/100,000 adult patients. They also found that the percentage of hospitals conducting ECT decreased from 14.8% to 10.6%, and the probability of inpatients receiving ECT for depression fell by 34%.74 In this survey, US community hospitals are defined as nonfederal, short-term, general, or specialty hospitals, including both public and private facilities and academic medical centers. Both the data from the NIMH Sample Survey and the National Inpatient Sample Survey are reflective of patients who received ECT in inpatient settings; however, neither source delineated whether the inpatient treatments were administered specifically in the ICU. These data points emphasize the decline in use of ECT and hint toward the stigma-driven barriers in its consideration. This is particularly detrimental to patient care, as these barriers also detract from use in severe presentations where ECT may offer rapid therapeutic benefit, such as ICU-level cases of malignant catatonia, depression with suicidality, pharmacotherapy-resistant protracted delirium, agitation, and treatment-resistant psychosis.
Stigma surrounding ECT continues to exert a deleterious impact on its acceptance and application, including in critical care settings. It manifests through provider hesitancy, institutional barriers, and racialized inequities. The stigma impedes timely access to a highly effective and often life-saving intervention. Addressing the issue requires robust educational initiatives, structural investment in ECT services, and the dismantling of biases—both societal and clinical—that preclude equitable and evidence-based psychiatric care.
What Do Intensivists and Anesthesiologists Think of ECT?
From the intensivist’s perspective, ECT in critically ill patients demands careful multidisciplinary coordination and a nuanced approach to anesthetic management. Preprocedural evaluation must include a thorough assessment of preexisting comorbidities and mitigation of cardiovascular and anesthetic risks. ECT produces a robust autonomic response, typically characterized by an acute sympathetic surge with increases in HR and BP, mirroring the physiological stress of a cardiac stress test. This transient hemodynamic load may be poorly tolerated in patients with cardiomyopathy, pulmonary hypertension, or right ventricular dysfunction.
Neurologic considerations are equally important in medically complex patients. Maintenance of protective airway reflexes, prevention of aspiration, and anticipation of postictal delirium are essential.
Anesthetic induction and neuromuscular blockade should be individualized to balance hemodynamic stability with adequate seizure quality. A meta-analysis of 82 studies reported a low all-cause mortality rate associated with ECT (0.42 deaths per 1,000 patients), with approximately 29% attributed to cardiac complications.75
Stress-induced cardiomyopathy, or Takotsubo cardiomyopathy (TCM), has been linked with ECT.76,77 Clinically indistinguishable from acute coronary syndrome, TCM presents with chest pain, dyspnea, ST-segment elevations, and elevated cardiac biomarkers. Echocardiography typically reveals apical or basal ballooning, without obstructive coronary lesions on angiography. Critical illness itself may precipitate TCM, necessitating cardiac imaging and clinical evaluation in the ICU setting.
What Happened to Mr A?
Mr A received bitemporal ECT that resulted in progressive and marked improvement in his mental status. He received 9 sessions. Lorazepam was gradually weaned, and he left the ICU. Maintenance ECT was not required.
Following ECT, Mr A became more alert, interactive, and emotionally expressive. His catatonic features resolved, and he became more motivated, engaged, and emotionally stable. He was subsequently transferred to inpatient rehabilitation, where he became actively invested in his recovery.
At outpatient follow-up, Mr A was in good spirits, oriented and optimistic, and committed to strengthening his upper body function despite being wheelchair-bound. He expressed a desire to regain independence and showed insight and resilience, indicating sustained remission of his psychiatric symptoms and successful reintegration into his rehabilitation goals.
