Objective: Antibiotics have been used extensively by clinicians to treat patients with PANDAS or PANS (pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections and pediatric acute-onset neuropsychiatric syndrome, respectively). This review examines the best-quality evidence supporting the practice-based use of antibiotics in these psychiatric conditions.
Data Sources: PubMed was searched for English-language articles published between January 1994 and July 2017 using the search terms [PANDAS OR PANS OR new-onset pediatric OCD] AND [antibiotics OR macrolides OR beta-lactams].
Study Selection: Randomized clinical trials, observational studies, and case reports concerning antibiotic use in PANDAS/PANS were reviewed. Four publications were included in the quantitative synthesis.
Data Extraction: The evidence was rated using the GRADE (Grading of Recommendations, Assessment, Development, and Evaluation) method.
Results: Although the single studies conveyed no statistically significant results, there is some evidence for efficacy of antibiotic therapy in reducing neuropsychiatric symptoms in the PANDAS/PANS population.
Conclusions: Whereas the use of eradicating antibiotic therapy during active infections in PANDAS/PANS is well established, there is still a need for studies that improve the quality of evidence supporting use of antibiotics in this population independent of ongoing infections. Studies that compare antibiotics with other therapies, as well as studies that assess safety and efficacy of long-term use of antibiotic therapy in PANDAS/PANS, are still lacking. However, the available research supports evidence of a subgroup in the pediatric OCD population that is sensitive to antibiotic treatment and immunomodulatory therapy, independent of ongoing infectious conditions. Thus, more studies are warranted in the overall OCD spectrum.
Antibiotics for PANDAS?
Limited Evidence: Review and Putative Mechanisms of Action
ABSTRACT
Objective: Antibiotics have been used extensively by clinicians to treat patients with PANDAS or PANS (pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections and pediatric acute-onset neuropsychiatric syndrome, respectively). This review examines the best-quality evidence supporting the practice-based use of antibiotics in these psychiatric conditions.
Data Sources: PubMed was searched for English-language articles published between January 1994 and July 2017 using the search terms [PANDAS OR PANS OR new-onset pediatric OCD] AND [antibiotics OR macrolides OR beta-lactams].
Study Selection: Randomized clinical trials, observational studies, and case reports concerning antibiotic use in PANDAS/PANS were reviewed. Four publications were included in the quantitative synthesis.
Data Extraction: The evidence was rated using the GRADE (Grading of Recommendations, Assessment, Development, and Evaluation) method.
Results: Although the single studies conveyed no statistically significant results, there is some evidence for efficacy of antibiotic therapy in reducing neuropsychiatric symptoms in the PANDAS/PANS population.
Conclusions: Whereas the use of eradicating antibiotic therapy during active infections in PANDAS/PANS is well established, there is still a need for studies that improve the quality of evidence supporting use of antibiotics in this population independent of ongoing infections. Studies that compare antibiotics with other therapies, as well as studies that assess safety and efficacy of long-term use of antibiotic therapy in PANDAS/PANS, are still lacking. However, the available research supports evidence of a subgroup in the pediatric OCD population that is sensitive to antibiotic treatment and immunomodulatory therapy, independent of ongoing infectious conditions. Thus, more studies are warranted in the overall OCD spectrum.
Prim Care Companion CNS Disord 2018;20(3):17r02232
To cite: Burchi E, Pallanti S. Antibiotics for PANDAS? Limited evidence: review and putative mechanisms of action. Prim Care Companion CNS Disord. 2018;20(3):17r02232.
To share: https://doi.org/10.4088/PCC.17r02232
© Copyright 2018 Physicians Postgraduate Press, Inc.
aDepartment of Scienze della Salute, University of Florence, Italy
bDepartment of Psychiatry and Behavioral Sciences, Albert Einstein College of Medicine, Bronx, New York
cDepartment of Psychiatry and Behavioral Sciences, Stanford University Medical Center, Stanford, California
dInstitute of Neuroscience, Florence, Italy
*Corresponding author: Elisabetta Burchi, MD, Department of Neurofarba, University of Florence, AOU Careggi, Vle della maternità Pad 8b, 50141, Florence, Italy ([email protected]).
The link between neuropsychiatric symptoms and infections was first elucidated in the early 1990s.1,2 These findings led to the definition of PITANDS (pediatric infection–triggered autoimmune neuropsychiatric disorders)3 and then to the more specific PANDAS (pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections) proposed as an entity with abrupt prepubertal onset of obsessive-compulsive disorder (OCD) and tic symptoms occurring in association with group A streptococcus (GAS) infections.4 However, the difficulty in operationalizing the association between GAS infection and onset and exacerbations of OCD and tic symptoms led to the definition of a new umbrella category called PANS (pediatric acute-onset neuropsychiatric syndrome).5 Although this category was developed to include acute-onset OCD cases that were not triggered by infections, the largest survey sample6 conducted on the PANS population to date showed that infection was implicated as the primary inciting factor in 65% of patients, and streptococcus was associated in 54% of patients. A recent study,7 the first to assess prevalence rates of PANDAS within a pediatric OCD sample (N = 136), found that 5% of pediatric OCD outpatients met the criteria for PANS or PANDAS, suggesting that this association is a relevant health issue.
Despite the controversy about the PANDAS hypothesis on pathogenesis,8 given the strong link between neuropsychiatric symptoms and streptococcus in patients with PANDAS, many clinicians use antibiotics in this population. According to the National Institute of Mental Health website,9 “the best treatment for acute episodes of PANDAS is to eradicate the strep infection causing the symptoms if it is still present” and “it may be helpful to use antibiotics as prophylaxis against strep infections,” considering the beneficial outcome observed in patients with rheumatic fever and Sydenham chorea.
Recently, the PANS/PANDAS Consortium proposed a set of practice-based guidelines10 for patient management that suggests an initial course of antibiotics for all PANS cases and secondary antimicrobial prophylaxis of streptococcal infections for children with PANDAS who have severe neuropsychiatric symptoms or recurrent exacerbations. Although there is accumulating evidence11,12 that immune dysregulation may contribute to the pathophysiology of not only PANDAS but also OCD as a whole, immunomodulatory therapies still have to be sufficiently investigated in the overall OCD population.
We reviewed the literature to assess levels of evidence for the use of antibiotics in the young OCD population and, specifically, in PANDAS/PANS patients, taking into account the method suggested by the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE)13 working group.
METHODS
A systematic review of PubMed was performed between January 1994 and July 2017 using the search terms [PANDAS OR PANS OR new-onset pediatric OCD] AND antibiotics OR macrolides OR beta-lactams (Figure 1). The search was limited to English-language articles. Randomized clinical trials (RCTs), observational studies, and case reports describing the efficacy of antibiotics in definitive or putative PANDAS or PANS patients were reviewed.
