Editor’s Note
The Psychiatric Consultation Service at Massachusetts General Hospital (MGH) sees medical and surgical inpatients with comorbid psychiatric symptoms and conditions. Such consultations require the integration of medical and psychiatric knowledge. During their twice-weekly rounds, Dr Stern and other members of the Consultation Service discuss the diagnosis and management of conditions confronted. These discussions have given rise to rounds reports that will prove useful for clinicians practicing at the interface of medicine and psychiatry.
Dr Stern is chief of the Psychiatric Consultation Service at MGH and is a professor of psychiatry at Harvard Medical School (HMS); Drs Celano and Gross are clinical fellows in psychiatry at HMS and fellows in adult psychiatry at MGH and McLean Hospital; Dr Huffman is director of Inpatient Psychiatry Services at MGH; Drs Freudenreich, Kontos, and Nejad are attending physicians on the Psychiatric Consultation Service at MGH; Ms Repper-DeLisi is a psychiatric nurse clinician on the Nursing Consultation Service at MGH; Dr Thompson is director of the Medical Intensive Care Unit at MGH and an associate professor of medicine at HMS.
Dr Stern is an employee of the Academy of Psychosomatic Medicine; has served on the speaker’s board of Reed Elsevier; is a stock shareholder in WiFiMed (Tablet MD); and has received royalties from Mosby/Elsevier and McGraw Hill. Dr Freudenreich has served as a consultant to Beacon Health Strategies and has received grant/research support from Pfizer and honoraria from Reed Medical Education.
Drs Celano, Gross, Huffman, Kontos, Nejad, and Thompson and Ms Repper-DeLisi report no financial or other affiliations relevant to the subject of this article.
Corresponding author: Anne F. Gross, MD, Department of Psychiatry, Massachusetts General Hospital, Warren 6 275 Charles St, Boston, MA 02114 ([email protected]).
Prim Care Companion J Clin Psychiatry 2010;12(1):e1-e11
Published online: February 25, 2010 (doi:10.4088/PCC.09r00938yel)
© Copyright 2010
Physicians Postgraduate Press, Inc.
The Assessment and Management of Agitation and Delirium in the General Hospital
Have you ever wondered what is responsible for a patient’s agitation and confusion? Have you debated about what the workup of agitation should involve? Have you been uncertain about how best to manage agitation? If so, then the following case vignette and discussion should provide the forum for answers to these and other questions related to the differential diagnosis, the assessment, and the treatment of agitation in the medically ill.
CASE VIGNETTE
Mr A, a 60-year-old man, had an unwitnessed fall from approximately 15 ft while painting his house. He was found some time later by passers-by, who called emergency medical services. At the scene, he was combative and moving all 4 extremities. He was electively intubated and brought to the hospital for further care.
Upon arrival at the emergency department, his Glasgow Coma Scale was 10. His vital signs were a heart rate of 88 beats/min, a blood pressure of 117/80 mm Hg, a respiratory rate of 14 breaths/min, an oxygen saturation of 98% on Fio2 100%, and a temperature of 98°F. He was 6 ft tall, weighed 258 lb, and had a body mass index of 35. A neurologic examination was nonfocal; Mr A’s eyes were midline and his extraocular muscles showed full range.
His laboratory studies were significant for a white blood cell count of 20,000 with 90% neutrophils. A comprehensive metabolic panel, liver function tests, urinalysis, urine toxicology screen, and serum toxicology screens were all unremarkable. A noncontrast computerized tomography scan of the head revealed multiple, small hemorrhagic contusions within the right parietal and bilateral inferior frontal lobes with minimal surrounding edema, scattered subarachnoid hemorrhages, and a nondepressed right parietal lobe fracture. C-spine imaging revealed a ventral epidural hematoma with no significant cord compression and a left alar ligamentous avulsive injury but no fracture. His chest x-ray showed a right-sided sixth rib fracture and bilateral upper and lower lobe airspace opacities, consistent with aspiration pneumonia.
Mr A’s medical history included hypertension, obstructive sleep apnea (for which he received continuous positive airway pressure), and thyroid nodules (for which he had undergone a thyroidectomy). His psychiatric history was unremarkable.
Treatment of his head injuries involved use of nimodipine for bleeding prophylaxis and levetiracetam for seizure prophylaxis; for his neck injury, he was maintained in an Aspen collar; for his pneumonia, he was started on antibiotics (vancomycin and cefepime). Mr A was transferred to the surgical intensive care unit (ICU) for further treatment.
In the surgical ICU, he remained intubated and was sedated with a propofol drip (initially at 300 mg/h, then titrated to a Richmond Agitation Sedation Scale score of −1 to +1; he received intravenous [IV] hydromorphone 0.25-0.5 mg q 3 hours prn for pain; he received 1.25 mg on hospital day 2 and 1.25 mg on hospital day 3). His respiratory status slowly improved; on hospital day 5, his sedation was weaned, and he was extubated. When sputum cultures revealed abundant methicillin-sensitive staphylococcus aureus, his antibiotic regimen was changed to ampicillin. Although his respiratory status was improving, his cognition remained impaired. He had periods of relative clarity and incoherent speech, as well as periods of oversedation and agitation, especially at night. He frequently pulled at his IV lines, and he removed his Foley catheter 3 times before he was adequately restrained with 2-point soft restraints. On hospital day 6, he received 1 dose of haloperidol (10 mg IV) for agitation; this was modestly effective, but his QTc lengthened (from 459 ms to 584 ms). Haloperidol was discontinued. When he became agitated later that day, he received olanzapine (5 mg); its use was not associated with QTc prolongation (his QTc ranged between 420 ms and 450 ms); olanzapine decreased his agitation. The psychiatry department was then consulted for further recommendations regarding management of his agitation.
The psychiatric consultant found Mr A sleeping in 2-point restraints. He was easily arousable and intermittently confused. He was unable to state his location or to recount the events that led to his hospitalization; he identified his location as a hospital when provided with choices. His speech was soft and slurred, and it was fluent with normal prosody (emotional melody and tone of language); however, he made frequent paraphasic (substitution of an inappropriate word) errors. He had no abnormal movements, but he frequently pulled at his nasogastric tube. He said his mood was “happy,” and he appeared inappropriately bright. He responded appropriately to some direct questions but at times would respond with answers unrelated to the questions asked. He did not appear to be preoccupied. He was oriented to person but not to the month or year; he registered 3 words but recalled none at 5 minutes; with prompts, he could provide the days of the week forward but not backward. He perseverated at times (persistently giving the days of the week even after he had completed the task).
On physical examination, Mr A’s temperature maximum was 101.5°F. There was wide variability in his heart rate and blood pressure; his heart rate ranged from 36 to 96 beats/min and his systolic blood pressure ranged from 100 to 190 mm Hg. His oxygen saturation was 99% while receiving 4 L via nasal cannulae. Mr A’s neck was supple, and he was not diaphoretic.
On neurologic examination, cranial nerves II-X were intact. There was no evidence of hemineglect. No snout, palmomental, or glabellar reflexes were elicited. Grip strength was 5/5 bilaterally, and Mr A was able to move his toes on command. His upper extremities revealed no rigidity, cogwheeling, or tremor.
Laboratory studies revealed a calcium level of 9.1 mEq/L, a magnesium level of 1.9 mEq/L, and a potassium level of 3.6 mEq/L. Serum glucose levels were within normal limits. A complete blood count revealed a mild normocytic anemia and a borderline elevated white blood cell count. His thyroid-stimulating hormone level was 0.43. Rapid plasma reagin and human immunodeficiency virus tests had been negative the year before; they were not repeated.