CONCLUSION
Although ECT is a highly effective treatment for several severe psychiatric conditions (eg, refractory depression, catatonia, mania, psychosis), administering ECT in ICUs presents myriad challenges, as critically ill individuals often have multiple organ dysfunction, are hemodynamically unstable, and rely on life-support measures. Clinical urgency is most often defined by the presence of life-threatening features and failure of first-line therapies. Performing ECT safely in the ICU depends upon interdisciplinary collaboration, involving psychiatrists, intensivists, anesthesiologists, and nursing staff. The informed consent process for ECT in the critically ill is shaped by varying state legal frameworks, the urgency typical of ICU settings, and the involvement of a complex multidisciplinary team. Whenever possible, the patient’s autonomy is respected by allowing them to choose the nature of treatment. However, if the patient’s abnormal mental status precludes full participation in the informed consent process, and the patient is deemed to lack the capacity to make this treatment decision, autonomy can still be honored by referencing advance care planning documents or wishes. Mortality directly attributed to ECT is exceptionally rare. When complications do occur in patients receiving ECT, the underlying mechanisms are attributed to hemodynamic alterations, the effects of anesthesia, and convulsions.
Article Information
Published Online: March 5, 2026. https://doi.org/10.4088/PCC.25f04072
© 2026 Physicians Postgraduate Press, Inc.
Submitted: September 2, 2025; accepted December 1, 2025.
To Cite: Ibrahim S, Cumming D, Robbins-Welty GA, et al. Administering electroconvulsive therapy to critically ill individuals: risks, monitoring, and benefits. Prim Care Companion CNS Disord 2026;28(2):25f04072.
Author Affiliations: Department of Psychiatry, McGill University Health Care Center, Montreal, Canada (Ibrahim); Department of Psychiatry and Psychology, Neurological Institute, Cleveland Clinic. Cleveland, Ohio (Cumming); Departments of Medicine, Psychiatry and Behavioral Sciences, and Family and Preventative Medicine, Emory School of Medicine and Grady Memorial Hospital, Atlanta, Georgia (Robbins-Welty); Tristar Centennial Medical Center, Nashville, Tennessee (Bond); Salem VA Health Care System, Salem, Virginia (Mastronardi); Wake Forest University School of Medicine, Winston-Salem, North Carolina (Dragonetti); Department of Psychiatry. Oregon Health and Science University, Portland, Oregon (Roy); Virginia Commonwealth University School of Medicine, Richmond, Virginia (Bui); Department of Psychiatry and Behavioral Sciences at Stanford University School of Medicine, Stanford, California (Salaheldin); Department of Anesthesia, Perioperative and Pain Medicine, Stanford HealthCare, Stanford, California (Lorenzo); Massachusetts General Hospital/Harvard Medical School, Boston, Massachusetts (Henry, Stern).
Corresponding Author: Samah Ibrahim, MD, Department of Psychiatry, McGill University Health Care Center, Montreal, Canada ([email protected]).
Financial Disclosure: None.
Funding/Support: None.
Clinical Points
- Electroconvulsive therapy (ECT) can be lifesaving for critically ill individuals with severe psychiatric and neuropsychiatric conditions (eg, malignant catatonia, neuroleptic malignant syndrome, refractory status epilepticus, refractory autoimmune encephalitis, and prolonged delirium with catatonic features), and it should be considered when these life-threatening neuropsychiatric syndromes are refractory to standard medical treatments.
- Some US states treat ECT like any other medical procedure, while others have introduced limitations for surrogate consent for ECT when the patient is unable to consent for themselves.
- Cardiac monitoring (eg, electrocardiography, noninvasive blood pressure monitoring, pulse oximetry) in ECT is essential.
- Risks associated with ECT can be mitigated with appropriate collaboration, education, use of safety protocols, and communication.
