Objective: Patients with schizophrenia have increased risk of atherosclerotic diseases. It is already known that lifestyle-related disorders and the use of antipsychotics are closely related with the progression of atherosclerosis in psychiatric patients. Stroke as well as coronary heart disease play an important role in the cause of death in Asia and Japan. Thus, we studied the prevalence of cerebrovascular disease in psychiatric inpatients in Japan using brain magnetic resonance imaging (MRI).
Method: This cross-sectional study was performed from January 2012 to December 2013. Study participants were 152 hospitalized patients (61 men and 91 women) in the Department of Psychiatry at Kohnodai Hospital, National Center for Global Health and Medicine, Ichikawa City, Japan. Mean ages were 50.0 and 57.1 years old for men and women, respectively. The diagnoses (DSM-IV-TR criteria) of participants were schizophrenia (69.1%), mood disorder (18.4%), and other mental disorders (12.5%). We checked physical status, metabolic status of glucose and lipid levels, and brain MRI within 1 week of admission.
Results: The study group showed a significantly high prevalence of diabetes and low high-density lipoprotein (HDL) cholesterolemia in both sexes (n = 61 in men, n = 91 in women, P < .05). In the study group, serum fasting plasma glucose and hemoglobin A1c levels were significantly high (n = 152, P < .05), but serum HDL cholesterol and total cholesterol were significantly low in both sexes (n = 61 in men, n = 90 in women, P < .05), and triglycerides were low in men (n = 61, P < .05). Silent brain infarction was recognized at a higher rate (n = 98, P < .05) compared with healthy controls.
Conclusions: Participants in this study had an increased ratio of silent brain infarction compared with Japanese healthy controls, accompanied with higher ratios of diabetes and low HDL cholesterol.
Increased Silent Brain Infarction Accompanied
With High Prevalence of Diabetes and Dyslipidemia
in Psychiatric Inpatients: A Cross-Sectional Study
ABSTRACT
Objective: Patients with schizophrenia have increased risk of atherosclerotic diseases. It is already known that lifestyle-related disorders and the use of antipsychotics are closely related with the progression of atherosclerosis in psychiatric patients. Stroke as well as coronary heart disease play an important role in the cause of death in Asia and Japan. Thus, we studied the prevalence of cerebrovascular disease in psychiatric inpatients in Japan using brain magnetic resonance imaging (MRI).
Method: This cross-sectional study was performed from January 2012 to December 2013. Study participants were 152 hospitalized patients (61 men and 91 women) in the Department of Psychiatry at Kohnodai Hospital, National Center for Global Health and Medicine, Ichikawa City, Japan. Mean ages were 50.0 and 57.1 years old for men and women, respectively. The diagnoses (DSM-IV-TR criteria) of participants were schizophrenia (69.1%), mood disorder (18.4%), and other mental disorders (12.5%). We checked physical status, metabolic status of glucose and lipid levels, and brain MRI within 1 week of admission.
Results: The study group showed a significantly high prevalence of diabetes and low high-density lipoprotein (HDL) cholesterolemia in both sexes (n = 61 in men, n = 91 in women, P < .05). In the study group, serum fasting plasma glucose and hemoglobin A1c levels were significantly high (n = 152, P < .05), but serum HDL cholesterol and total cholesterol were significantly low in both sexes (n = 61 in men, n = 90 in women, P < .05), and triglycerides were low in men (n = 61, P < .05). Silent brain infarction was recognized at a higher rate (n = 98, P < .05) compared with healthy controls.
Conclusions: Participants in this study had an increased ratio of silent brain infarction compared with Japanese healthy controls, accompanied with higher ratios of diabetes and low HDL cholesterol.
Prim Care Companion CNS Disord 2015;17(2):doi:10.4088/PCC.14m01713
© Copyright 2015 Physicians Postgraduate Press, Inc.
Submitted: August 18, 2014; accepted October 22, 2014.
Published online: March 26, 2015.
Corresponding author: Tetsuto Kanzaki, MD, PhD, 15-8, Shiomi-cho, Choshi City, Chiba 288-0025, Japan ([email protected]).
The average life expectancy of patients with schizophrenia is approximately 15 years shorter than that of the general population in the United States.1 Coronary heart disease is the cause of more than 50% of deaths in patients with schizophrenia in the United States.1 Crump et al2 reported that the leading causes of death in people with schizophrenia in Sweden were cardiovascular disease and cancer. These results indicate that patients with schizophrenia have the tendency to suffer from atherosclerotic diseases.