A diagnosis of delirium was made. Contributing factors were thought to include hypoxia, pneumonia, and traumatic brain injury. Recommendations included the following: to continue to optimize his respiratory status, to treat his infection, and to perform frequent neurologic checks observing for signs of increased intracranial pressure. Pharmacologic interventions for agitation included use of olanzapine (2.5 mg/2.5 mg/5 mg) and valproic acid (125 mg/125 mg/500 mg); olanzapine and haloperidol were also available on a prn basis. Recommendations for an alternative antibiotic were made, as ampicillin can prolong the QTc. Repletion of electrolytes and a check of a daily electrocardiogram (EKG) were recommended to reduce the risk of torsades de pointes.
Over the next several days, Mr A’s olanzapine was increased (to 10 mg po tid), valproic acid was increased (to 1,000 mg po bid), and haloperidol was tapered. On this regimen, his cognition improved slowly. Although he was suffering from a Wernicke’s aphasia, his attention and orientation improved (to where he could state the year, month, and name of the hospital). Olanzapine was tapered slowly, and valproic acid was continued (due to its antiepileptic properties and its ability to decrease impulsivity).
On hospital day 14, he was transferred to a rehabilitation center that specializes in the care of patients with an acute brain injury.
WHAT IS DELIRIUM?
Broadly defined, delirium designates a disturbance of brain or mental function characterized by a clouded consciousness and confusion, a disturbance that is usually acute and transient (ie, potentially reversible) and with a restitutio ad integrum if the underlying etiology can be treated. The etymology of delirium is also informative. Delirium is derived from the Latin terms de, meaning down or away from, and lira, meaning a furrow or track in the fields; that is, to be off the track. In the case of a delirious person, his or her brain is off track. Other equally descriptive terms include acute brain failure, acute confusional state, or encephalopathy.
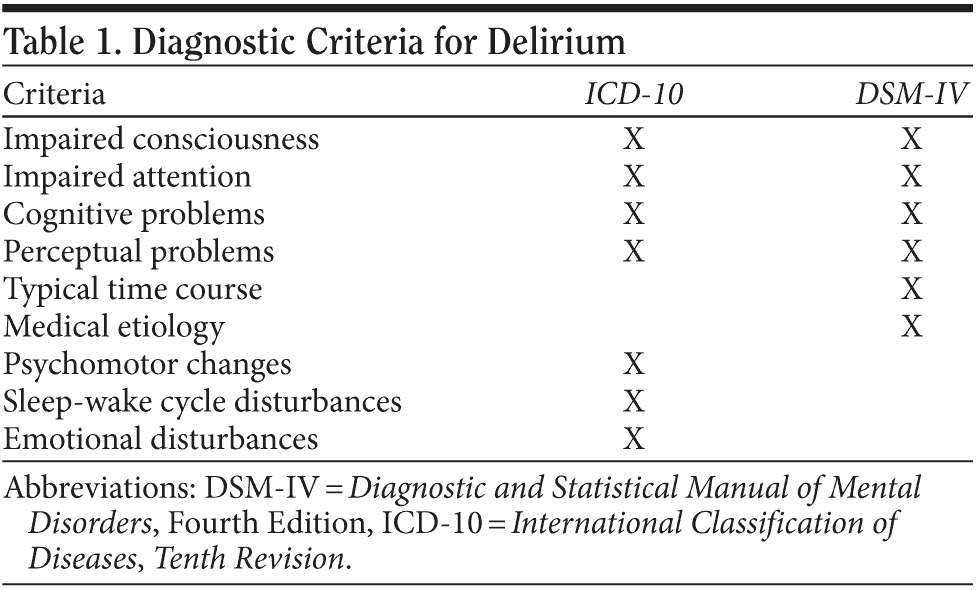
According to Chédru and Geschwind1 and Mesulam and associates,2 inattention is the sine qua non of delirium, with difficulties sustaining, focusing, or shifting attention. Table 1 contrasts the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision3 and the International Classification of Diseases, Tenth Revision4 operationalized diagnostic criteria. Both classification systems identify inattention and a disturbance in consciousness as the hallmarks of delirium. Ancillary cognitive features that can overshadow these core features include perceptual disturbances and psychosis (eg, illusions, visual hallucinations, or persecutory ideation that tends to be fleeting and poorly formed), language problems (eg, rambling speech or perseveration), or problems with memory and orientation. Fluctuation in symptomatology over the course of the day is characteristic, as is a disturbance in the sleep-wake cycle.5 A patient can be agitated or apathetic.6 In an extreme case, a patient is agitated and awake at night, while barely arousable during the day. Affective changes ranging from euphoria to abulia can also be observed. Not all features of the syndrome must be present; this accounts for why delirious patients look different from one another.
In a widely used algorithm that aids in the recognition of delirium, the Confusion Assessment Method inquires about the presence of a common alteration in mental status (one involving both an acute nature and a fluctuating course and characterized by inattentiveness) in addition to either disorganized thinking or an altered level of consciousness (defined as anything but alert).7
WHAT CONDITIONS LOOK LIKE DELIRIUM?
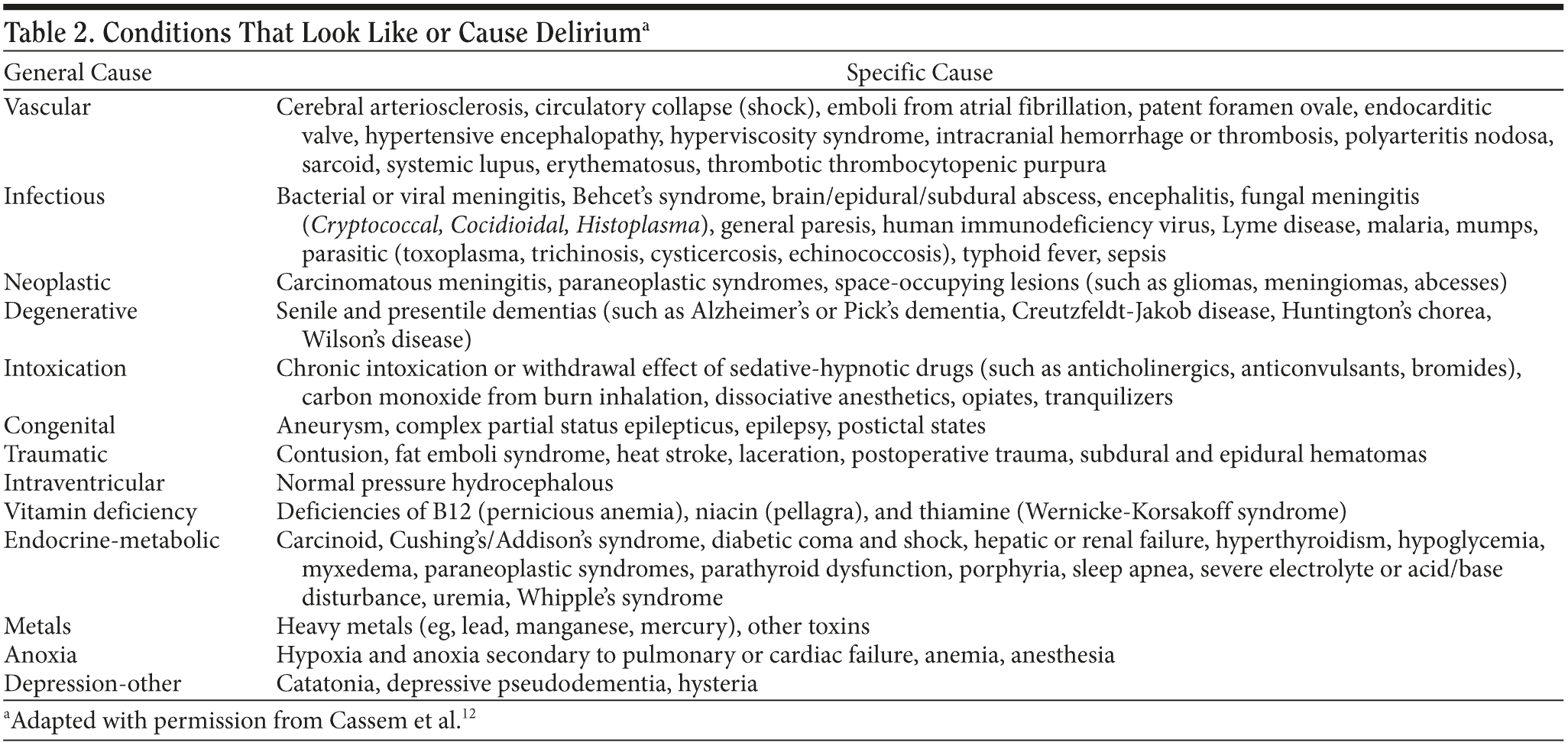
One of the conditions most commonly confused with delirium is dementia, a condition considered to have an irreversible and progressive alteration in brain function, typically with an insidious onset and without fluctuations. In general, patients with dementia are alert and fail to exhibit clouded mentation. However, dementia is a risk factor for delirium; the 2 diagnoses are not mutually exclusive, and they may be very difficult to separate from one another.8 Several other psychiatric syndromes can be confused with delirium, eg, Bell’s mania (also called delirious mania),9 a category of acute and transient psychotic disorder,10 chronic psychosis, depression, and catatonia.11 A more complete differential diagnosis is provided in Table 2.