References (77)

- Espinoza RT, Kellner CH. Electroconvulsive therapy. N Engl J Med. 2022;386(7):667–672. PubMed CrossRef
- Dessens FM, van Paassen J, van Westerloo DJ, et al. Electroconvulsive therapy in the intensive care unit for the treatment of catatonia: a case series and review of the literature. Gen Hosp Psychiatry. 2016;38:37–41. PubMed CrossRef
- Bulteau S, Laforgue EJ, Chimot L, et al. Management of emergency electroconvulsive therapy in the intensive care unit for life-threatening psychiatric conditions: a case series. J ECT. 2018;34(1):55–59. PubMed CrossRef
- Lee HM, Kisicki MD, Hong J, et al. Importance of multispecialty coordination of care in an electroconvulsive therapy patient with musculoskeletal, neurological, and vascular injuries: a case report. J ECT. 2025;41(1):71–73. PubMed CrossRef
- Wilkinson ST, Kitay BM, Harper A, et al. Barriers to the implementation of electroconvulsive therapy (ECT): results from a nationwide survey of ECT practitioners. Psychiatr Serv. 2021;72(7):752–757. PubMed CrossRef
- Ali NA, Owiti F, Kigamwa P, et al. A qualitative exploration of gaps and challenges in knowledge and practices of electroconvulsive therapy by key personnel in public and private mental health units in Kenya. Front Psychiatry. 2019;10:697. PubMed CrossRef
- Ferrafiat V, Wachtel L, Dhossche D, et al. Catatonia is an outpatient reality. What do we do now?. Schizophr Res. 2024;264:233–235. PubMed CrossRef
- Hirjak D, Rogers JP, Wolf RC, et al. Catatonia. Nat Rev Dis Prim Springer Nat Ltd. 2024;10(1):49. PubMed CrossRef
- Smith AC, Holmes EG. Catatonia: a narrative review for hospitalists. Am J Med Open. 2023;10:100059. PubMed CrossRef
- Hirjak D, Sartorius A, Kubera KM, et al. Antipsychotic-induced motor symptoms in schizophrenic psychoses—Part 2: catatonic symptoms and neuroleptic malignant syndrome. Nervenarzt. 2019;90(1):12–24. PubMed CrossRef
- Rogers JP, Oldham MA, Fricchione G, et al. Evidence-based consensus guidelines for the management of catatonia: recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2023;37(4):327–369. PubMed CrossRef
- Arnold OH. Investigations into the question of acute fatal catatonia. Wien Z Nervenheilkd Grenzgeb. 1949;2(3):376. PubMed
- Wijdicks EFM, Ropper AH. Neuroleptic malignant syndrome. N Engl J Med. 2024;391(12):1130–1138. PubMed CrossRef
- Saijo T, Takano A, Suhara T, et al. Electroconvulsive therapy decreases dopamine D(2)receptor binding in the anterior cingulate in patients with depression: a controlled study using positron emission tomography with radioligand [(1)(1)C]FLB 457. J Clin Psychiatry. 2010;71(6):793–799. PubMed CrossRef
- Kimura M, Oda Y, Oishi K, et al. Effects of repeated electroconvulsive shocks on dopamine supersensitivity psychosis model rats. Schizophr Res. 2021;228:1–6. PubMed CrossRef
- Trollor JN, Sachdev PS. Electroconvulsive treatment of neuroleptic malignant syndrome: a review and report of cases. Aust N Z J Psychiatry. 1999;33(5):650–659. PubMed CrossRef
- Luchini F, Lattanzi L, Bartolommei N, et al. Catatonia and neuroleptic malignant syndrome: two disorders on a same spectrum? Four case reports. J Nerv Ment Dis. 2013;201(1):36–42. PubMed CrossRef
- Kuhlwilm L, Schonfeldt-Lecuona C, Gahr M, et al. The neuroleptic malignant syndrome-a systematic case series analysis focusing on therapy regimes and outcome. Acta Psychiatr Scand. 2020;142(3):233–241. PubMed CrossRef
- Dowd DK, Nunes D, Shah PD, et al. Electroconvulsive therapy for super refractory status epilepticus: a scoping review. Epilepsia. 2025;66(8):2657–2668. PubMed CrossRef
- Lambrecq V, Villega F, Marchal C, et al. Refractory status epilepticus: electroconvulsive therapy as a possible therapeutic strategy. Seizure. 2012;21(9):661–664. PubMed CrossRef