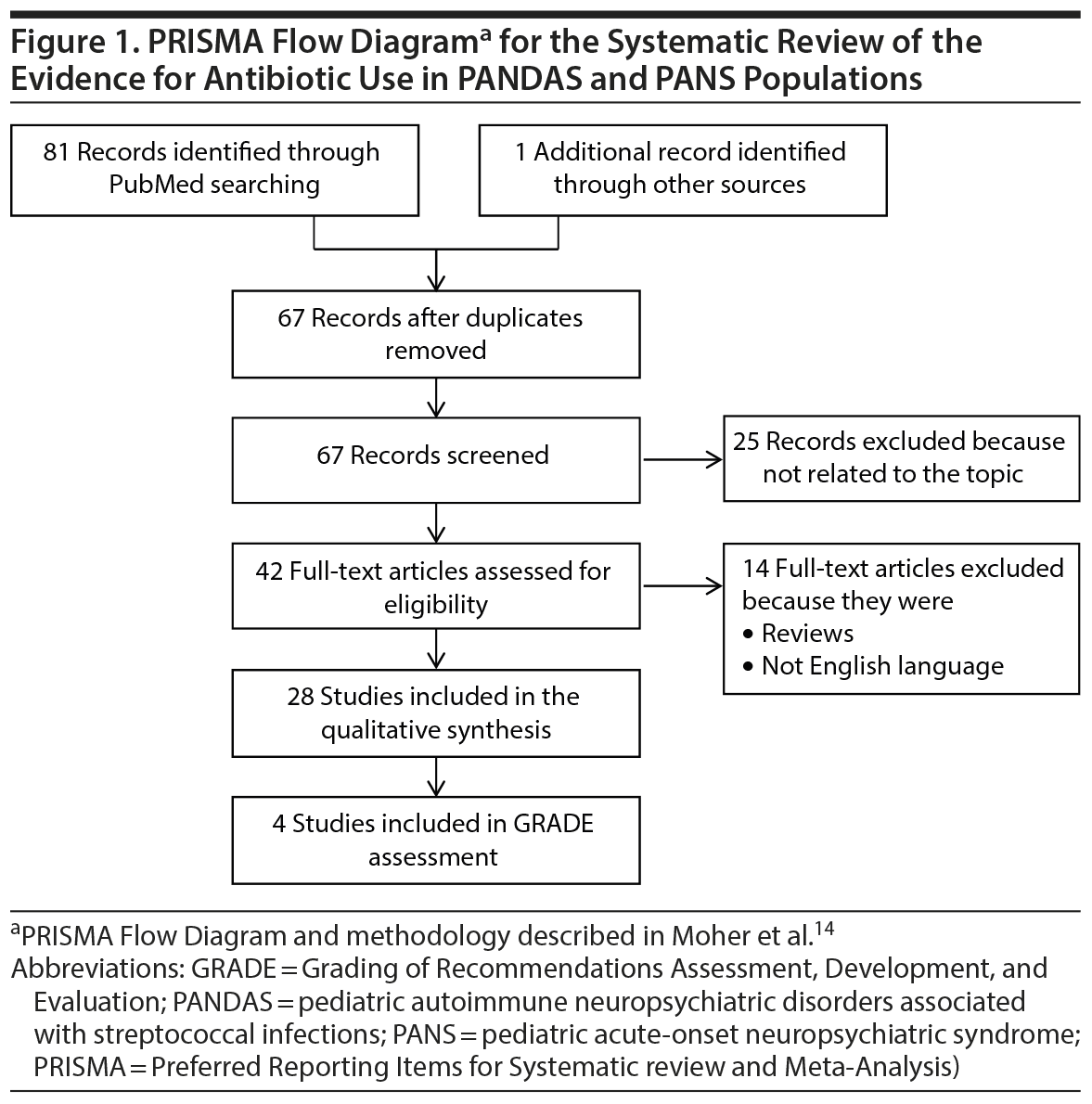
Studies that did not report a comparison group or objective measures of effect were excluded from the quantitative synthesis. Studies were rated independently by 2 researchers (E.B. and S.P.) using the GRADE method, which systematically analyzes the factors important to interpret the quality of evidence—study limitations, inconsistency, indirectness, imprecision, publication bias—and the magnitude of effect for each outcome considered.13 Following this approach, the quality of evidence is rated high, moderate, low, or very low. Twenty-eight studies were included in the qualitative analysis, 415–18 of which were included in the GRADE assessment.
RESULTS
PANDAS and Antibiotic Therapy
RCTs. To date, there are 2 published RCTs15,16 studying antibiotic efficacy in children with streptococcal-triggered OCD and tic disorders, specifically proposing as primary outcome measures the reduction in the number of both GAS reinfections and neuropsychiatric exacerbations in patients previously diagnosed with PANDAS. Garvey et al15 failed to provide support for the use of a 4-month prophylaxis with penicillin, with no significant differences in the number of infections or in ratings of tic or OCD severity between the active and placebo arms. Nevertheless, the results described relative efficacy of antibiotic compared to placebo, and the lack of statistical significance could be influenced by the carryover effect related to the crossover design, use of off-study antibiotics, and the subclinical score of OCD symptoms at baseline.15 Conversely, Snider et al16 reported statistically significant reductions in GAS infections and neuropsychiatric exacerbations in PANDAS patients treated for 1 year with prophylaxis with penicillin or azithromycin compared to the previous year. However, the effect size of antibiotics could have been overestimated due to the risk of bias with the study design, including lack of placebo control, the regression toward the mean phenomenon, and retrospective collection of the baseline year data.16 Interestingly, the observed rate of neuropsychiatric exacerbations in the intervention group (0.5 per person-year for those treated with penicillin and 0.9 for those treated with azithromycin)16 was similar to the rate reported in a case-control study19 comparing PANDAS with classic OCD in which children who met criteria for PANDAS had 0.56 exacerbations per person-year (versus 0.28 exacerbations per person-year for control subjects) but in the absence of prophylactic antibiotic treatment.
Observational studies. Murphy and Pichichero20 identified and followed 12 new-onset PANDAS cases among approximately 4,000 patients infected with GAS across a 3-year time period and evaluated antibiotic therapy in relation to acute GAS tonsillopharyngitis at the index episode and during recurrences. In every instance, PANDAS behavior was associated with evidence of acute GAS infection, and an early intervention with eradicating antibiotic therapy corresponded to remission of neuropsychiatric symptoms in 12 of 12 PANDAS cases. Despite the lack of a comparison group, which prevented inclusion in the current quantitative synthesis, this study20 conveys important clinical significance given the magnitude of the intervention in relation to neuropsychiatric symptoms during acute PANDAS episodes. Other studies19,21–24 supported the role of untreated GAS infection in triggering OCD and tic disorders in a subgroup of the pediatric population, also suggesting the preventive therapeutic role of eradicating antibiotic therapy.