WHAT IS RESPONSIBLE FOR DELIRIUM?
The medical causes of delirium are legion. An organized approach may be facilitated by use of the mnemonic “WHHHIMP,” indicating 7 potentially fatal etiologies: Wernicke’s encephalopathy, hypoxemia, hypoglycemia, hypertensive crisis, intracranial bleeding, meningitis/encephalitis, and poisoning.12 Table 3 provides a more extensive list of etiologies and expands on the original WHHHIMP mnemonic.13
HOW SHOULD DELIRIUM BE ASSESSED?
Awareness of several principles may reduce the likelihood of diagnostic errors while dealing with delirium.
Delirium “always” has a medical cause. Since delirium, by definition, always has a medical cause, the identification of such a cause (including a rare cause of delirium, eg, paraneoplastic syndrome) must be aggressively pursued. In a minority of cases of delirium, no etiology can be identified. If delirium is overlooked, a patient’s symptoms are often falsely attributed to a psychiatric syndrome and medical treatments are foregone.
Delirium is often multifactorial. In many cases, more than 1 factor is responsible for the development of delirium. Failing to understand this may lead to premature closure and to the overlooking of potentially reversible causes of delirium.
Delirium has no etiologic specificity. Unfortunately, delirium is a rather nonspecific response pattern of the brain. While many authors, most prominent among them the German psychiatrist Karl Bonhoeffer (the father of the theologian Dietrich Bonhoeffer),14 have attempted to delineate etiology-specific deliria, the clinical phenomenology does not provide enough guidance as to help with the etiology.
Delirium is potentially life threatening. Delirium should be suspected in anyone with an alteration in mental status; when detected, a workup should be initiated urgently.
Delirium can be subtle. In milder, subsyndromal cases or in cases with lucid periods, delirium can be missed and misdiagnosed.15 The delirium-specific aspects of the mental state (ie, inattention and impaired consciousness) can be subtle or overshadowed by more striking psychopathology (such as psychosis or oppositional behavior), leading to the inappropriate application of psychiatric diagnoses. In 1 case series of consecutively referred inpatients with delirium, 43% of patients were psychotic.16
Our patient, Mr A, fell off a ladder and developed delirium (characterized by an acute onset and by prominent problems with attention and consciousness). At least 1 of the etiologies was obvious: his head trauma. However, since delirium is often multifactorial, other causes (eg, hypoxemia due to aspiration pneumonia) were appropriately considered.
HOW SHOULD AGITATION AND DELIRIUM BE ASSESSED?
Since the core principles in the treatment of delirium are to identify and to correct the etiologic factor(s) that contribute to the patient’s symptoms, a careful review of the patient’s medical history and information from collateral sources may provide the necessary clues and information, especially when the patient is an unreliable historian or when his or her underlying medical condition (eg, intubation) prevents a clinician from obtaining an extensive or accurate history. One should also be cynical and consider why Mr A fell off the ladder. Was he intoxicated? Might he have been experiencing delirium as a consequence of substance withdrawal? In addition, laboratory and radiologic data may also shed light on contributing factors to the patient’s problems.
As use of, and withdrawal from, pharmacologic agents are among the most common iatrogenic causes of delirium, a review of current and past medications should always be conducted. Delirium may occur when certain drugs and substances associated with physiologic dependence are discontinued abruptly or when central nervous system-active drugs reach supratherapeutic levels (with resultant neurotoxicity). Pharmacokinetic and pharmacodynamic drug interactions may further exacerbate delirium; however, management generally entails discontinuing or reducing the dose of the offending drug(s) whenever possible. If necessary, administration of the necessary antidote(s), eg, naloxone, flumazenil, or physostigmine, may be required.
WHICH DRUGS CAN PRECIPITATE DELIRIUM WHEN ABRUPTLY WITHDRAWN?
Drug withdrawal syndromes are common in general hospital settings.17 Moreover, emergent medical and surgical admissions may result in the sudden discontinuation of abused drugs, and a history of chronic drug use may be difficult to establish in an intubated patient. In addition, the physical signs of withdrawal are nonspecific (eg, increased temperature, tachycardia, or diaphoresis) and may mimic the signs of medical illness. Furthermore, while certain laboratory results (such as elevated liver enzymes and mean corpuscular volume) may indicate heavy alcohol use, no laboratory test can confirm the diagnosis.
Alcohol. Alcohol withdrawal may also be accompanied by agitation and psychosis. Its treatment often consists of the administration of increasing doses of benzodiazepines to achieve autonomic system control and to prevent alcohol withdrawal seizures. In certain cases, adjunctive treatment with neuroleptics or α2 agonists is required, especially for cases of delirium tremens manifest by an altered mental status and by agitation.18,19
Sedative-hypnotic withdrawal. Sedative hypnotics, like alcohol, also result in γ-aminobutyric acid (GABA) receptor withdrawal, and, thus, their withdrawal presents in a fashion similar to that of alcohol withdrawal. Symptom onset is a function of the half-life of the agent in question; the shorter the half-life, the quicker the onset of withdrawal. Withdrawal symptoms are generally more intense with agents (such as lorazepam, oxazepam, and alprazolam) associated with a half-life of 10-20 hours. Treatment of withdrawal from these agents consists of using a drug from the same class, but with a longer half-life, such as diazepam or chlordiazepoxide.17
Narcotic withdrawal. Delirium tends to be a rare complication of opioid withdrawal; such patients are usually able to provide an accurate history of their drug use, which prompts replacement therapy. The major clinical dilemma arises when differentiating symptoms associated with narcotic withdrawal from those due to an underlying medical illness. The syndrome is generally mild (with abdominal cramping, nausea, emesis, diarrhea, rhinorrhea, and general discomfort). Treatment involves use of a longer half-life agent, such as methadone, which can be tapered during the hospital course. Use of clonidine may also help to reduce noradrenergic signs of withdrawal; antiemetics may mitigate nausea and emesis.