- Yang X, Wang X. Potential mechanisms and clinical applications of mild hypothermia and electroconvulsive therapy on refractory status epilepticus. Expert Rev Neurother. 2015;15(2):135–144. PubMed CrossRef
- Beatino MF, Weiss F, Torrigiani S, et al. Autoimmune encephalitis in catatonic and treatment-resistant psychotic patients referred to electroconvulsive therapy: two case reports and systematic review. J ECT. 2024;41(4):257–267. PubMed CrossRef
- Ali S, Welch CA, Park LT, et al. Encephalitis and catatonia treated with ECT. Cogn Behav Neurol. 2008;21(1):46–51. PubMed CrossRef
- Nielsen RM, Olsen KS, Lauritsen AOE, et al. Electroconvulsive therapy as a treatment for protracted refractory delirium in the intensive care unit-Five cases and a review. J Crit Care. 2014;29(5):881.e1–881.e8816. e6. PubMed
- Lupke K, Warren N, Teodorczuk A, et al. A systematic review of modified electroconvulsive therapy (ECT) to treat delirium. Acta Psychiatr Scand. 2023;147(5):403–419. PubMed CrossRef
- Gugger JJ, Saad M, Smith C. Neuroleptic-induced catatonia in two hospitalized patients. J Pharm Pract. 2012;25(2):250–254. PubMed CrossRef
- Grover S, Ghosh A, Ghormode D. Do patients of delirium have catatonic features? An exploratory study. Psychiatry Clin Neurosci. 2014;68(8):644–651. PubMed CrossRef
- Jaimes-Albornoz W, Ruiz de Pellon-Santamaria A, Nizama-Via A, et al. Catatonia in older adults: a systematic review. World J Psychiatry. 2022;12(2):348–367. PubMed
- Wilson JE, Oldham MA, Francis A, et al. Catatonia: American psychiatric association resource document. J Acad Consult Liaison Psychiatry. 2025.
- Shenai N, White CD, Azzam PN, et al. Practical and legal challenges to electroconvulsive therapy in malignant catatonia. Harv Rev Psychiatry. 2016;24(3):238–241. PubMed CrossRef
- Deng ZD, Hardesty DE, Lisanby SH, et al. Electroconvulsive therapy in the presence of deep brain stimulation implants: electric field effects. Annu Int Conf IEEE Eng Med Biol Soc. 2010;2010:2049–2052. PubMed CrossRef
- Robbins-Welty GA, Slauer RD, Brown MM, et al. Palliative electroconvulsive therapy: a descriptive cohort study. J Acad Consult Liaison Psychiatry. 2025;66(3):215–223. PubMed CrossRef
- Sexton MT, Kim A, McGonigle T, et al. In-hospital catatonia, delirium, and coma and mortality: results from the delirium and catatonia prospective cohort investigation. Schizophr Res. 2024;263:223–228. PubMed CrossRef
- Romanowicz M, Sola CL. Electroconvulsive therapy-responsive catatonia in a medically complicated patient. J ECT. 2010;26(3):234–237. PubMed CrossRef
- Oldham MA. The probability that catatonia in the hospital has a medical cause and the relative proportions of its causes: a systematic review. Psychosomatics. 2018;59(4):333–340. PubMed CrossRef
- Mankad MV BJ, Weiner RD, Krystal AD. Clinical manual of electroconvulsive therapy. American Psychiatric Publishing, Inc;2010.
- Livingston R, Wu C, Mu K, et al. Regulation of electroconvulsive therapy: a systematic review of US state laws. J ECT. 2018;34(1):60–68. PubMed CrossRef
- Beydler EM, Katzell LM, Dragonetti J, et al. Legislation limiting access to electroconvulsive therapy is increasing: a call for reasonable and evidence-based legislation. J ECT. 2025.
- Boone K, Geerts PJ, Van de Velde N, et al. Relatives’ knowledge, attitudes, and experiences toward electroconvulsive therapy: a systematic review. J ECT. 2024;41:161–173. PubMed CrossRef
- Boone K, Geerts PJ, Van de Velde N, et al. Relatives’ knowledge, attitudes, and experiences toward electroconvulsive therapy: a systematic review. J ECT. 2024.