Hypertension, diabetes mellitus, dyslipidemia, visceral-type obesity, and smoking are risk factors for atherosclerosis. There are many reports of a high prevalence of diabetes in patients with schizophrenia.3–6 The causes of diabetes are thought to be related to these patients’ lifestyles, which include unhealthy eating habits, shortage of exercise, and smoking.7–9 Schizophrenic patients also have a high incidence of dyslipidemia.10 Sasaki et al11 reported that serum high-density lipoprotein (HDL) cholesterol (HDL-C) decreased in patients with schizophrenia. Furthermore, Sugawara et al12 reported that the prevalence of metabolic syndrome was higher in Japanese schizophrenic patients under age 60 years old compared with the general population. It is also reported that the prevalence of smoking is higher in these patients than in the general population.1,13
Most schizophrenic patients are administered typical or atypical antipsychotics. Prah et al14 reported that, in 2007 in the United Kingdom, 15.0% of the prescriptions were for typical antipsychotics and 51.2% were for atypical antipsychotics among schizophrenic patients in primary care. It was also reported that antipsychotic prescriptions changed from typical antipsychotics (1997: 71.7%, 1999: 25.2%, 2002: 5.7%) to atypical antipsychotics for patients with mental disorders among Texas veterans.15 In Japanese schizophrenic inpatients in 2008,16 44.2% received typical antipsychotics and 55.8% received atypical antipsychotics. Some atypical antipsychotics cause adverse effects on metabolism, such as diabetes and dyslipidemia.4,17 These side effects also increase risks for atherosclerosis.
Cancer, heart disease, and cerebrovascular disease are main causes of death in the general population in Japan. Saku et al18 reported that the standardized mortality ratio of cancer in Japanese patients with schizophrenia, followed up from 1982 to 1985, was almost the same as the general population. However, there are no reports that state the mortality rates of coronary heart disease and strokes in schizophrenic patients in Japan. Schizophrenic patients under 45 years old demonstrated a 2-fold increased risk of developing strokes compared with controls in Taiwan.19 Therefore, it is probable that strokes as well as coronary heart disease have an essential role in cause of death and quality of life in the schizophrenic patients of Japan and the Asia-Pacific region.20
Magnetic resonance imaging (MRI) of the brain is commonly used in the diagnosis of stroke in Japan. Silent brain infarction (SBI) is described as lesions imaged by MRI as cerebral infarctions but without any recognized clinical symptoms and signs. SBI frequently occurs in healthy elderly individuals and is thought to more than double the risk of subsequent stroke, dementia, and cognitive decline.21,22
In the present study, we investigated brain MRI and lipid and glucose metabolism of psychiatric inpatients in Japan. High prevalence of SBI with diabetes and dyslipidemia was reported.
METHOD
Study Subjects
This study was performed from January 2012 to December 2013 at Kohnodai Hospital, National Center for Global Health and Medicine, Ichikawa City, Japan. Study participants were 152 hospitalized patients (61 men and 91 women) in the Department of Psychiatry at Kohnodai Hospital. The psychiatric diagnoses (per DSM-IV-TR criteria) of participants were schizophrenia, brief psychiatric disorders, and schizoaffective disorders (n = 105, 69.1%); mood disorders (n = 28, 18.4%); and other mental disorders (Alzheimer’s disease, stimulant psychosis, and somatoform disorders) (n = 19, 12.5%). Ninety-seven patients were diagnosed with schizophrenia, 6 with brief psychiatric disorders, 2 with schizoaffective disorders, 18 with bipolar disorders, and 10 with major depressive disorders. Mean ages of the patients were 50.0 and 57.1 years for men and women, respectively.
The study protocol was approved by the Ethics Committees of Chiba Institute of Science, Choshi City, Japan, and National Center for Global Health and Medicine, Ichikawa City, Japan. All participants were provided with a written informed consent form, and explanation and participation agreement were performed in accordance with the Declaration of Helsinki principles.
Diagnosis of Study Participants
The psychiatric diagnoses were made using DSM-IV-TR criteria. The definition of hypertension was systolic blood pressure ≥ 140 mm Hg and/or diastolic blood pressure ≥ 90 mm Hg. Diabetes mellitus was defined as hemoglobin A1c (HbA1c) ≥ 6.5% and fasting plasma glucose ≥ 126 mg/dL. High low-density lipoprotein (LDL) cholesterolemia (fasting serum LDL cholesterol [LDL-C] ≥ 140 mg/dL) or low high-density lipoprotein (HDL) cholesterolemia (fasting serum HDL cholesterol [HDL-C] < 40 mg/dL) or hypertriglyceridemia (fasting serum triglyceride ≥ 150 mg/dL) are described as dyslipidemia. Patients are also counted as having hypertension, diabetes, or dyslipidemia if they use antihypertensive (Ca antagonists, angiotensin receptor blockers, diuretics, and β-blockers) or hypoglycemic (insulins, biguanides, sulfonylureas, α-glucosidase inhibitors, thiazolidines, and dipeptidyl peptidase-4 [DDP-4] inhibitors) or antidyslipidemic drugs (statins, fibrates, and ezetimibe), respectively.