HOW CAN AGITATION AND DELIRIUM BE MANAGED?
The management of patients with delirium must take into account several overarching goals: to ensure the safety of the patient and staff, to identify and treat the underlying causes, and to improve the patient’s level of function.
Cognitive dysfunction, behavioral dyscontrol, and other behavioral changes associated with delirium often endanger the patient or medical staff.20 The psychiatric consultant should always assess for the risk of suicide, for self-harm, and for violence; then, a plan to minimize these risks should be implemented. Self-destructive or suicidal acts may result from underlying cognitive dysfunction (along with sensory and perceptual impairment, hallucinations, or delusions). In addition, a patient may be impulsive and attempt to get out of bed, to wander, and to fall (which may lead to further injury or death) and attempt to remove IV lines, tubes, or catheters.21
Removal of potentially dangerous items from the room and the surrounding area, institution of sitters to provide greater supervision, utilization of physical restraints, and initiation of pharmacologic treatment are often necessary.
WHICH PHARMACOLOGIC AGENTS EFFECTIVELY TREAT AGITATION AND DELIRIUM?
Although the best treatment of delirium is one that treats the problem specifically, the symptoms of delirium often decrease with use of pharmacologic agents. Antipsychotics are most frequently used for control of agitation, paranoia, and psychosis associated with delirium. In addition, depending on the etiology of the agitation, use of benzodiazepines, narcotics, and other sedatives may also be employed.
Haloperidol. Haloperidol, administered intravenously, is the preferred treatment for agitated delirious patients (as described by the guidelines of the American Psychiatric Association22) despite the fact that this route of administration has not been approved by the US Food and Drug Administration and that it now carries a “black box warning” associated with the risk of ventricular arrhythmia, including torsades de pointes.
Haloperidol has strong dopamine D2 receptor-binding affinity; this may explain its efficacy in the treatment of delirious patients. It has minimal anticholinergic activity and has few active metabolites. The effects of haloperidol on blood pressure, pulmonary artery pressure, heart rate, and respiration are mild when compared to those of IV benzodiazepines and other neuroleptics. In addition, it has little effect on respiratory drive. Intravenous administration of haloperidol is preferable to intramuscular administration since drug absorption after intramuscular administration may be inconsistent, especially in critically ill patients with hemodynamic instability. In addition, painful intramuscular injections can exacerbate paranoia, agitation, and treatment noncompliance. Moreover, use of IV haloperidol is less likely to produce extrapyramidal symptoms (EPS) than is intramuscular or even oral haloperidol.12
IV haloperidol has a mean distribution time of 11 minutes, with tranquilizing effects generally seen in 10-20 minutes. The mean half-life of haloperidol when given intravenously is approximately 24 hours. Generally, the dose of haloperidol is titrated to the severity of the underlying agitation. If agitation is mild, it is reasonable to start with 0.5-2.5 mg of IV haloperidol, if it is moderate, 5-10 mg IV is given, and if severe, 10 mg IV is given at the outset. The clinician should adjust the dose to the clinical course, and IV doses can be repeated every 15-30 minutes until calm is achieved. If marked agitation persists, the previous dose can be doubled after 30 minutes. Successive doublings may follow, as the goal of ameliorating the agitation is sought. Although rarely needed, for persistent agitation, continuous infusions of 5-10 mg/h can be initiated.12
Other typical antipsychotics. IV droperidol may also reduce agitation in delirious patients. Droperidol is more sedating than haloperidol, and, thus, care should be taken when used in patients with underlying respiratory compromise. In addition, it is a more potent α1-adrenergic receptor antagonist and thus is more likely to cause hypotension.12
Other neuroleptics that are occasionally administered parenterally for agitation include perphenazine and chlorpromazine; however, they are more often associated with hypotension, arrhythmias, and anticholinergic effects than is haloperidol.12
Atypical antipsychotics. Atypical antipsychotics (including risperidone, olanzapine, quetiapine, aripiprizole, ziprasidone, and clozapine) have also been used for the treatment of delirium. While the number of studies is limited, there does not seem to be any significant difference with regard to the efficacy of atypical antipsychotics, and none has shown a greater efficacy or an improved safety profile when compared to the use of IV haloperidol among delirious patients.23-25
Benzodiazepines. There have been few controlled studies of the efficacy of benzodiazepines as monotherapy for the treatment of delirium. The limited data available suggest that benzodiazepine monotherapy is generally ineffective as a treatment for most cases of delirium, ie, delirium that is not associated with alcohol or GABA receptor withdrawal syndromes.26,27 However, adding a benzodiazepine as an adjunctive agent to haloperidol in the management of a severely agitated patient can be helpful, as benzodiazepines may reduce the EPS (especially akathisia) of haloperidol.28 In addition, in several open studies, the combination of IV lorazepam with IV haloperidol has resulted in greater efficacy and a shorter duration of the delirium when compared with use of haloperidol alone.29,30 Of the benzodiazepines, lorazepam is most commonly used, as it has no active metabolites and is least likely to cause respiratory depression.
Narcotics. When pain may be an aggravating factor or a cause of delirium, narcotics may be useful. Morphine is the most commonly used agent in the medical setting. Morphine has very weak anticholinergic effects; however, its antihistaminergic activity may cause hypotension. Hydromorphone, a more potent narcotic, is also well tolerated in appropriate doses. Fentanyl and meperidine should be avoided in patients with an altered mental status; each has anticholinergic effects, and the accumulation of normeperidine, an active metabolite of meperidine, may cause neurotoxicity and worsening delirium.31 Methadone, with a long half-life, is also often used for pain control, and it helps to provide stable therapeutic levels of pain relief. In addition, it has been associated with N-methyl-d-aspartate receptor antagonism and decreased mesolimbic dopamine release that may help explain the lower rates of altered mental status changes seen.32,33 Respiratory depression and hypotension are side effects of all narcotics, and these should be monitored closely.
Valproate. The addition of IV valproate may be considered in the management of severe agitation when conventional therapy is inadequate or when problematic side effects emerge from the use of antipsychotics.34 Its use may be particularly helpful in patients who exhibit symptoms of disinhibition or impulsivity during episodes of agitation.35
Propofol. When agitation fails to respond to more traditional medications, IV sedation may be required. Propofol is a short-acting, IV-administered hypnotic agent. It is used for the induction and maintenance of general anesthesia, for sedation of mechanically ventilated adults, and for procedural sedation.36 Clinically, propofol is not considered an analgesic, so opioids (such as fentanyl) are often combined with propofol to help alleviate pain in critically injured patients. It has several mechanisms of action, both through potentiation of GABA-A receptor activity and also acting as a sodium channel blocker.37 Recent research has also suggested the endocannabinoid system may contribute significantly to propofol’s anesthetic action and to its unique properties.38
Dexmedetomidine. Dexmedetomidine is a highly selective α2-adrenoreceptor agonist that has been shown to have both sedative and analgesic effects. Compared with clonidine, an α2-agonist used for the treatment of hypertension, dexmedetomidine has an α2 : α1-adrenoreceptor ratio of approximately 1,600 : 1 (7 to 8 times higher than clonidine), making it primarily a sedative-anxiolytic like propofol.39 It has no GABAergic activity or any anticholinergic activity, and a recent study in cardiac surgery patients has shown low rates of postoperative delirium following its administration.40 Case reports have also highlighted its usefulness in the treatment of alcohol withdrawal delirium.41,42
WHAT ARE THE COMMON SIDE EFFECTS OF
THE ANTIPSYCHOTICS USED TO TREAT DELIRIUM?