In a sample of 109 children with tics or OCD, Murphy et al25 found that those diagnosed with PANDAS (n = 41) were significantly more likely to have had a remission of neuropsychiatric symptoms during antibiotic therapy than those without PANDAS (relative risk = 3.98). No dose or type of antibiotic was specified.25
Case reports. The first poststreptococcal case of OCD reporting use of antibiotics appeared in 1996.26 Since then, other case reports27–37 have been published describing patients with PANDAS prescribed antibiotics at the index episode. In every instance, the antibiotics β-lactams or macrolides were used.
PANS and Antibiotic Therapy
RCTs. Murphy and collegues17 conducted a placebo-controlled trial examining the preliminary efficacy and tolerability of 4-week treatment with azithromycin in patients with PANS; 61% met criteria for PANDAS. Both study arms showed improvement during the trial. There was no significant difference in the Children’s Yale-Brown Obsessive Compulsive Scale (CY-BOCS) scores, but OCD severity on the Clinical Global Impressions-Severity of Illness scale was significantly reduced in the azithromycin group compared to the placebo group. Greater tic severity was associated with enhanced treatment response. The azithromycin group showed a potential for cardiac risk, with a trend toward significantly greater electrocardiography corrected QT interval (QTc) (P = .060) at the end of week 4.17
Observational studies and case reports. In a retrospective survey sample6 that included 698 patients with clinical diagnoses of PANS, 90% of patients with infection-associated onset (65% of the total) reported receiving antibiotics for the infection, with 59% having a resolution of infection and 31% having a resolution of PANS symptoms following this intervention. Some case reports36–41 described efficacy of antibiotic therapy in infection-associated PANS cases.
New-Onset Pediatric OCD and Antibiotic Therapy
Our query identified an additional RCT18 that was included in the quantitative synthesis, although it did not include participants who strictly met PANDAS or PANS criteria. This study18 evaluated the efficacy of a cephalosporin (cefdinir) in a sample of patients comparable to the PANS population. Murphy et al18 examined the safety and efficacy of cefdinir (a third-generation cephalosporin) in the treatment of children aged 4–13 years old with new-onset OCD or tics. No evidence of infection was required. Although no significant group differences were detected between the cefdinir and placebo groups, large within-group treatment effects were observed in the cefdinir group using the CY-BOCS and Yale Global Tic Severity Scale (YGTSS), identifying youth with tics as a subgroup more sensitive to antibiotic treatment. The effect size of the intervention could have been underestimated given that only a small number of subjects were not receiving antibiotics in the placebo arm.18
DISCUSSION
To date, only 4 studies15–18 yield objective measures of effect provided by antibiotic therapy in reducing neuropsychiatric symptoms in pediatric OCD samples. These studies are heterogeneous, differing in type of antibiotic used, duration of treatment, selection criteria of participants, study design, and primary outcome measures. Using the GRADE approach13 and distinguishing statistical significance from magnitude of effect, we stratified these studies according to 3 main outcomes: reduction in OCD symptom severity, reduction in tic symptom severity, and reduction in number of neuropsychiatric exacerbations using the CY-BOCS and the YGTSS (Table 1).
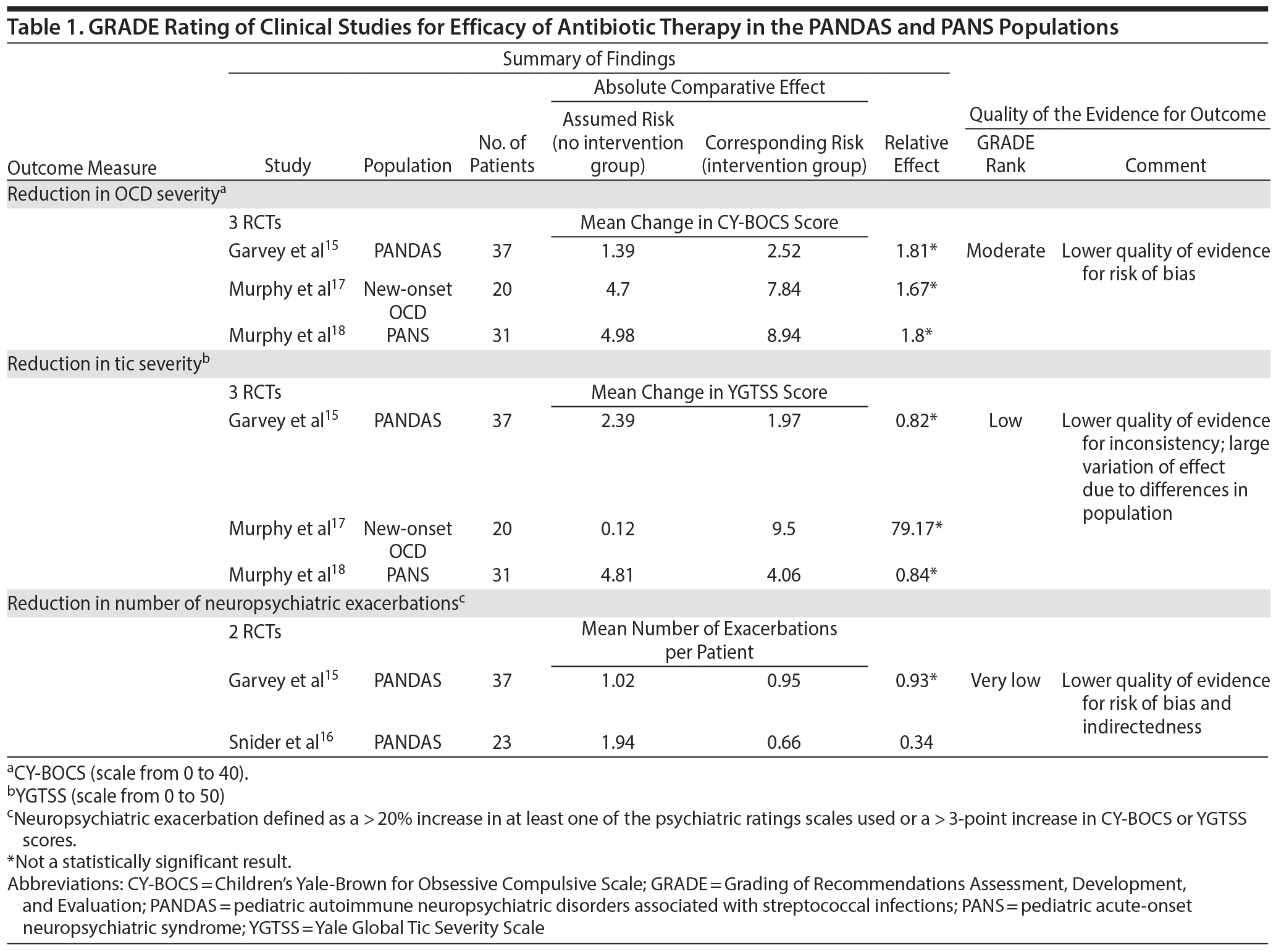
Despite the heterogeneity across studies that prevented a meta-analysis of results, collectively the 4 studies15–18 suggest some benefit from antibiotic treatment in the PANDAS/PANS population. Even if the quality of the evidence for the outcomes considered was not highly rated (Table 1), the strength of recommendation for the use of antibiotics in this population, which would depend on the balance between desirable and undesirable effects, is not compromised.