The diagnosis of metabolic syndrome was made using the definition of the Japan Society for the Study of Obesity.23 Metabolic syndrome was diagnosed when waist circumference (male ≥ 85 cm, female ≥ 90 cm) plus 2 of the following criteria were met: high blood pressure (≥ 130/85 mm Hg), reduced HDL-C (< 40 mg/dL) and/or raised triglycerides (≥ 150 mg/dL), and raised fasting hyperglycemia (≥ 110 mg/dL).23 Cerebral infarction was diagnosed by the presence of neurologic symptoms and signs corresponding to brain imaging. We counted a past history of coronary intervention as coronary heart disease. The definition of smokers was patients who smoked until 1 month before admission.
Data Collection
Patients’ demographic data and medical history were obtained from their medical records. Body mass index (BMI) was calculated by their height and weight. Waist circumference was measured at a level midway between the lowest rib and the iliac crest. Hospital staff measured blood pressure on the right arm of a patient before breakfast. Blood samples were obtained from patients after 12-hour starvation. We collected these data from patients within a week of admission.
Total cholesterol and triglycerides were assayed by enzymatic method, and HDL-C was assayed by direct method. LDL-C was calculated using the Friedewald formula from total cholesterol, triglycerides, and HDL-C (total cholesterol – triglycerides ÷ 5 – HDL-C). HbA1c was measured by the high-performance liquid chromatography method, and plasma glucose was measured by the enzymatic method.
To find the difference in glucose and lipid metabolism and risk factors of atherosclerosis between psychiatric patients and the healthy Japanese standard population, we used The National Health and Nutrition Survey in Japan 2011 produced by the Japanese Ministry of Health, Labor, and Welfare as the control group.26 This survey was performed in 7,047 people (3,230 were male, 3,817 were female) chosen at random from all districts of Japan in November 2011. The age range of the target population was from 1 to over 70 years old.
Imaging
The MRI examinations were performed in 5-mm thickness with 2-mm slice gap using a 1.5 Tesla MRI system (Siemens Magnetom Symphony, Siemens Medical Solutions USA Inc, Malvern, Pennsylvania). Ninety-eight patients underwent T1- and T2-weighted MRI and fluid-attenuated inversion recovery (FLAIR) of the brain as described previously.24 The FLAIR images were used to distinguish infarcts from dilated perivascular spaces. We diagnosed SBI as follows: (1) spotty area ≥ 3 mm in diameter showing high density in T2 and FLAIR images and low density in T1 image, (2) lack of neurologic signs explained by MRI lesions, and (3) no medical history of clinical symptoms of stroke.
The control group for SBI included 790 elderly volunteers (330 women and 460 men; mean age = 61.0 years; range, 40–88 years).25 All of the volunteers were living independently at home with no apparent history of stroke or dementia.
Statistics
Means or ratios in the control group were calculated by adjusting sex and age configuration to the patient group. Next, we estimated means or ratios with 95% CIs of the patient group. Then, we compared the patient group with the control group. All analyses were conducted using statistical software SAS, version 9.3 (SAS Institute, Inc, Cary, North Carolina).
RESULTS
Demographic Data
Table 1 shows the profile of study patients. We calculated the rate of hypertension, diabetes mellitus, dyslipidemia, and metabolic syndrome as complicated lifestyle-related diseases. The ratio of hypertension was significantly low in women compared with the Japanese standard (Table 2). Sex and age–adjusted diabetic ratio of the standard group was 0.099 and 0.073 in men and women, respectively. The 95% CI of diabetic ratio in the study group was 0.103–0.328 in men and 0.104–0.270 in women, indicating that prevalence of diabetes mellitus was significantly higher than the Japanese standard in both sexes (Table 2).