Antipsychotics used for the treatment of delirium are associated with a variety of adverse effects. These include anticholinergic effects, EPS, neuroleptic malignant syndrome (NMS), orthostatic hypotension, and, perhaps most importantly, the risk of QT interval prolongation and torsades de pointes ventricular arrhythmia.
Anticholinergic side effects (including dry mouth, urinary retention, constipation, tachycardia, and confusion) result from blockade of muscarinic cholinergic receptors. They are most often found with use of low-potency typical antipsychotics and clozapine; these agents are infrequently used to treat delirium. Among the antipsychotics typically used to treat delirium (eg, haloperidol and most atypical antipsychotics), anticholinergic side effects are rare. However, higher doses of the atypical antipsychotics olanzapine and quetiapine can result in anticholinergic effects, and one must monitor patients for these side effects when using these medications at high doses.
EPS—dystonia, akathisia, and parkinsonism—occur most often with antipsychotics that are potent D2 receptor blockers. Among agents most frequently used to treat delirium, these include haloperidol, risperidone, and, at higher doses, olanzapine. Interestingly, however, EPS are much less common with use of IV administration of haloperidol43,44; such rates are seen much less often when haloperidol is given via the IV route than orally or intramuscularly. The reason that EPS occur at markedly lower rates via the IV route is not known, though presumably this phenomenon is related to the avoidance of first-pass metabolism through the gut and the liver with IV administration.
NMS is a syndrome associated with the administration of antipsychotics. Symptoms of NMS include confusion, fever, autonomic instability, and a lead-pipe rigidity; substantially elevated levels of creatine phosphokinase (CPK) are very common. Though it is more commonly considered in patients who have primary psychotic illnesses, NMS has been observed in patients who receive antipsychotics for the treatment of delirium.45 Risk factors for NMS appear to include the type of antipsychotic used (NMS appears to occur more commonly with agents associated with EPS such as oral haloperidol) and appears to occur more frequently when these agents are given at higher doses.45,46 There is some question about whether treatment with atypical antipsychotics causes an “incomplete” form of NMS, with fewer motor symptoms and less of an elevation of CPK, but the existing literature suggests that such a syndrome appears to be associated with clozapine but not the atypicals (such as risperidone) that do have significant D2 receptor activity.47,48
Orthostatic hypotension, a significant drop in blood pressure upon sitting or standing, is related to blockade of α1 receptors. Among antipsychotics commonly used to treat delirium, orthostatic hypotension is most common among patients receiving quetiapine or risperidone. This is an important effect since patients with delirium—who have ongoing medical issues and are often deconditioned—are already at substantial risk of falls.12
One final and important adverse effect associated with antipsychotics is prolongation of the corrected QT interval (QTc). The QT interval is measured on the EKG from the beginning of the QRS complex to the end of the T wave, and it represents ventricular depolarization and repolarization within the heart. The QT interval has a characteristic length of 400-450 ms when corrected for heart rate; when the QTc is over 500 ms, it is considered as clearly prolonged. When the QTc prolongs, it serves as a substantial risk factor for torsades de pointes, a malignant polymorphic ventricular arrhythmia that may result in sudden death.12
It appears that all antipsychotics can cause QTc prolongation. Though there has not been a systematic study of the comparative QTc prolongation caused by antipsychotics in delirious patients, a study of antipsychotic administration in healthy volunteers found differential QTc prolongation depending on the agent.49 In that study, olanzapine and (oral) haloperidol appeared to cause the least prolongation of the QTc. Though haloperidol appears to be associated with limited prolongation of the QTc when given orally, IV haloperidol has been associated with QTc prolongation and with over 2 dozen cases of torsades de pointes; therefore, clinicians should be aware of significant QTc changes when this agent is used to treat patients with delirium.12
HOW DOES ONE MANAGE THE RISK OF Torsades de Pointes IN A PATIENT RECEIVING ANTIPSYCHOTICS FOR DELIRIUM?
First, it is important to monitor the QTc interval before and after administration of antipsychotics. Fortunately, the vast majority of patients admitted to medical or surgical units have had a baseline EKG from admission. If the patient’s baseline QTc is > 500 ms, or if the QTc prolongs to that length or increases by an absolute value of 60 ms with administration of an antipsychotic, alternatives should be considered.
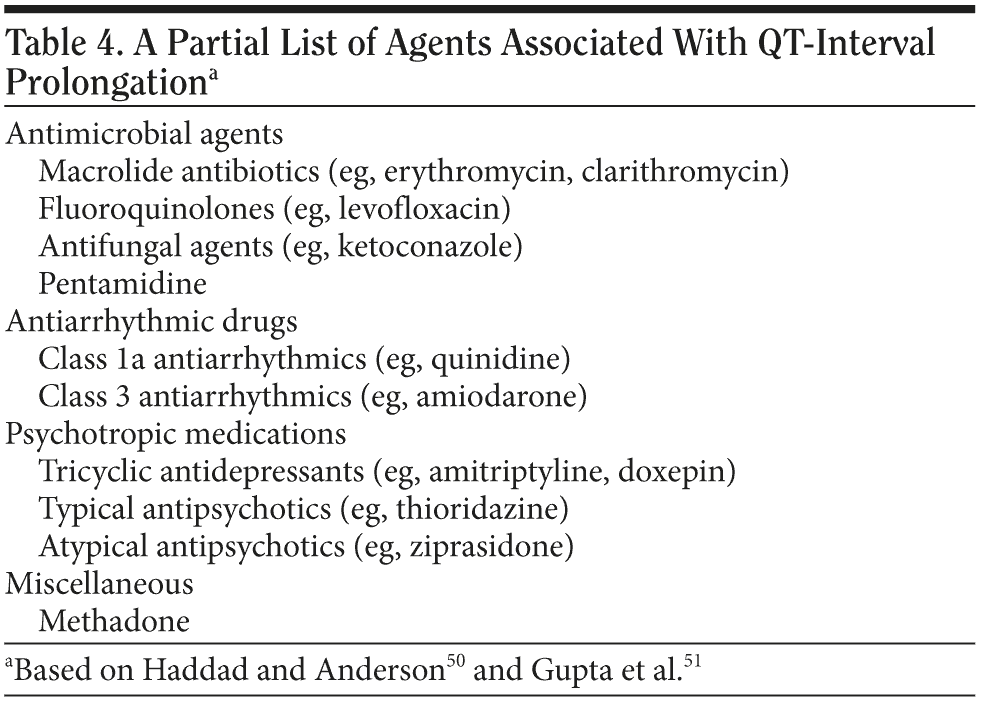
In addition to simply monitoring the QTc, measures should be taken to reduce the risk of QTc prolongation. When antipsychotics (and especially IV haloperidol) are used, other QT-prolonging medications (such as macrolide antibiotics, fluoroquinolones, tricyclic antidepressants, or amiodarone) (Table 4)50,51 should be avoided when other alternatives are available. Low potassium and low magnesium are associated with both QTc prolongation and torsades de pointes, and, where possible, levels of potassium and magnesium should be repleted to high-normal levels.
If the QTc interval becomes prolonged, several alternatives are available, depending on the clinical situation and the extent of the QTc prolongation. If the QTc interval has increased to slightly over 500 ms and the patient clearly would benefit from use of an antipsychotic, using a low dose of an antipsychotic that may have a lesser propensity for QT prolongation (eg, olanzapine) is an option. If the QTc has been substantially prolonged or rather high doses of an agent are needed, benzodiazepines can be used urgently for more immediate sedation and treatment of agitation. Finally, if agitation or confusion are mild and can be managed temporarily with nonpharmacologic methods, it is often the case that with time and repletion of potassium or magnesium, the QTc will normalize and antipsychotics can be restarted.50,51
WHAT SIDE EFFECTS ARE ASSOCIATED WITH OTHER PHARMACOLOGIC AGENTS USED TO TREAT DELIRIUM?