Antibiotics and Mechanisms for
Treatment Efficacy in PANDAS/PANS
On the basis of the immune-driven hypothesis for pathogenesis of PANDAS42–45 and given the results of studies46,47 describing mechanisms of action of antibiotics, we can hypothesize some possible mechanisms for treatment efficacy of antibiotics in this clinical population that supersede their anti-infective activities.
Immune-Driven Pathogenesis
Some preclinical studies48,49 have tried to elucidate the pathogenesis of PANDAS. Yaddanapudi et al48 found a strict relationship between GAS exposure and the syndrome, demonstrating that passive transfer of streptococcus-induced antibodies reproduces behavioral disturbances in a mouse model of PANDAS. Lotan and colleagues49 tested the efficacy of ampicillin treatment in a rat model of GAS-related neuropsychiatric disorders comparing controls and GAS-exposed rats in the ampicillin condition. The findings showed that ampicillin prevented some of the behavioral alterations induced by GAS-antigen exposure, IgG (immunoglobulin G)deposition in the thalamus, and the increase in the level of TH (tyrosine hydroxylase) and D1 and D2 receptors in the striatum without reducing the level of sera anti-GAS antibodies.49
However, in humans, the pathogenesis is thought to be more complicated, and it seems that PANDAS represents a subgroup of patients with tics or OCD whose symptoms are vulnerable to GAS infection, although not exclusively. The proposed pathogenesis of PANDAS implicates the production of antibodies in response to GAS infection, which cross the blood-brain barrier, and, due to molecular mimicry, cross-react with neuronal proteins that interfere with neuronal signaling.42–45 According to this hypothesis, a patient’s immunogenetics as a whole—through specific human leukocyte antigens, type of T response, specificity, affinity, and concentration of the autoantibodies induced—become critical in eventually determining the disorder.
Recent preclinical studies45 have provided insight into the link between the central nervous system (CNS) and cellular immunity. In particular, in a mouse model, Dileepan et al45 illustrated the neuroimmune consequences of repeated intranasal GAS infections, with the pivotal role of Th17 cellular response in promoting blood-brain barrier breakdown, IgG deposition, microglial activation, and loss of excitatory synaptic proteins. This study45 suggested that in children with PANDAS, once autoantibody levels are primed by previous GAS infections, flare-ups may be produced by other common pathogens including the influenza virus, mycoplasma, and staphylococcus aureus that are able to induce a robust Th17 cellular response.50,51
This hypothesis might explain why subjects with new-onset first-episode OCD are more likely to respond to antibiotic treatment than those with a recurrent OCD episode17 and why only a minority (7.5%–25%) of the clinical exacerbations in a PANDAS population had evidence of an association with group A beta-hemolytic streptococcal infections infections.19
β-Lactams and Macrolides: Actions on CNS
β-Lactams and macrolides are the classes of antibiotics studied in PANDAS15,16 and PANS17 because they are the antibiotics recommended for eradication of GAS infections. In addition to the anti-infective activities, they provide other ancillary effects46,47 that may convey part of the treatment benefit reported in the PANDAS/PANS populations (Table 2).
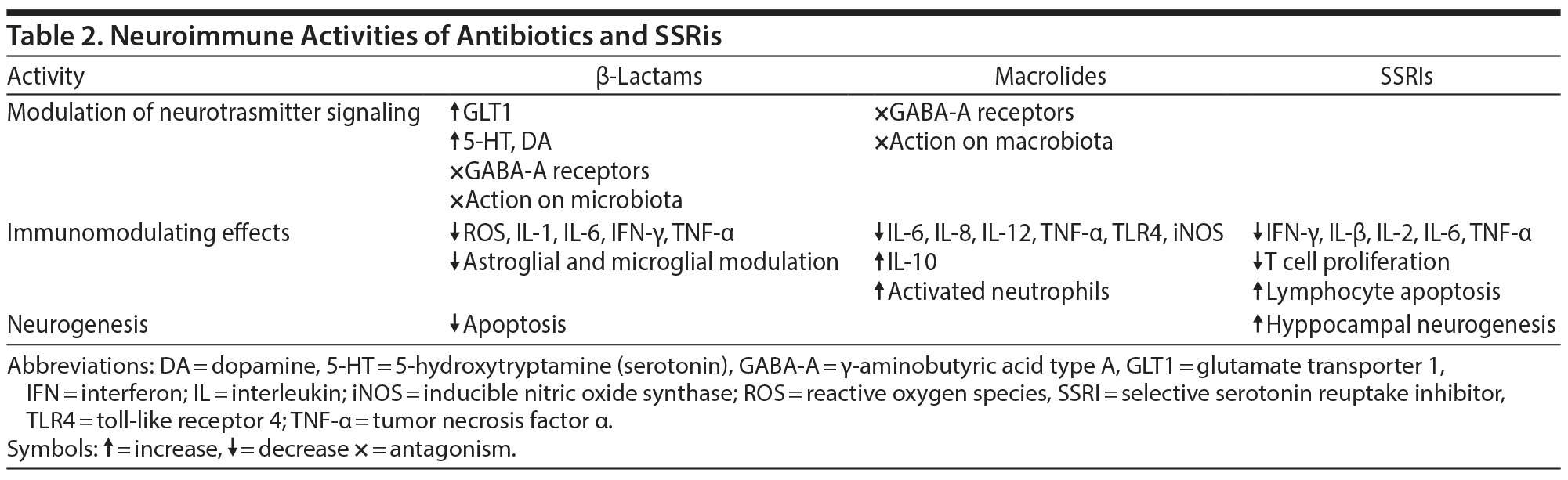
β-Lactam molecules have neuroprotective, antioxidant, analgesic, and immunomodulatory capabilities.46,52,53 They have been found to facilitate serotonin or dopamine release, to block γ-aminobutyric acid (GABA)-A receptors, to modulate astroglia and microglia, to down-regulate reactive oxygen, and to have anti-inflammatory properties. Notably, β-lactam molecules minimize glutamate neurotoxicity, increasing expression of glutamate transporter 1 GLT1 by activation of promoter for GLT1 in a concentration-dependent manner.53
While GLT1 dysfunction is implicated in several neurologic disorders,52 increasing evidence also suggests a dysregulation of glutamatergic signaling in patients with OCD,54 and 1 study45 raises the possibility that synaptic pruning of glutamatergic connections is increased in PANDAS.
