Prim Care Companion CNS Disord 2022;24(6):21cr03198
To cite: Sadh K, Murthy MS, Dutta D, et al. Autoimmune encephalitis following severe traumatic brain injury. Prim Care Companion CNS Disord. 2022;24(6):21cr03198.
To share: https://doi.org/10.4088/PCC.21cr03198
© 2022 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, National Institute of Mental Health and Neurosciences, Bangalore, India
bDepartment of Neurology, National Institute of Mental Health and Neurosciences, Bangalore, India
*Corresponding author: Kamaldeep Sadh, MD, Department of Psychiatry, National Institute of Mental Health and Neurosciences, Bangalore, 560029, India ([email protected]).
Autoimmune encephalitis (AIE) presents with a wide range of psychiatric and neurologic problems, including behavioral changes, anxiety, agitation, personality changes, hallucinations, cognitive deterioration, and disorientation.1 Here, we report a case of AIE following severe traumatic brain injury (TBI), focusing on the challenges in diagnosis and management.
Case Report
A 50-year-old man with no contributory family or personal history presented to the emergency services of a tertiary neuropsychiatric hospital. He was brought to the hospital by his son for the treatment of his acute psychiatric symptoms. It was reported that 1½ months ago, he had a road traffic accident and sustained a severe head injury with hemorrhagic contusions in the bilateral basal frontal lobe and left anterior temporal lobes, intraparenchymal hematoma in the left basal frontal lobe, extradural hematoma in the left temporal region, and subdural hematoma in the left posterior parietal region. He had lost consciousness and was conservatively treated in the intensive care unit of a local hospital for 2 days and was discharged after 15 days on prophylactic antiepileptics (sodium valproate 500 mg/d, gabapentin 450 mg/d, and lacosamide 200 mg/d). Five days following discharge, he exhibited irritability, aimless wandering, crying spells, and confusion; was misidentifying family members; had an unsteady gait; and reported persecutory ideas. He was started on olanzapine 7.5 mg/d and quetiapine 200 mg/d by a psychiatrist; however, no improvement was noted. These complaints were fluctuating initially, with gradual worsening for 20 days when he presented to us with symptoms of increased aggression, urinary incontinence, and slurred speech and was admitted to the psychiatry inpatient unit.
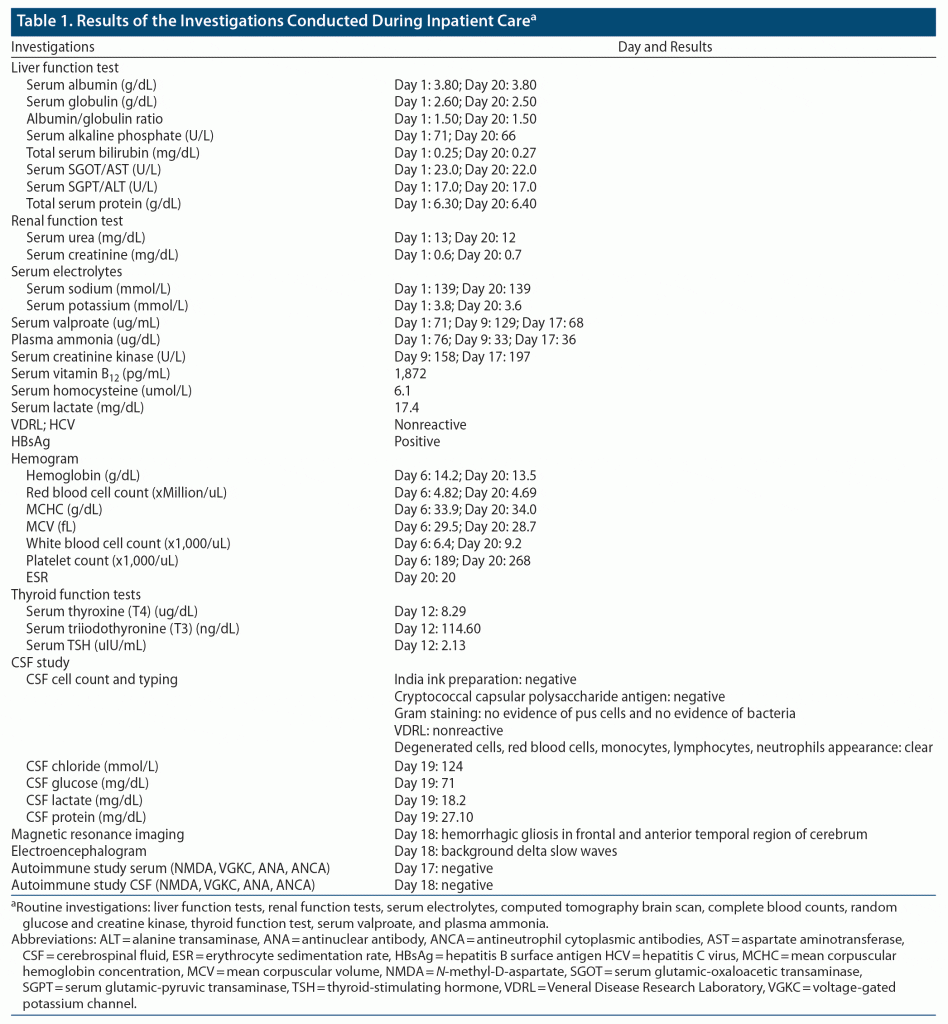
Routine investigations to rule out organic causes were within normal limits (Table 1). A computed tomography brain scan revealed bilateral frontal gliosis. Coronavirus disease 2019 testing (reverse transcription–polymerase chain reaction) was negative. He was started on tablet risperidone 2 mg at bedtime and tablet clonazepam 0.5 mg at bedtime. He continued to be disoriented to time, place, and person and was restless, dysarthric, and uncooperative for a detailed examination. He was initially diagnosed with post–head injury delirium and was treated with tablet valproate 1 g/d, risperidone 4 mg/d, trihexyphenidyl 2 mg/d, and clonazepam 1 mg/d (which was later tapered and stopped). On day 4 of his admission, there was mild improvement in irritability, and the Hindi Mental Status Examination (HMSE)2 score was 8/31, suggestive of cognitive impairment. Subsequently, given persisting disorientation and rigidity, risperidone was decreased to 3 mg/d and trihexyphenidyl to 1 mg/d. On day 8, he became drowsy with a reduced response to verbal commands. Repeat serum electrolytes were within normal limits. Valproate was reduced to 750 mg/d (sodium valproate = 129 μg/mL, plasma ammonia = 33 μg/dL) and risperidone to 1 mg/d. A neurology consultation was completed, and a serum autoimmune profile was advised, which was negative. Brain magnetic resonance imaging showed hemorrhagic gliosis in the bilateral frontal and anterior temporal lobe. Repeat serum valproate was 68 μg/mL. He continued to