A Case Report of Cobalamin Deficiency-Induced Mania in a Patient With Total Gastrectomy Due to Gastric Adenocarcinoma:
Further Insights Into the Pathogenesis of Mania
Cobalamin deficiency has hematologic, neurologic, and psychiatric sequelae.1 Toward this end, we present a patient with no prior psychiatric history who developed mania status post-gastrectomy for gastric cancer with an otherwise unremarkable workup save immeasurable cobalamin levels.
Case Report
Our patient is an 84-year-old black man with past medical history of total gastrectomy (TG) due to gastric adenocarcinoma in 2012 (on oral B12 supplementation since the time of TG) who presented to our hospital in August 2019 due to "altered mental status." The patient had paranoid delusions 3 days prior to presentation. Premorbid, he had no history of psychiatric illness, substance misuse, or recent medical illness and was normally alert, oriented, and able to live independently. Computed tomography (CT) of the head, electroencephalogram, and electrocardiogram were unremarkable. Further assessment was negative for any infectious, thyroid, or metabolic etiology save blood hemoglobin (hematocrit) level = 12.1 g/dL (37%), mean corpuscular volume = 102 µm3, serum vitamin B12 level < 150 pg/mL, serum homocysteine level = 108.4 µmol/L, and serum methylmalonic acid level = 3,445 µmol/L.
On our evaluation, the patient described his mood as "great." His affect was expansive but appropriate to thought content. At this time, he was oriented to person and place, but not time. He was distracted by irrelevant stimuli in the room and had pressured speech that was difficult to interrupt. He had a Young Mania Rating Scale (YMRS)2 score of 30, with a Mini-Mental State Examination (MMSE)3 score of 24.
We began treatment with ziprasidone 5 mg intramuscular (IM) twice daily and vitamin B12 IM 1,000 μg once daily in addition to oral B complex supplement. By day 4 after treatment began, the patient’s YMRS score was 19, and 3 days later it had reduced to 12; his YMRS score was 0 after 2 weeks of the aforementioned treatment, at which time he experienced no auditory/verbal hallucinations. Subsequently, ziprasidone was discontinued. At that time, our patient’s serum vitamin B12 level had returned to within normal limits (ie, 793 pg/mL). Magnetic resonance imaging of head occurred at this time but did not demonstrate any acute findings.
At discharge 24 days after admission, our patient was alert and oriented ×— 3 and had an MMSE score of 27 at that time. He was discharged on treatment with cyanocobalamin 1,000 μg/mL IM every week for 3 weeks and then once monthly. At 6-month follow-up, our patient’s symptoms showed no recurrence.
Discussion