Although ceftriaxone has been preferred in vivo as a neuroprotectant against many of glutamate/GLT1-dependent neurologic diseases because of a large volume of distribution, penicillin also has been shown to be highly active in stimulating GLT1 protein expression in vivo.53
Clavulanate, a β-lactamase inhibitor commonly used in conjunction with amoxicillin, readily crosses the blood-brain barrier. Through reductions in glutamate and enhancements in dopamine and trophic factors, clavulanate has shown significant potential as an antidepressant and anxiolytic agent.52
The 14- and 15-membered ring macrolides are known for their immunomodulating properties that seem exceptionally suited for the treatment of chronic inflammatory diseases.47 These effects are permitted by their capacity to concentrate in lymphocytic cells, where they have a dampening effect on the proinflammatory response.
Azithromycin, which has poor CNS penetration, is known to accumulate in leukocytes to a considerably higher degree than any other macrolide antibiotic. Recent findings55 suggest that it might be the macrolide of choice in the treatment of inflammasome-driven diseases in dose regimens lower than that used in antimicrobial chemotherapy. The efficacy of azithromycin during GAS infection and possibly during postinfection exacerbations seen in PANDAS could also be related to its impact on innate immunity, reducing TLR4 (toll-like receptor 4) and IL-12 (interleukin 12). TLR4 mutations to D299G and T399I were associated with vulnerability to recurrent GAS infection.56
Antibiotics may provide other indirect effects on cerebrospinal fluid through action on the composition of the commensal microbiota,57 which is involved in the production of neurotransmitters, gut hormone release and hypothalamic-pituitary-adrenal axis, and cytokine release and in the control of Th17/Treg (T helper 17 cells and regulatory T cells) balance.58
In brief, antibiotic efficacy in the PANDAS/PANS population could supersede the direct effect on GAS and may also be conveyed by immunomodulatory and neuroprotective effects—preventing the formation of autoantibodies, the expansion of Th17 response, the disruption of blood-brain barrier, or the activation of microglia—and by the action on the microbiota.
Safety of Antibiotic Treatment
With regard to β-lactams,59 they all act as GABA-A antagonists in a dose-dependent manner, an effect which may be potentiated by nonsteroidal anti-inflammatory drugs. This effect is not noticeable in concentrations within the therapeutic range, but patients with renal impairment or comorbidities, which may lower the seizure threshold, would be at risk. With regard to macrolides,59 clarithromycin is known to cause drug-induced mania, and azithromycin has been found to have a neurotoxic dose-dependent effect related to action on GABA-A receptors.
Other concerns with use of macrolides for long-term treatment are the QTc increase, which is based on peak concentration60 and duration of treatment,17 and risk of development of resistance.61 Although therapy with macrolides is indicated in some chronic inflammatory diseases, an increase of macrolide-resistant bacteria occurred as a result of long-term, low-dose use in some cases.62 A study in Italy63 identified that 32% of GAS isolates exhibit resistance to macrolides. Macrolide resistance during the 1990s in other European countries was between 1% and 7%.63 Macrolide resistance could be associated with serious consequences such as acute rheumatic fever, therefore patients should be monitored.61
Prevention of GAS Infection
Considering the pathogenetic role of GAS in the index episode of PANDAS, as well as the association with other neuropsychiatric disorders,64 prevention of GAS infections would obviously represent a crucial therapeutic intervention. Although GAS vaccines have been defined as impeded vaccines mainly for the complex epidemiology of GAS infections, the worldwide prevalence of severe GAS disease—approximately 18.1 million cases65—has led to efforts to develop a vaccine against pharyngitis and impetigo that should also prevent acute rheumatic fever, rheumatic heart disease, and serious invasive disease. A review66 outlined a clinical development strategy for the registration of a candidate vaccine for GAS pharyngitis, and one has been proposed recently as a vaccine candidate.67
Antibiotics Versus Other
Treatment Options in PANDAS/PANS
We found no studies comparing antibiotic therapy to other therapies in the young OCD population. Research concerning immune-based treatments is particularly relevant in the PANDAS OCD subtype, which has been found to be at elevated risk for behavioral activation following selective serotonin reuptake inhibitor (SSRI) initiation.68
Recently, Williams and colleagues69 failed to demonstrate superiority of intravenous immunoglobulin over placebo.70 To date, RCTs assessing the symptomatic efficacy of SSRIs or comparing SSRIs with other treatments in the PANDAS population are lacking as are RCTs comparing the effect of different SSRIs on immune biomarkers. To our knowledge, only 1 study72 was proposed with the objective of determining the safety and efficacy of SSRI + antibiotic compared to SSRI + placebo for the treatment of OCD symptoms in patients with PANDAS, but it was not completed due to lack of funds.
With regard to cognitive-behavioral therapy (CBT), even if cognitive-behavioral intervention for OCD symptoms in the PANDAS subtype demonstrated promising preliminary efficacy,72 CBT does not appear to prevent a full episode of GAS-triggered exacerbations. Nadeau et al73 conducted the first pilot trial of CBT augmentation in youth meeting criteria for PANS and presenting with an incomplete response to antibiotic treatment. Subjects received a minimum of 4 weeks of antibiotic treatment and had a CY-BOCS total score > 16. Relative to pretreatment, youth who completed CBT in conjunction with antibiotic treatment showed significant reductions on the CY-BOCS of 49% and 50% at posttreatment and follow-up, respectively. This open trial73 showed a great effect size of the combined therapy in a resistant population but did not utilize a control condition, thus it is unclear to what extent continuation of antibiotic treatment accounted for OCD symptom reductions.
CONCLUSIONS
Antibiotics Versus
No Antibiotics in PANDAS/PANS
The objective of this review was to rate the quality of the best-available evidence for efficacy of antibiotic therapy in reducing OCD and tic symptom severity and number of neuropsychiatric exacerbations in the PANDAS/PANS population. The review did not intend to support recommendations that reflect the extent to which a guideline panel is confident that desirable effects of an intervention outweigh undesirable effects.