- Tang VM, Pasricha AN, Blumberger DM, et al. Should benzodiazepines and anticonvulsants be used during electroconvulsive therapy?: a case study and literature review. J ECT. 2017;33(4):237–242. PubMed CrossRef
- Roberts DJ, Hall RI. Drug absorption, distribution, metabolism and excretion considerations in critically ill adults. Expert Opin Drug Metab Toxicol. 2013;9(9):1067–1084. PubMed CrossRef
- Kadiyala PK, Kadiyala LD. Anaesthesia for electroconvulsive therapy: an overview with an update on its role in potentiating electroconvulsive therapy. Indian J Anaesth. 2017;61(5):373–380. PubMed CrossRef
- Althagafi A. Effects of caffeine, theophylline, and aminophylline on electroconvulsive therapy: a review of evidence. J Microsc Ultrastruct. 2022;10(3):103–106. PubMed CrossRef
- Enns M, Peeling J, Sutherland GR. Hippocampal neurons are damaged by caffeine-augmented electroshock seizures. Biol Psychiatry. 1996;40(7):642–647. PubMed CrossRef
- Rasmussen KG, Zorumski CF. Electroconvulsive therapy in patients taking theophylline. J Clin Psychiatry. 1993;54(11):427–431. PubMed
- Sawayama E, Takahashi M, Inoue A, et al. Moderate hyperventilation prolongs electroencephalogram seizure duration of the first electroconvulsive therapy. J ECT. 2008;24(3):195–198. PubMed CrossRef
- Yang M, McGraw D, Ross C. Characterization of flumazenil utilization in electroconvulsive therapy. J ECT. 2025;15.
- MacPherson RD, Loo CK, Barrett N. Electroconvulsive therapy in patients with cardiac pacemakers. Anaesth Intensive Care. 2006;34(4):470–474. PubMed CrossRef
- Parikh DA, Garg SN, Dalvi NP, et al. Outcome of four pretreatment regimes on hemodynamics during electroconvulsive therapy: a double-blind randomized controlled crossover trial. Ann Card Anaesth. 2017;20(1):93–99. PubMed CrossRef
- Mizrak A, Koruk S, Ganidagli S, et al. Premedication with dexmedetomidine and midazolam attenuates agitation after electroconvulsive therapy. J Anesth. 2009;23(1):6–10. PubMed CrossRef
- Durmus I, Sezen O, Cevik B, et al. Comparison of the effects of dexmedetomidine and remifentanil on seizure duration, hemodynamics, and recovery time in electroconvulsive therapy. J ECT. 2025;20.
- Hoyer C, Kranaster L, Janke C, et al. Impact of the anesthetic agents ketamine, etomidate, thiopental, and propofol on seizure parameters and seizure quality in electroconvulsive therapy: a retrospective study. Eur Arch Psychiatry Clin Neurosci. 2014;264(3):255–261. PubMed CrossRef
- Wang A, Tsivitis A, Ma S, et al. The safety and efficacy of sugammadex for reversing neuromuscular blockade in younger children and infants. Expert Opin Drug Saf. 2024;23(7):845–853. PubMed CrossRef
- Kovac AL, Goto H, Pardo MP, et al. Comparison of two esmolol bolus doses on the haemodynamic response and seizure duration during electroconvulsive therapy. Can J Anaesth. 1991;38(2):204–209. PubMed CrossRef
- Castelli I, Steiner LA, Kaufmann MA, et al. Comparative effects of esmolol and labetalol to attenuate hyperdynamic states after electroconvulsive therapy. Anesth Analg. 1995;80(3):557–561. PubMed CrossRef
- van der Starre PJ, Lemmens HJ, Chandel A, et al. The effects of esmolol and labetalol on cerebral blood flow velocity during electroconvulsive therapy. Eur J Anaesthesiol. 2008;25(2):174–176. PubMed CrossRef
- Rogers JP, Zandi MS, David AS. The diagnosis and treatment of catatonia. Clin Med (Lond). 2023;23(3):242–245. PubMed CrossRef
- Thirthalli J, Sinha P, Sreeraj VS. Clinical practice guidelines for the use of electroconvulsive therapy. Indian J Psychiatry. 2023;65(2):258–269. PubMed CrossRef
- Patkar AA, Hill KP, Weinstein SP, et al. ECT in the presence of brain tumor and increased intracranial pressure: evaluation and reduction of risk. J ECT. 2000;16(2):189–197. PubMed CrossRef
- Buday J, Albrecht J, Mares T, et al. Brain tumors and electroconvulsive therapy: a literature overview of the last 80 years. Front Neurol. 2020;11:723. PubMed CrossRef
- Elias A, Das S, Kirkland J, et al. Safety of electroconvulsive therapy in the context of physiological and medical complexity: a state-of-the art review. PCN Rep. 2025;4(1):e70051. PubMed CrossRef
- Schnellbacher GJ, Vogt FJ, Hein M, et al. Cardiac effects of electroconvulsive therapy - a systematic review. World J Biol Psychiatry. 2025:1–15.