Due to severe malabsorption, as in our patient, TG patients are at high risk of vitamin B12 deficiency.4 Nonetheless, the pathogenesis of vitamin B12-induced mood disorders is poorly understood, and that of mania even less well understood. While our patient did present with anemia and macrocytosis, about a fifth of patients presenting with nervous system disorders caused by vitamin B12 deficiency do not have anemia or macrocytosis.5
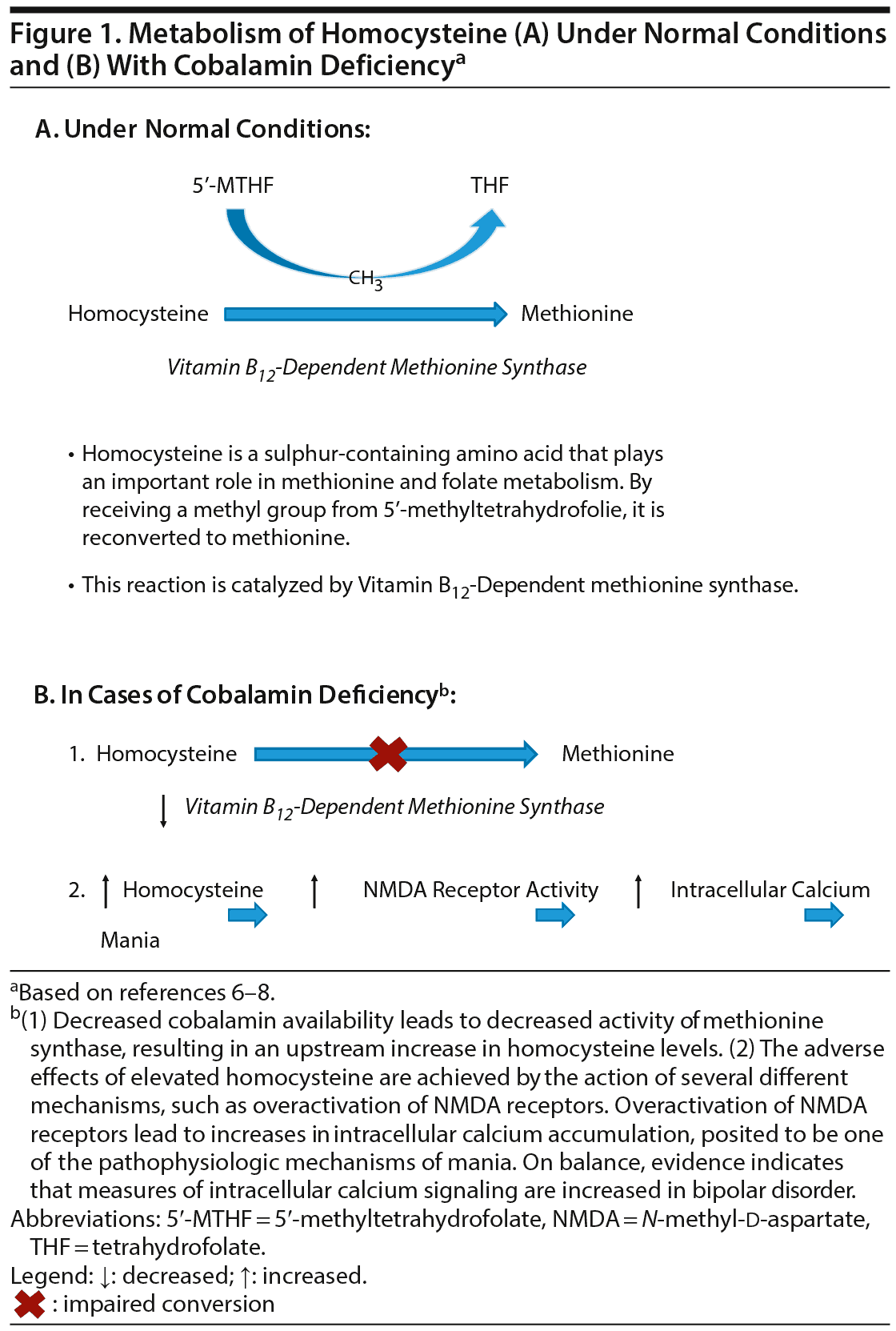
Our proposed pathogenic model of mania due to cobalamin deficiency (Figure 1)6-8 starts by addressing that vitamin B12 is a pivotal cofactor in 2 enzymatic reactions.9 Impaired activity of methionine synthase, as a consequence of vitamin B12 deficiency, will lead to accumulation of homocysteine. It has been proposed that alterations in glutamatergic neurotransmission may be the link between homocysteine and primary mania. Homocysteine could have a role as a partial excitatory agonist on the N-methyl-d-aspartate (NMDA) subtype of glutamate receptors and also on modulatory sites.10 Thus, long-term activation of NMDA receptors due to hyperhomocysteinemia could result in increased calcium ion influx and elevated intracellular second messenger calcium, similar to in primary mania.
Finally, our decision to select short-term intramuscular treatment was to ensure adherence in a medical unit unfamiliar with treating psychiatric symptoms. The advantage of intramuscular ziprasidone (compared to oral formulation) is that it eliminates bioavailability problems related to absorption and first-pass metabolism and maintains stable plasma concentrations. Ziprasidone bioavailability is significantly decreased when the drug is administered orally without food, and the drug undergoes first-pass metabolism at the liver. Thus, only a small portion of the dose reaches the systemic circulation. These bioavailability difficulties can be successfully overcome by the parenteral administration of the drug.11 Ziprasidone, in particular, was selected because of several clinical variables, including a less sedating profile than that of olanzapine and decreased risk of extrapyramidal symptoms compared to haloperidol.12
Given our proposed pathogenesis of mania secondary to cobalamin deficiency as related to partial agonism of NMDA glutamate receptors by accumulated homocysteine, a potential consideration for therapeutic benefit could have been to administer an NMDA antagonist to manage our patient’s manic/psychotic symptoms. A brief review of the literature found that although certain NMDA receptor antagonists are promising in the treatment of mania or psychosis, the available data for their use in these disorders is limited. Therefore, we elected not to treat with any NMDA receptor antagonists.13,14
In conclusion, we recommend that if a patient develops mania or a related neuropsychiatric disorder after TG, vitamin B12, methylmalonic acid, and total homocysteine levels should be measured and appropriate treatment subsequently initiated.