Given the available evidence and following the GRADE approach, we are moderately confident in the effect estimate for efficacy of antibiotics in reducing OCD symptoms in the PANDAS/PANS population. However, our confidence in the effect estimate is limited for reduction of tic symptoms and very limited with regard to reduction in number of neuropsychiatric exacerbations.
Further Studies
Despite the identification of PANDAS dating back to 1998,4 to date there are only 4 RCTs15–18 studying the efficacy of antibiotic therapy in this population. Beyond the recommendation to assess the presence of infection when PANS is suspected8 and eradicate infection when present,10 conclusive studies that assess superiority of antibiotic versus placebo or other therapies in the PANDAS/PANS population are still lacking. However, the available studies suggest the existence of a pediatric OCD subtype sensitive to antibiotic treatment, independent of evidence of infection.17,18
These results, along with the poor remission rates associated with standard therapies provided by the largest multisite RCT74 conducted in a general pediatric OCD population, support the existence of different subtypes of pediatric OCD, particularly a subgroup characterized by sensitivity to immunomodulatory therapies.
It is probable that neuroinflammation or autoimmunity occur not just in childhood OCD cases, but also in adult OCD. A recent study12 strongly supported the neuroinflammatory theory of OCD that goes beyond the involvement of basal ganglia to include the cortico-striato-thalamo-cortical circuit. If neuroinflammation is involved in most OCD cases, immunomodulatory therapies, including antibiotic agents used as neuromodulators and immunomodulators, should be also investigated in adult OCD.
To improve the quality of studies concerning the efficacy of antibiotic treatment in the PANDAS/PANS population and to identify an immunotherapy-susceptible subgroup in the overall OCD population, RCTs in youth with new-onset tic and OCD symptoms and in adults with OCD are needed. These RCTs should be of sufficient duration of time and compare prophylactic antibiotic treatment (both at eradicating and low doses) with placebo, SSRIs, CBT, and other immunomodulatory therapies.
Submitted: October 13, 2017; accepted November 20, 2017.
Published online: May 3, 2018.
Potential conflicts of interest: None.
Funding/support: None.
REFERENCES
1. Murphy TK, Kurlan R, Leckman J. The immunobiology of Tourette’s disorder, pediatric autoimmune neuropsychiatric disorders associated with streptococcus, and related disorders: a way forward. J Child Adolesc Psychopharmacol. 2010;20(4):317–331. PubMed CrossRef
2. Hornig M. The role of microbes and autoimmunity in the pathogenesis of neuropsychiatric illness. Curr Opin Rheumatol. 2013;25(4):488–795. PubMed CrossRef
3. Allen AJ, Leonard HL, Swedo SE. Case study: a new infection-triggered, autoimmune subtype of pediatric OCD and Tourette’s syndrome. J Am Acad Child Adolesc Psychiatry. 1995;34(3):307–311. PubMed CrossRef
4. Swedo SE, Leonard HL, Garvey M, et al. Pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections: clinical description of the first 50 cases. Am J Psychiatry. 1998;155(2):264–271. PubMed CrossRef
5. Swedo SE, Leckman JF, Rose NR. From Research Subgroup to Clinical Syndrome: modifying the PANDAS criteria to describe PANS (pediatric acute-onset neuropsychiatric syndrome). Pediatr Therapeut. 2012;2:113. CrossRef
6. Calaprice D, Tona J, Parker-Athill EC, et al. A survey of pediatric acute-onset neuropsychiatric syndrome characteristics and course. J Child Adolesc Psychopharmacol. 2017;27(7):607–618. PubMed CrossRef
7. Jaspers-Fayer F, Han SHJ, Chan E, et al. Prevalence of acute-onset subtypes in pediatric obsessive-compulsive disorder. J Child Adolesc Psychopharmacol. 2017;27(4):332–341. PubMed CrossRef
8. Chiarello F, Spitoni S, Hollander E, et al. An expert opinion on PANDAS/PANS: highlights and controversies. Int J Psychiatry Clin Pract. 2017;21(2):91–98. PubMed CrossRef
9. National Institute of Mental Health. Guidelines published for treating PANS/PANDAS. https://www.nimh.nih.gov/news/science-news/2017/guidelines-published-for-treating-pans-pandas.shtml. Accessed April 10, 2018.
10. Cooperstock MS, Swedo SE, Pasternack MS, et al; and for the PANS/PANDAS Consortium. Clinical management of pediatric acute-onset neuropsychiatric syndrome: part III: treatment and prevention of infections. J Child Adolesc Psychopharmacol. 2017;27(7):594–606. CrossRef.