A number of other agents (including benzodiazepines, propofol, and narcotic analgesics) are sometimes given to patients with delirium. These treatments are not typically associated with anticholinergic effects, orthostatic hypotension, EPS, or QTc prolongation. However, these agents can generate side effects (eg, oversedation, respiratory depression, and gait disturbance and with benzodiazepines, risk of paradoxical disinhibition) that limit their use among patients with delirium. Furthermore, these agents frequently worsen confusion (indeed, they are common causes of delirium), and for this reason, they are much less commonly used to treat delirium.
Finally, the selective α2 agonist dexmedetomidine is generally well tolerated; the most common side effect of treatment appears to be bradycardia.52,53 At this stage, its use is more limited by reduced availability and experience (especially outside of ICUs) than by its side effect profile.
HOW COSTLY IS DELIRIUM?
Remarkably, the care of delirious patients accounts for approximately 1% of the annual US gross domestic product54 and is associated with increased length of stay, complications, and poor outcomes (including increased mortality).55-59 In a prospective study of 224 consecutive mechanically ventilated medical ICU patients (without coma), costs were significantly higher (1.6 times higher) for those with at least 1 episode of delirium versus those without delirium, after adjustment for age, comorbidity, severity of illness, degree of organ dysfunction, nosocomial infections, hospital mortality, and other potential confounders. Given that delirium developed in the majority of mechanically ventilated medical ICU patients,60 the cost savings and reduction of suffering would be substantial if the course of delirium could be shortened or, better yet, prevented.
CAN WE PREVENT DELIRIUM?
Given the high prevalence of delirium in the general hospital, its occurrence seems inevitable. Those who are elderly or who get sick enough are prone to mental status changes. While for some, the proverbial “horse has already left the barn” by the time of hospital admission, the majority of cases of delirium develop after admission.61
To prevent delirium, one must start with an analysis of risk factors. Risk factors for delirium can be divided into 3 overlapping categories: modifiable risk factors, nonmodifiable risk factors, and risks that may not be risks at all. Modifiable risk factors include sleep deprivation, impairments of vision and/or hearing, dehydration, iatrogenic and other complications of being an inpatient (most notably, having an infection), and polypharmacy62; depressive illness is also a risk for delirium.63 While it is a correctable condition, it may not be a correctable risk factor; aside from elective surgery, there are few inpatient circumstances wherein it can be addressed before delirium onset. Nonmodifiable risk factors include advanced age, acute and chronic illness, and so-called “organic” brain diseases (most notably the dementias),62 especially when executive dysfunction is present.63 Our patient, Mr A, will forever be considered at high risk for delirium because of his traumatic brain injury. Finally, we come to risks that may not be risks at all (eg, restraints and Foley catheters).64 The implementation of these measures often conveys that a mental status change or a deliriogenic condition is already afoot.
It is also crucial to ask what we seek to prevent. Delirium is not, strictly speaking, a disease; rather, it is a cluster of symptoms that is shared by a vast number of other diseases and conditions. Therefore, when delirium prevention is sought (eg, by paying proactive attention to the risk for alcohol withdrawal), is this delirium prevention or the prevention of illnesses that themselves may or may not subsequently trigger delirium? The distinction may not really matter. Conversely, surrogate terms for delirium such as confusion or agitation only capture a component of a broader syndrome. If these symptoms are prevented, is delirium actually being prevented or just ameliorated?
Keeping the above points in mind, prevention strategies can be divided into positive (“doing something”) and negative (“not doing something”) categories. Among the positive strategies are some old standbys (eg, providing frequent active and passive orientation cues that include displays of calendars, use of note boards, nursing reminders, and lengthy family visitations). In what is perhaps the best-known study of delirium prevention, Inouye and colleagues65 implemented either usual care or an extensive array of orientation, mobilization, sleep correction, sensory optimization, and hydration protocols among 852 geriatric patients admitted to either a regular or a specialized unit. Delirium incidence (as measured by the Confusion Assessment Method) decreased significantly by these means, though severity and recurrence rates were unaffected.65
Other positive interventions include proactive geriatrics consultation that may decrease delirium incidence66 and prophylactic low-dose haloperidol (~1.5 mg/d) that may reduce the severity and duration of delirium; however, the latter was not shown to be strictly preventative.67 Data from a large, randomized, double-blind, placebo-controlled study of olanzapine and placebo in elderly joint replacement patients68 revealed that 10 mg of olanzapine administered perioperatively reduced the incidence of postoperative delirium (from 40% to 14%) and increased the likelihood of discharge to home (versus a rehabilitation facility). A similar strategy using risperidone reduced the incidence of delirium.69 Negative prevention measures include avoidance of medications associated with the onset of delirium (eg, benzodiazepines and those agents with anticholinergic properties). While the delirium prevention literature is sparse, it is not surprising that little attention has been paid to negative strategies; in fact, it has been shown conclusively (albeit inadvertently) that benzodiazepines reliably worsen delirium27; nonetheless, this class of medications is still prescribed frequently for the “sedation” of the agitated and delirious patient.
Obstacles to delirium prevention include a small and shaky evidence base, hindered by suboptimal study designs70 and difficulty implementing nonpharmacologic strategies (as they are highly labor and training intensive). The latter issue was demonstrated by Inouye et al71 in the very hospital wherein they conducted the partially successful prevention trial. This problem may reflect an underemphasis in practice on nonpharmacologic techniques for delirium prevention or mitigation at the same time that there is a philosophical overemphasis on the idea that environment is the dominant factor in deliriogenesis. This can lead to a paradoxical situation in which there is a strong push to pharmacologically “sedate” a patient, while the pathophysiologic workup for mental status change is stalled since the patient is thought to suffer from an entity such as the so-called “ICU psychosis” (in which the ICU itself is mistakenly held to be the etiologic culprit). Whether we mitigate or prevent delirium and whether we prevent its primary causes or its secondary manifestation may not matter from a patient-suffering point of view, but it does matter in terms of how staff and family members are affected by the delirious patient.
CONCLUSION
Delirium is a common and dread complication associated with myriad conditions and medications. Early recognition and timely treatment can reduce morbidity, hospital length of stay, health care costs, and mortality. Educational efforts directed at the prevention, the diagnosis, and the effective resolution of the syndrome are sorely needed.
REFERENCES
1. Chédru F, Geschwind N. Disorders of higher cortical functions in acute confusional states. Cortex. 1972;8(4):395-411. PubMed
2. Mesulam MM, Waxman SG, Geschwind N, et al. Acute confusional states with right middle cerebral artery infarctions. J Neurol Neurosurg Psychiatry. 1976;39(1):84-89. doi:10.1136/jnnp.39.1.84 PubMed
3. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
4. World Health Organization. The ICD-10 Classification of Mental and Behavioural Disorders: Diagnostic Criteria for Research. Geneva, Switzerland: World Health Organization; 1993.