- Bryson EO, Popeo D, Briggs M, et al. Electroconvulsive therapy (ECT) in patients with cardiac disease: hemodynamic changes. J ECT. 2013;29(1):76–77. PubMed CrossRef
- Singh A, Kar SK. How electroconvulsive therapy works?: understanding the neurobiological mechanisms. Clin Psychopharmacol Neurosci. 2017;15(3):210–221. PubMed CrossRef
- Kerner N, Prudic J. Current electroconvulsive therapy practice and research in the geriatric population. Neuropsychiatry (London). 2014;4(1):33–54. PubMed CrossRef
- Tess AV, Smetana GW. Medical evaluation of patients undergoing electroconvulsive therapy. N Engl J Med. 2009;360(14):1437–1444. PubMed CrossRef
- Bustamante JF, Gerken P, Castro VA, et al. ECT in the general hospital: a new challenge for C-L psychiatry teams? Review of three patients treated at the ICU. J Acad Consultation-Liaison Psychiatry. 2023;64:S44–S45. CrossRef
- Sackeim HA. Modern electroconvulsive therapy: vastly improved yet greatly underused. JAMA Psychiatry. 2017;74(8):779–780. PubMed CrossRef
- Jones KC, Salemi JL, Dongarwar D, et al. Racial/ethnic disparities in receipt of electroconvulsive therapy for elderly patients with a principal diagnosis of depression in inpatient settings. Am J Geriatr Psychiatry. 2019;27(3):266–278. PubMed CrossRef
- Kaster TS, Blumberger DM, Gomes T, et al. Risk of suicide death following electroconvulsive therapy treatment for depression:A propensity score-weighted, retrospective cohort study in Canada. Lancet Psychiatry. 2022;9(6):435–446. PubMed CrossRef
- Black Parker C, McCall WV, Spearman-McCarthy EV, et al. Clinicians’ racial bias contributing to disparities in electroconvulsive therapy for patients from racial-ethnic minority groups. Psychiatr Serv. 2021;72(6):684–690. PubMed CrossRef
- Thompson JW, Weiner RD, Myers CP. Use of ECT in the United States in 1975, 1980, and 1986. Am J Psychiatry. 1994;151(11):1657–1661. PubMed CrossRef
- Thirthalli J. Underutilization of electroconvulsive therapy (ECT): a call for urgent attention. Indian J Psychol Med. 2025;47(2):103–107. PubMed CrossRef
- Duma A, Maleczek M, Panjikaran B, et al. Major adverse cardiac events and mortality associated with electroconvulsive therapy: a systematic review and meta-analysis. Anesthesiology. 2019;130(1):83–91. PubMed CrossRef
- Sharp RP, Welch EB. Takotsubo cardiomyopathy as a complication of electroconvulsive therapy. Ann Pharmacother. 2011;45(12):1559–1565. PubMed CrossRef
- Narayanan A, Russell MD, Sundararaman S, et al. Takotsubo cardiomyopathy following electroconvulsive therapy: an increasingly recognised phenomenon. BMJ Case Rep. 2014;2014:bcr2014206816. PubMed CrossRef
This PDF is free for all visitors!