11. Teixeira L, Rodrigues DH, Marques AH, et al. Searching for the immune basis of obsessive-compulsive disorder. Neuroimmunomodulation. 2014;21(2–3):152–158. PubMed CrossRef
12. Attwells S, Setiawan E, Wilson AA, et al. Inflammation in the neurocircuitry of obsessive-compulsive disorder. JAMA Psychiatry. 2017;74(8):833–840. PubMed CrossRef
13. Guyatt GH, Oxman AD, Vist GE, et al; GRADE Working Group. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650):924–926. PubMed CrossRef
14. Moher D, Liberati A, Tetzlaff J, et al; PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. PubMed CrossRef
15. Garvey MA, Perlmutter SJ, Allen AJ, et al. A pilot study of penicillin prophylaxis for neuropsychiatric exacerbations triggered by streptococcal infections. Biol Psychiatry. 1999;45(12):1564–1571. PubMed CrossRef
16. Snider LA, Lougee L, Slattery M, et al. Antibiotic prophylaxis with azithromycin or penicillin for childhood-onset neuropsychiatric disorders. Biol Psychiatry. 2005;57(7):788–792. PubMed CrossRef
17. Murphy TK, Brennan EM, Johnco C, et al. A double-blind randomized placebo-controlled pilot study of azithromycin in youth with acute-onset obsessive-compulsive disorder. J Child Adolesc Psychopharmacol. 2017;27(7):640–651. PubMed CrossRef
18. Murphy TK, Parker-Athill EC, Lewin AB, et al. Cefdinir for recent-onset pediatric neuropsychiatric disorders: a pilot randomized trial. J Child Adolesc Psychopharmacol. 2015;25(1):57–64. PubMed CrossRef
19. Kurlan R, Johnson D, Kaplan EL; Tourette Syndrome Study Group. Streptococcal infection and exacerbations of childhood tics and obsessive-compulsive symptoms: a prospective blinded cohort study. Pediatrics. 2008;121(6):1188–1197. PubMed CrossRef
20. Murphy ML, Pichichero ME. Prospective identification and treatment of children with pediatric autoimmune neuropsychiatric disorder associated with group A streptococcal infection (PANDAS). Arch Pediatr Adolesc Med. 2002;156(4):356–361. PubMed CrossRef
21. Mell LK, Davis RL, Owens D. Association between streptococcal infection and obsessive-compulsive disorder, Tourette’s syndrome, and tic disorder. Pediatrics. 2005;116(1):56–60. PubMed CrossRef
22. Schrag A, Gilbert R, Giovannoni G, et al. Streptococcal infection, Tourette syndrome, and OCD: is there a connection? Neurology. 2009;73(16):1256–1263. PubMed CrossRef
23. Wang HC, Lau CI, Lin CC, et al. Streptococcal infections are associated with increased risk of pediatric neuropsychiatric disorders: a Taiwanese population-based cohort study. J Clin Psychiatry. 2016;77(7):e848–e854. PubMed CrossRef
24. Perrin EM, Murphy ML, Casey JR, et al. Does group A beta-hemolytic streptococcal infection increase risk for behavioral and neuropsychiatric symptoms in children? Arch Pediatr Adolesc Med. 2004;158(9):848–856. PubMed CrossRef
25. Murphy TK, Storch EA, Lewin AB, et al. Clinical factors associated with pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections. J Pediatr. 2012;160(2):314–319. PubMed CrossRef
26. Tucker DM, Leckman JF, Scahill L, et al. A putative poststreptococcal case of OCD with chronic tic disorder, not otherwise specified. J Am Acad Child Adolesc Psychiatry. 1996;35(12):1684–1691. PubMed CrossRef
27. Snider LA, Swedo SE. Childhood-onset obsessive-compulsive disorder and tic disorders: case report and literature review. J Child Adolesc Psychopharmacol. 2003;13(suppl 1):S81–S88. PubMed CrossRef
28. Chmelik E, Awadallah N, Hadi FS, et al. Varied presentation of PANDAS: a case series. Clin Pediatr (Phila). 2004;43(4):379–382. PubMed CrossRef
29. van Toorn R, Weyers HH, Schoeman JF. Distinguishing PANDAS from Sydenham’s chorea: case report and review of the literature. Eur J Paediatr Neurol. 2004;8(4):211–216. PubMed CrossRef
30. Dranitzki Z, Steiner I. PANDAS in siblings: a common risk? Eur J Neurol. 2007;14(6):e4. PubMed CrossRef
31. Fonseca L, Guerra J, Neves N, et al. PANDAS (Pediatric Autoimmune Neuropsychiatric Disorders Associated With Streptococcal Infection): a case report. J Neuropsychiatry Clin Neurosci. 2010;22(3):352f.e9–e10. PubMed CrossRef
32. Maguire GA, Viele SN, Agarwal S, et al. Stuttering onset associated with streptococcal infection: a case suggesting stuttering as PANDAS. Ann Clin Psychiatry. 2010;22(4):283–284. PubMed
33. Alexander AA, Patel NJ, Southammakosane CA, et al. Pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections (PANDAS): an indication for tonsillectomy. Int J Pediatr Otorhinolaryngol. 2011;75(6):872–873. PubMed CrossRef
34. Lewin AB, Storch EA, Murphy TK. Pediatric autoimmune neuropsychiatric disorders associated with Streptococcus in identical siblings. J Child Adolesc Psychopharmacol. 2011;21(2):177–182. PubMed CrossRef
35. Das A, Radhakrishnan A. A case of PANDAS with Kleine-Levin type periodic hypersomnia. Sleep Med. 2012;13(3):319–320. PubMed CrossRef
36. Toufexis MD, Hommer R, Gerardi DM, et al. Disordered eating and food restrictions in children with PANDAS/PANS. J Child Adolesc Psychopharmacol. 2015;25(1):48–56. PubMed CrossRef
37. Frankovich J, Thienemann M, Rana S, et al. Five youth with pediatric acute-onset neuropsychiatric syndrome of differing etiologies. J Child Adolesc Psychopharmacol. 2015;25(1):31–37. PubMed CrossRef
38. Sokol MS, Gray NS. Case study: an infection-triggered, autoimmune subtype of anorexia nervosa. J Am Acad Child Adolesc Psychiatry. 1997;36(8):1128–1133. PubMed CrossRef
39. Sokol MS. Infection-triggered anorexia nervosa in children: clinical description of four cases. J Child Adolesc Psychopharmacol. 2000;10(2):133–145. PubMed CrossRef
40. Calkin CV, Carandang CG. Certain eating disorders may be a neuropsychiatric manifestation of PANDAS: case report. J Can Acad Child Adolesc Psychiatry. 2007;16(3):132–135. PubMed
41. Ercan TE, Ercan G, Severge B, et al. Mycoplasma pneumoniae infection and obsessive-compulsive disease: a case report. J Child Neurol. 2008;23(3):338–340. Published online Dec 13, 2007. PubMed CrossRef
42. Kirvan CA, Swedo SE, Heuser JS, et al. Mimicry and autoantibody-mediated neuronal cell signaling in Sydenham chorea. Nat Med. 2003;9(7):914–920. PubMed CrossRef
43. Doyle F, Cardoso F, Lopes L, et al. Infusion of Sydenham’s chorea antibodies in striatum with up-regulated dopaminergic receptors: a pilot study to investigate the potential of SC antibodies to increase dopaminergic activity. Neurosci Lett. 2012;523(2):186–189. PubMed CrossRef