5. Meagher DJ, Moran M, Raju B, et al. Phenomenology of delirium. Assessment of 100 adult cases using standardised measures. Br J Psychiatry. 2007;190(2):135-141. doi:10.1192/bjp.bp.106.023911 PubMed
6. Meagher D. Motor subtypes of delirium: past, present and future. Int Rev Psychiatry. 2009;21(1):59-73. doi:10.1080/09540260802675460 PubMed
7. Inouye SK, van Dyck CH, Alessi CA, et al. Clarifying confusion: the confusion assessment method. A new method for detection of delirium. Ann Intern Med. 1990;113(12):941-948. PubMed
8. Meagher D, Trzepacz PT. Phenomenological distinctions needed in DSM-V: delirium, subsyndromal delirium, and dementias. J Neuropsychiatry Clin Neurosci. 2007;19(4):468-470. PubMed
9. Karmacharya R, England ML, Ong×¼r D. Delirious mania: clinical features and treatment response. J Affect Disord. 2008;109(3):312-316. doi:10.1016/j.jad.2007.12.001 PubMed
10. Marneros A, Pillmann F, Haring A, et al. Features of acute and transient psychotic disorders. Eur Arch Psychiatry Clin Neurosci. 2003;253(4):167-174. doi:10.1007/s00406-003-0420-y PubMed
11. Swartz C, Francis A, Malur C. Delirium or catatonic disorder due to a general medical condition. J ECT. 2002;18(3):167-168, author reply 168. doi:10.1097/00124509-200209000-00012 PubMed
12. Cassem NH, Murray GB, Lafayette JM, et al. Delirious patients. In: Stern TA, Fricchione GL, Cassem NH, et al, eds. Massachusetts General Hospital Handbook of General Hospital Psychiatry. Fifth edition. Philadelphia, PA: Mosby/Elsevier; 2004;119-134. doi:10.1016/B978-0-323-02767-0.50016-4
13. Caplan JP, Cassem NH, Murray GB, et al. Delirium. In: Stern TA, Rosenbaum JF, Fava M, et al, eds. Massachusetts General Hospital Comprehensive Clinical Psychiatry. Philadelphia, PA: Mosby/Elsevier; 2008:217-229.
14. Ströhle A, Wrase J, Malach H, et al. Karl Bonhoeffer (1868-1948). Am J Psychiatry. 2008;165(5):575-576. doi:10.1176/appi.ajp.2008.07061031 PubMed
15. Meagher DJ, Maclullich AM, Laurila JV. Defining delirium for the International Classification of Diseases, 11th Revision. J Psychosom Res. 2008;65(3):207-214. doi:10.1016/j.jpsychores.2008.05.015 PubMed
16. Webster R, Holroyd S. Prevalence of psychotic symptoms in delirium. Psychosomatics. 2000;41(6):519-522. doi:10.1176/appi.psy.41.6.519 PubMed
17. Kosten TR, O’ Connor PG. Management of drug and alcohol withdrawal. N Engl J Med. 2003;348(18):1786-1795. doi:10.1056/NEJMra020617 PubMed
18. Palestine ML, Alatorre E. Control of acute alcoholic withdrawal symptoms: a comparative study of haloperidol and chlordiazepoxide. Curr Ther Res Clin Exp. 1976;20(3):289-299. PubMed
19. Crippen D, Ermakov S. Continuous infusions of haloperidol in critically ill patients. Crit Care Med. 1995;23(1):214-215. doi:10.1097/00003246-199501000-00041 PubMed
20. Breitbart W, Payne DK. Pain. In: Holland J, ed. Psycho-oncology. New York, NY: Oxford University Press; 1998:450-467.
21. Lakatos BE, Capasso V, Mitchell MT, et al. Falls in the general hospital: association with delirium, advanced age, and specific surgical procedures. Psychosomatics. 2009;50(3):218-226. doi:10.1176/appi.psy.50.3.218 PubMed
22. American Psychiatric Association. Practice Guidelines for the Treatment of Psychiatric Disorders: Compendium 2006. Washington, DC: American Psychiatric Publishing; 2006.
23. Han CS, Kim YK. A double-blind trial of risperidone and haloperidol for the treatment of delirium. Psychosomatics. 2004;45(4):297-301. PubMed
24. Lacasse H, Perreault MM, Williamson DR. Systematic review of antipsychotics for the treatment of hospital-associated delirium in medically or surgically ill patients. Ann Pharmacother. 2006;40(11):1966-1973. doi:10.1345/aph.1H241 PubMed
25. Lonergan E, Britton AM, Luxenberg J, et al. Antipsychotics for delirium. Cochrane Database Syst Rev. 2007;(2):CD005594. PubMed
26. Lonergan E, Luxenberg J, Areosa Sastre A, et al. Benzodiazepines for delirium. Cochrane Database Syst Rev. 2009;(1)CD006379.
27. Breitbart W, Marotta R, Platt MM, et al. A double-blind trial of haloperidol, chlorpromazine, and lorazepam in the treatment of delirium in hospitalized AIDS patients. Am J Psychiatry. 1996;153(2):231-237. PubMed
28. Menza MA, Murray GB, Holmes VF, et al. Controlled study of extrapyramidal reactions in the management of delirious, medically ill patients: intravenous haloperidol versus intravenous haloperidol plus benzodiazepines. Heart Lung. 1988;17(3):238-241. PubMed
29. Fernandez F, Levy JK, Mansell PW. Management of delirium in terminally ill AIDS patients. Int J Psychiatry Med. 1989;19(2):165-172. PubMed
30. Adams F, Fernandez F, Andersson BS. Emergency pharmacotherapy of delirium in the critically ill cancer patient. Psychosomatics. 1986;27(suppl):33-38. PubMed
31. Fogarty T, Murray GB. Psychiatric presentation of meperidine toxicity. J Clin Psychopharmacol. 1987;7(2):116-117. PubMed
32. Choi DW, Viseskul V. Opioids and non-opioid enantiomers selectively attenuate N-methyl-d-aspartate neurotoxicity on cortical neurons. Eur J Pharmacol. 1988;155(1-2):27-35. doi:10.1016/0014-2999(88)90399-8 PubMed
33. Smith MT. Neuroexcitatory effects of morphine and hydromorphone: evidence implicating the 3-glucuronide metabolites. Clin Exp Pharmacol Physiol. 2000;27(7):524-528. doi:10.1046/j.1440-1681.2000.03290.x PubMed
34. Bourgeois JA, Koike AK, Simmons JE, et al. Adjunctive valproic acid for delirium and/or agitation on a consultation-liaison service: a report of six cases. J Neuropsychiatry Clin Neurosci. 2005;17(2):232-238. PubMed
35. Lindenmayer JP, Kotsaftis A. Use of sodium valproate in violent and aggressive behaviors: a critical review. J Clin Psychiatry. 2000;61(2):123-128. PubMed
36. Siegel MD. Management of agitation in the intensive care unit. Clin Chest Med. 2003;24(4):713-725. doi:10.1016/S0272-5231(03)00104-7 PubMed
37. Kotani Y, Shimazawa M, Yoshimura S, et al. The experimental and clinical pharmacology of propofol, an anesthetic agent with neuroprotective properties. CNS Neurosci Ther. 2008;14(2):95-106. doi:10.1111/j.1527-3458.2008.00043.x PubMed
38. Guindon J, LoVerme J, Piomelli D, et al. The antinociceptive effects of local injections of propofol in rats are mediated in part by cannabinoid CB1 and CB2 receptors. Anesth Analg. 2007;104(6):1563-1569.doi:10.1213/01.ane.0000263278.05423.a3 PubMed
39. Carollo DS, Nossaman BD, Ramadhyani U. Dexmedetomidine: a review of clinical applications. Curr Opin Anaesthesiol. 2008;21(4):457-461. doi:10.1097/ACO.0b013e328305e3ef PubMed