have disorientation, slurring of speech, hypomimia, and gait difficulty. Cerebrospinal fluid (CSF) analysis was normal. The CSF AIE panel was negative. The electroencephalogram showed diffuse bilateral slowing of background rhythm (Table 1). He was subsequently transferred to the neurology department. The possibility of AIE was considered, and the patient was started on 5 cycles of plasma exchange. After 3 cycles, 10%–20% improvement was noted in rigidity. Speech output had increased; however, fluctuating orientation continued. After 5 cycles, there was mild improvement in behavioral symptoms, rigidity, and gait, and he was discharged on the 10th day of his admission in the neurology department. The patient was discharged on tablet quetiapine 50 mg/d. No objective cognitive assessment was possible, as the patient continued to be uncooperative. Due to the pandemic and lockdown, video-based follow-up was done on the 28th day of discharge. Significant (80%–90%) improvement in irritability, gait, speech, and orientation was reported by the patient’s family members. The HMSE done through video consultation was 22 of 31. The patient continued to show progressive improvement after discharge, with no worsening or recurrence of symptoms.
Discussion
Given progressive improvements in our patient’s clinical condition in response to plasma exchange and after ruling out metabolic and infective causes of delirium and absence of improvement with psychotropics or after removal of valproate, a final diagnosis of AIE was considered. TBI is known to trigger an autoimmune antibody response with the central nervous system.3 TBI causes excitotoxicity, inflammation, oxidative damage, and synaptic injury. TBI may directly induce structural or functional brain changes in sensory and other information-processing networks, causing cognitive and motor disturbances. It has been shown that the N-methyl-D-aspartate receptor density is reduced in the neurons after a TBI to cope with the increase in extracellular excitatory neurotransmitters.4
However, only a few autoimmune antibodies can be tested. Autoimmune encephalitis can be caused by several unidentified autoantibodies, and TBI and stroke can trigger an autoimmune response, causing cognitive disturbances.
This case demonstrates that the differential diagnosis for post-TBI delirium should include autoimmune encephalitis in addition to evaluation for other causes. There are no reported cases, to our knowledge, of autoimmune encephalitis following severe TBI. Psychotic symptoms and the subacute onset of the cognitive disturbances following brain trauma may precipitate a psychiatric referral. In such situations, a high index of suspicion to new-onset neurologic symptoms or worsening of existing symptoms during the presentation without assumption of these as simply sequelae of the primary head injury become important. Hence, AIE should be considered in the differential diagnosis of subacute presentations of post-TBI delirium.
Published online: November 10, 2022.
Relevant financial relationships: None.
Funding/support: None.
Patient consent: Written informed consent was received from the patient’s son to publish the case report, and information has been de-identified to protect anonymity.
References (4)

- Leypoldt F, Armangue T, Dalmau J. Autoimmune encephalopathies. Ann N Y Acad Sci. 2015;1338(1):94–114. PubMed CrossRef
- Ganguli M, Ratcliff G, Chandra V, et al. A Hindi version of the MMSE: the development of a cognitive screening instrument for a largely illiterate rural elderly population in India. Int J Geriatr Psychiatry. 1995;10(5):367–377. CrossRef
- Javidi E, Magnus T. Autoimmunity after ischemic stroke and brain injury. Front Immunol. 2019;10:686. PubMed CrossRef
- Park Y, Luo T, Zhang F, et al. Downregulation of Src-kinase and glutamate-receptor phosphorylation after traumatic brain injury. J Cereb Blood Flow Metab. 2013;33(10):1642–1649. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!