Published online: August 20, 2020.
Potential conflicts of interest: Dr Spiegel serves on the speakers’ bureaus for Allergen, Alkermers, and Otsuka but has no conflict of interest in preparation of this report. Drs Cho, Malik, Ramirez, Yuna, and McLean have no disclaimer or conflict of interest to report.
Funding/support: None.
Patient consent: When our patient was euthymic, he verbally agreed to allow us to publish this case report. He also consented under the premise that the information published was de-identified, which we strictly adhered to in this report.
REFERENCES
1.Lachner C, Steinle NI, Regenold WT. The neuropsychiatry of vitamin B12 deficiency in elderly patients. J Neuropsychiatry Clin Neurosci. 2012;24(1):5-15. PubMed CrossRef
2.Young RC, Biggs JT, Ziegler VE, et al. A rating scale for mania: reliability, validity and sensitivity. Br J Psychiatry. 1978;133(5):429-435. PubMed CrossRef
3.Folstein MF, Folstein SE, McHugh PR. "Mini-mental state": a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189-198. PubMed CrossRef
4.Bilici A, Sonkaya A, Ercan S, et al. The changing of serum vitamin B12 and homocysteine levels after gastrectomy in patients with gastric cancer: do they associate with clinicopathological factors? Tumour Biol. 2015;36(2):823-828. PubMed CrossRef
5.Reynolds E. Vitamin B12, folic acid, and the nervous system. Lancet Neurol. 2006;5(11):949-960. PubMed CrossRef
6.Robinson K. Homocysteine, B vitamins, and risk of cardiovascular disease. Heart. 2000;83(2):127-130. PubMed CrossRef
7.Djuric D, Jakovljevic V, Zivkovic V, et al. Homocysteine and homocysteine-related compounds: an overview of the roles in the pathology of the cardiovascular and nervous systems. Can J Physiol Pharmacol. 2018;96(10):991-1003. PubMed CrossRef
8.Harrison PJ, Geddes JR, Tunbridge EM. The emerging neurobiology of bipolar disorder. Trends Neurosci. 2018;41(1):18-30. PubMed CrossRef
9.Wolffenbuttel BHR, Wouters HJCM, Heiner-Fokkema MR, et al. The many faces of cobalamin (vitamin B12) deficiency. Mayo Clin Proc Innov Qual Outcomes. 2019;3(2):200-214. PubMed CrossRef
10.Gomez-Bernal GJ, Bernal-Perez M. Vitamin B12 deficiency manifested as mania: a case report. Prim Care Companion J Clin Psychiatry. 2007;9(3):238. PubMed CrossRef
11.Altamura AC, Sassella F, Santini A, et al. Intramuscular preparations of antipsychotics: uses and relevance in clinical practice. Drugs. 2003;63(5):493-512. PubMed CrossRef
12.Stip E, Zhornitsky S, Moteshafi H, et al. Ziprasidone for psychotic disorders: a meta-analysis and systematic review of the relationship between pharmacokinetics, pharmacodynamics, and clinical profile. Clin Ther. 2011;33(12):1853-1867. PubMed CrossRef
13.Zdanys K, Tampi RR. A systematic review of off-label uses of memantine for psychiatric disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(6):1362-1374. PubMed CrossRef
14.Kishi T, Iwata N. NMDA receptor antagonists interventions in schizophrenia: meta-analysis of randomized, placebo-controlled trials. J Psychiatr Res. 2013;47(9):1143-1149. PubMed CrossRef
aDepartment of Psychiatry and Behavioral Sciences, Eastern Virginia Medical School, Norfolk, Virginia
*Corresponding author: David R. Spiegel, MD, Eastern Virginia Medical School, Department of Psychiatry and Behavior Sciences, 825 Fairfax Ave, Norfolk, VA 23507 ([email protected]).
Prim Care Companion CNS Disord 2020;22(4):19l02565
To cite: Spiegel DR, Cho CM, Malik R, et al. A case report of cobalamin deficiency-induced mania in a patient with total gastrectomy due to gastric adenocarcinoma: further insights into the pathogenesis of mania. Prim Care Companion CNS Disord. 2020;22(4):19l02565.
To share: https://doi.org/10.4088/PCC.19l02565
© Copyright 2020 Physicians Postgraduate Press, Inc.
Enjoy this premium PDF as part of your membership benefits!