44. Cox CJ, Sharma M, Leckman JF, et al. Brain human monoclonal autoantibody from sydenham chorea targets dopaminergic neurons in transgenic mice and signals dopamine D2 receptor: implications in human disease. J Immunol. 2013;191(11):5524–5541. PubMed CrossRef
45. Dileepan T, Smith ED, Knowland D, et al. Streptococcus intranasal infection promotes CNS infiltration by streptococcal-specific Th17 cells. J Clin Invest. 2016;126(1):303–317. PubMed CrossRef
46. Ochoa-Aguilar A, Ventura-Martinez R, Sotomayor-Sobrino MA, et al. Review of antibiotic and non-antibiotic properties of beta-lactam molecules. Antiinflamm Antiallergy Agents Med Chem. 2016;15(1):3–14. PubMed CrossRef
47. Altenburg J, de Graaf CS, van der Werf TS, et al. Immunomodulatory effects of macrolide antibiotics, part 1: biological mechanisms. Respiration. 2011;81(1):67–74. PubMed CrossRef
48. Yaddanapudi K, Hornig M, Serge R, et al. Passive transfer of streptococcus-induced antibodies reproduces behavioral disturbances in a mouse model of pediatric autoimmune neuropsychiatric disorders associated with streptococcal infection. Mol Psychiatry. 2010;15(7):712–726. PubMed CrossRef
49. Lotan D, Cunningham M, Joel D. Antibiotic treatment attenuates behavioral and neurochemical changes induced by exposure of rats to group a streptococcal antigen. PLoS One. 2014;9(6):e101257. PubMed CrossRef
50. Maroof A, Yorgensen YM, Li Y, et al. Intranasal vaccination promotes detrimental Th17-mediated immunity against influenza infection. PLoS Pathog. 2014;10(1):e1003875. PubMed CrossRef
51. Frank KM, Zhou T, Moreno-Vinasco L, et al. Host response signature to Staphylococcus aureus alpha-hemolysin implicates pulmonary Th17 response. Infect Immun. 2012;80(9):3161–3169. PubMed CrossRef
52. Stock ML, Fiedler KJ, Acharya S, et al. Antibiotics acting as neuroprotectants via mechanisms independent of their anti-infective activities. Neuropharmacology. 2013;73:174–182. PubMed CrossRef
53. Rothstein JD, Patel S, Regan MR, et al. Beta-lactam antibiotics offer neuroprotection by increasing glutamate transporter expression. Nature. 2005;433(7021):73–77. PubMed CrossRef
54. Pittenger C, Bloch MH, Williams K. Glutamate abnormalities in obsessive compulsive disorder: neurobiology, pathophysiology, and treatment. Pharmacol Ther. 2011;132(3):314–332. PubMed CrossRef
55. Gualdoni GA, Lingscheid T, Schmetterer KG, et al. Azithromycin inhibits IL-1 secretion and non-canonical inflammasome activation. Sci Rep. 2015;5(1):12016. PubMed CrossRef
56. Liadaki K, Petinaki E, Skoulakis C, et al. Toll-like receptor 4 gene (TLR4), but not TLR2, polymorphisms modify the risk of tonsillar disease due to Streptococcus pyogenes and Haemophilus influenzae. Clin Vaccine Immunol. 2011;18(2):217–222. PubMed CrossRef
57. Turna J, Grosman Kaplan K, Anglin R, et al. What’s bugging the gut in OCD? a review of the gut microbiome in obsessive-compulsive disorder. Depress Anxiety. 2016;33(3):171–178. PubMed CrossRef
58. Ivanov II, Frutos Rde L, Manel N, et al. Specific microbiota direct the differentiation of IL-17-producing T-helper cells in the mucosa of the small intestine. Cell Host Microbe. 2008;4(4):337–349. PubMed CrossRef
59. Zareifopoulos N, Panayiotakopoulos G. Neuropsychiatric effects of antimicrobial agents. Clin Drug Investig. 2017;37(5):423–437. PubMed CrossRef
60. Albert RK, Schuller JL; COPD Clinical Research Network. Macrolide antibiotics and the risk of cardiac arrhythmias. Am J Respir Crit Care Med. 2014;189(10):1173–1180. PubMed CrossRef
61. Logan LK, McAuley JB, Shulman ST. Macrolide treatment failure in streptococcal pharyngitis resulting in acute rheumatic fever. Pediatrics. 2012;129(3):e798–e802. PubMed CrossRef
62. Valery PC, Morris PS, Byrnes CA, et al. Long-term azithromycin for Indigenous children with non-cystic-fibrosis bronchiectasis or chronic supportive lung disease (Bronchiectasis Intervention Study): a multicentre, double-blind, randomised controlled trial. Lancet Respir Med. 2013;1(8):610–620. PubMed CrossRef
63. Ilakkiya A, Parveen S, Kumar CN, et al. Macrolide resistance in streptococcus species. J Pharm Bioallied Sci. 2015;7(suppl 1):S26–S31. PubMed CrossRef
64. Leslie DL, Kozma L, Martin A, et al. Neuropsychiatric disorders associated with streptococcal infection: a case-control study among privately insured children. J Am Acad Child Adolesc Psychiatry. 2008;47(10):1166–1172.
bMed65. Carapetis JR, Steer AC, Mulholland EK, et al. The global burden of group A streptococcal diseases. Lancet Infect Dis. 2005;5(11):685–694. PubMed CrossRef
66. Schödel F, Moreland NJ, Wittes JT, et al. Clinical development strategy for a candidate group A streptococcal vaccine. Vaccine. 2017;35(16):2007–2014. Published online Mar 16, 2017. PubMed CrossRef
67. Kuo CF, Tsao N, Hsieh IC, et al. Immunization with a streptococcal multiple-epitope recombinant protein protects mice against invasive group A streptococcal infection. PLoS One. 2017;12(3):e0174464. PubMed CrossRef
68. Murphy TK, Storch EA, Strawser MS. Selective serotonin reuptake inhibitor induced behavioral activation in the PANDAS subtype. Prim Psychiatry. 2006;13(8):87–89.
69. Williams KA, Swedo SE, Farmer CA, et al. Randomized, controlled trial of intravenous immunoglobulin for pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections. J Am Acad Child Adolesc Psychiatry. 2016;55(10):860–867.e2. PubMed CrossRef
70. Perlmutter SJ, Leitman SF, Garvey MA, et al. Therapeutic plasma exchange and intravenous immunoglobulin for obsessive-compulsive disorder and tic disorders in childhood. Lancet. 1999;354(9185):
1153–1158. PubMed CrossRef
71. Pallanti S. Antibiotic Treatment and Intravenous Immunoglobulin Trial for PANDAS (ATIVPANDAS). ClinicalTrials.gov website. https://clinicaltrials.gov/ct2/show/NCT01769027.
72. Storch EA, Murphy TK, Geffken GR, et al. Cognitive-behavioral therapy for PANDAS-related obsessive-compulsive disorder: findings from a preliminary waitlist controlled open trial. J Am Acad Child Adolesc Psychiatry. 2006;45(10):1171–1178. PubMed CrossRef
73. Nadeau JM, Jordan C, Selles RR, et al. A pilot trial of cognitive-behavioral therapy augmentation of antibiotic treatment in youth with pediatric acute-onset neuropsychiatric syndrome-related obsessive-compulsive disorder. J Child Adolesc Psychopharmacol. 2015;25(4):337–343. PubMed CrossRef
74. Pediatric OCD Treatment Study Team. Cognitive-behavior therapy, sertraline, and their combination for children and adolescents with obsessive-compulsive disorder. JAMA. 2004;292(16):1969–1976. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!