40. Maldonado JR, Wysong A, van der Starre PJ, et al. Dexmedetomidine and the reduction of postoperative delirium after cardiac surgery. Psychosomatics. 2009;50(3):206-217. doi:10.1176/appi.psy.50.3.206 PubMed
41. Darrouj J, Puri N, Prince E, et al. Dexmedetomidine infusion as adjunctive therapy to benzodiazepines for acute alcohol withdrawal. Ann Pharmacother. 2008;42(11):1703-1705. doi:10.1345/aph.1K678 PubMed
42. Rovasalo A, Tohmo H, Aantaa R, et al. Dexmedetomidine as an adjuvant in the treatment of alcohol withdrawal delirium: a case report. Gen Hosp Psychiatry. 2006;28(4):362-363. doi:10.1016/j.genhosppsych.2006.03.002 PubMed
43. Fricchione GL, Nejad SH, Esses JA, et al. Postoperative delirium. Am J Psychiatry. 2008;165(7):803-812. doi:10.1176/appi.ajp.2008.08020181 PubMed
44. Tesar GE, Murray GB, Cassem NH. Use of high-dose intravenous haloperidol in the treatment of agitated cardiac patients. J Clin Psychopharmacol. 1985;5(6):344-347. PubMed
45. Seitz DP, Gill SS. Neuroleptic malignant syndrome complicating antipsychotic treatment of delirium or agitation in medical and surgical patients: case reports and a review of the literature. Psychosomatics. 2009;50(1):8-15. doi:10.1176/appi.psy.50.1.8 PubMed
46. Caroff SN, Mann SC. Neuroleptic malignant syndrome. Med Clin North Am. 1993;77(1):185-202. PubMed
47. Trollor JN, Chen X, Sachdev PS. Neuroleptic malignant syndrome associated with atypical antipsychotic drugs. CNS Drugs. 2009;23(6):477-492. doi:10.2165/00023210-200923060-00003 PubMed
48. Ananth J, Parameswaran S, Gunatilake S, et al. Neuroleptic malignant syndrome and atypical antipsychotic drugs. J Clin Psychiatry. 2004;65(4):464-470. PubMed
49. US Food and Drug Administration. Center for Drug Evaluation and Research Psychopharmacological Drugs Advisory Committee. Meeting transcript (for the approval of Zeldox [ziprasidone]); Bethesda, MD; July 19, 2000. Available at: www.fda.gov/ohrms/dockets/ac/00/transcripts/3619t1a.pdf .
50. Haddad PM, Anderson IM. Antipsychotic-related QTc prolongation, torsade de pointes and sudden death. Drugs. 2002;62(11):1649-1671. doi:10.2165/00003495-200262110-00006 PubMed
51. Gupta A, Lawrence AT, Krishnan K, et al. Current concepts in the mechanisms and management of drug-induced QT prolongation and torsade de pointes. Am Heart J. 2007;153(6):891-899. doi:10.1016/j.ahj.2007.01.040 PubMed
52. Riker RR, Shehabi Y, Bokesch PM. Dexmedetomidine vs midazolam for sedation of critically ill patients: a randomized trial. JAMA. 2009;301(5):489-499. PubMed doi:10.1001/jama.2009.56
53. Pandharipande PP, Pun BY, Herr DL. Effect of sedation with dexmedetomidine vs lorazepam on acute brain dysfunction in mechanically ventilated patients: the MENDS randomized controlled trial. JAMA. 2007;298(22):2644-2653. PubMed doi:10.1001/jama.298.22.2644
54. Socioeconomic Status and Health Chartbook. US Department of Health and Human Services publication PHS 98-1232. Hyattsville, MD: US Department of Health and Human Services; 1998.
55. Ely EW, Margolin R, Francis J, et al. Evaluation of delirium in critically ill patients: validation of the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU). Crit Care Med. 2001;29(7):1370-1379. doi:10.1097/00003246-200107000-00012 PubMed
56. Ely EW, Siegel MD, Inouye SK. Delirium in the intensive care unit: an under-recognized syndrome of organ dysfunction. Semin Respir Crit Care Med. 2001;22(2):115-126. PubMed doi:10.1055/s-2001-13826
57. Ely EW, Inouye SK, Bernard GR, et al. Delirium in mechanically ventilated patients: validity and reliability of the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU). JAMA. 2001;286(21):2703-2710. doi:10.1001/jama.286.21.2703 PubMed
58. Ely EW, Gautam S, Margolin R, et al. The impact of delirium in the intensive care unit on hospital length of stay. Intensive Care Med. 2001;27(12):1892-1900. doi:10.1007/s00134-001-1132-2 PubMed
59. Ely EW, Shintani A, Bernard G, et al. Delirium in the IU is associated with prolonged length of stay in the hospital and higher mortality [abstract]. Am J Respir Crit Care Med. 2002;165:A23.
60. Milbrandt EB, Deppen S, Harrison PL, et al. Costs associated with delirium in mechanically ventilated patients. Crit Care Med. 2004;32(4):955-962. doi:10.1097/01.CCM.0000119429.16055.92 PubMed
61. Levkoff SE, Evans DA, Liptzin B, et al. Delirium: the occurrence and persistence of symptoms among elderly hospitalized patients. Arch Intern Med. 1992;152(2):334-340. doi:10.1001/archinte.152.2.334 PubMed
62. O’ Keefe S. The prevention of delirium. In: Lindesay J, Rockwood K, Macdonald A, eds. Delirium in Old Age. New York, NY: Oxford University Press; 2002.
63. Greene NH, Attix DK, Weldon BC, et al. Measures of executive function and depression identify patients at risk for postoperative delirium. Anesthesiology. 2009;110(4):788-795. doi:10.1097/ALN.0b013e31819b5ba6 PubMed
64. Inouye SK, Charpentier PA. Precipitating factors for delirium in hospitalized elderly persons: predictive model and interrelationship with baseline vulnerability. JAMA. 1996;275(11):852-857. doi:10.1001/jama.275.11.852 PubMed
65. Inouye SK, Bogardus ST Jr, Charpentier PA, et al. A multicomponent intervention to prevent delirium in hospitalized older patients. N Engl J Med. 1999;340(9):669-676. doi:10.1056/NEJM199903043400901 PubMed
66. Marcantonio ER, Flacker JM, Wright RJ, et al. Reducing delirium after hip fracture: a randomized trial. J Am Geriatr Soc. 2001;49(5):516-522. doi:10.1046/j.1532-5415.2001.49108.x PubMed
67. Kalisvaart KJ, de Jonghe JFM, Bogaards MJ, et al. Haloperidol prophylaxis for elderly hip surgery patients at risk for delirium: a randomized placebo-controlled study. J Am Geriatr Soc. 2005;53(10):1658-1666. doi:10.1111/j.1532-5415.2005.53503.x PubMed
68. Larsen KA, Kelly SE, Stern TA, et al. Administration of olanzapine to prevent postoperative delirium in elderly joint replacement patients: a randomized controlled study. Psychosomatics. In press.
69. Prakanrattana U, Prapaitrakool S. Efficacy of risperidone for prevention of postoperative delirium in cardiac surgery. Anaesth Intensive Care. 2007;35(5):714-719. PubMed
70. Siddiqi N, Stockdale R, Britton AM, et al. Interventions for preventing delirium in hospitalised patients. Cochrane Database Syst Rev. 2007;2(2):CD005563. PubMed
71. Inouye SK, Bogardus ST, Williams CS, et al. The role of adherence on the effectiveness of nonpharmacologic interventions: evidence from the delirium prevention trial. Arch Intern Med. 2003;163(8):958-964. doi:10.1001/archinte.163.8.958