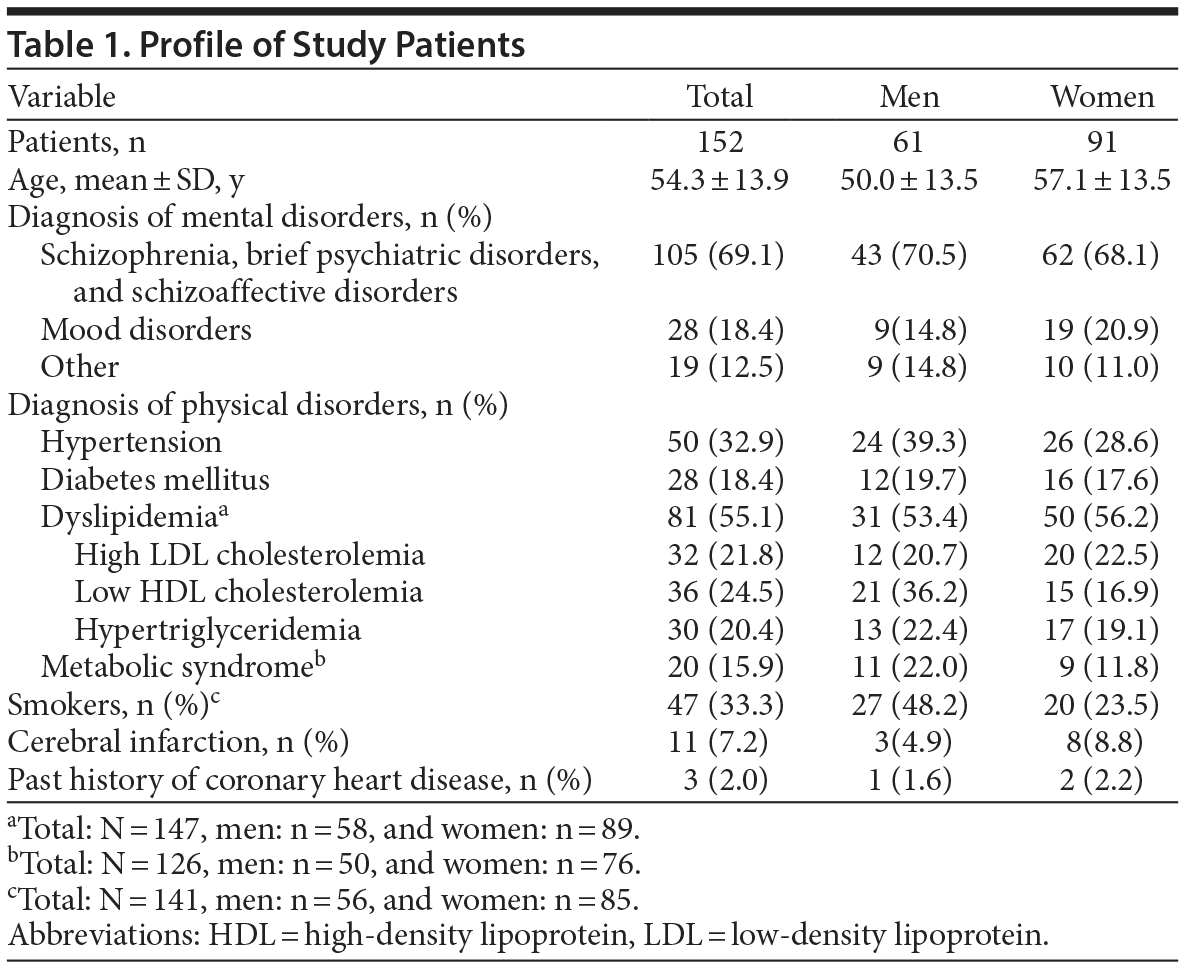
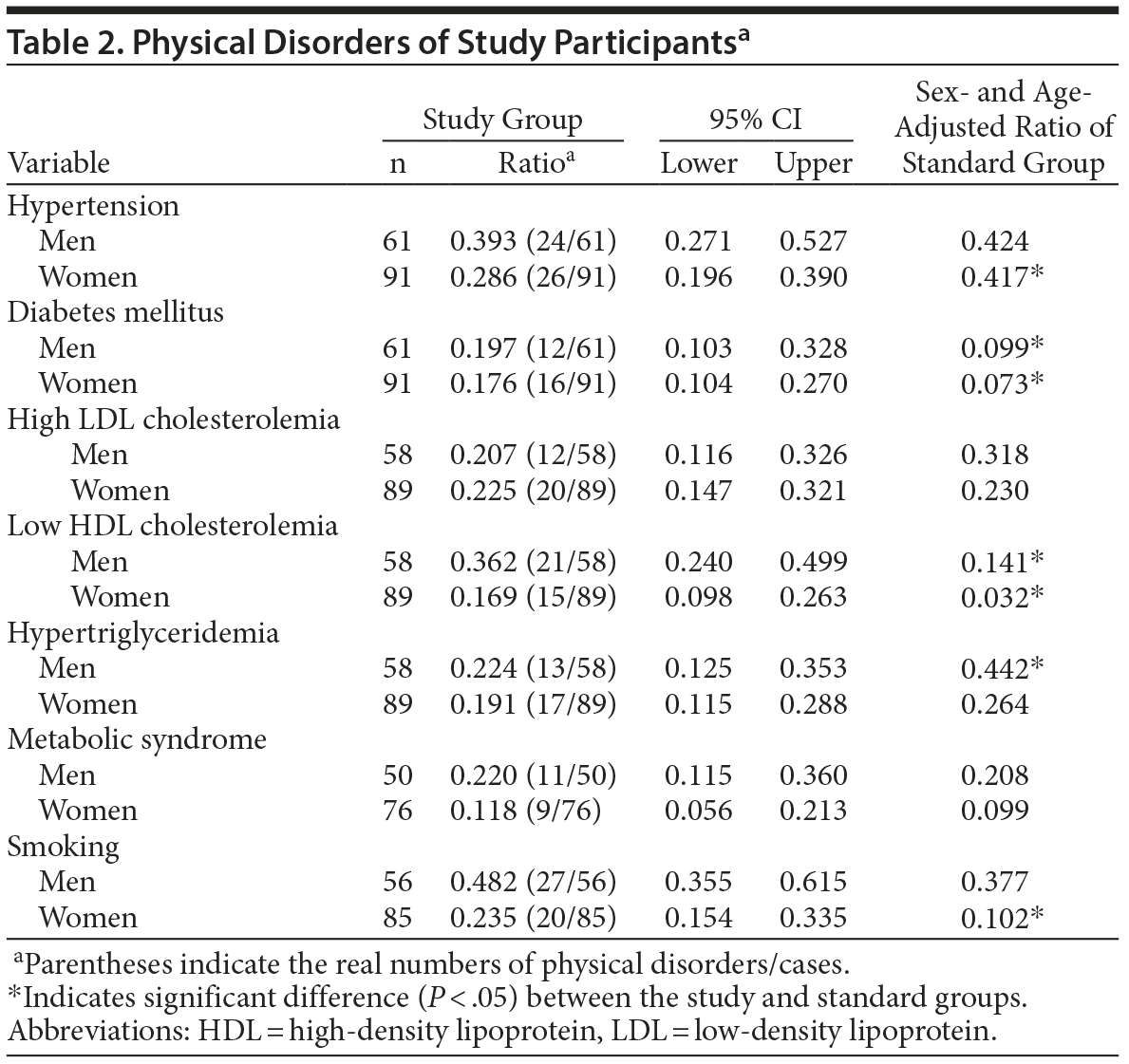
It is difficult to say whether the ratio of dyslipidemia is high or not because the definition of dyslipidemia is low HDL cholesterolemia (HDL-C < 40 mg/dL) or administration of antidyslipidemic agents in the National Health and Nutrition Survey in Japan 2011. However, the prevalence of low HDL-C was significantly higher in the study group than in the Japanese standard in both sexes as shown in Table 2. The ratio of high LDL-C was not significantly different between the study group and the Japanese standard population in both sexes, but the ratio of hypertriglyceridemia was significantly lower in men in the study group. The prevalence of metabolic syndrome in the study group was almost the same as the Japanese standard in both sexes as defined by the Japan Society for the Study of Obesity23 (Table 2).
Sex and age-adjusted prevalence of smoking in the standard group was 0.377 and 0.102 in men and women, respectively. The 95% CI of prevalence of smoking in the study group was 0.355–0.615 in men and 0.154–0.335 in women, indicating that prevalence of smoking was significantly higher in the study group than in the Japanese standard in women (Table 2).
Antipsychotics Mainly Administered
to Patients and Other Drugs Used
In the study group, 71.1% of patients took atypical antipsychotics, mainly risperidone, olanzapine, quetiapine, and clozapine; 19.1% took typical antipsychotics and 8.6% took other psychiatric medications (antidepressants and mood stabilizers) (Table 3). Therefore, over 90% of patients were treated with antipsychotics.

Twenty-four patients were treated with antihypertensive drugs. Eighteen patients were treated with Ca antagonists, 12 with angiotensin receptor blockers, 3 with β-blockers, and 1 with diuretics. Hypoglycemic drugs were administrated to 16 diabetic patients. Insulins were administered to 2 patients, DDP-4 inhibitors to 10 patients, thiazolidines to 5 patients, biguanides to 4 patients, and sulfonylureas and α-glucosidase inhibitors to 2 patients each. Eighteen patients took antidyslipidemic drugs, 15 took statins, 5 took ezetimibe, and 2 took fibrates. These drugs were used for more than 3 months.
Physical Characteristics of Study Patients
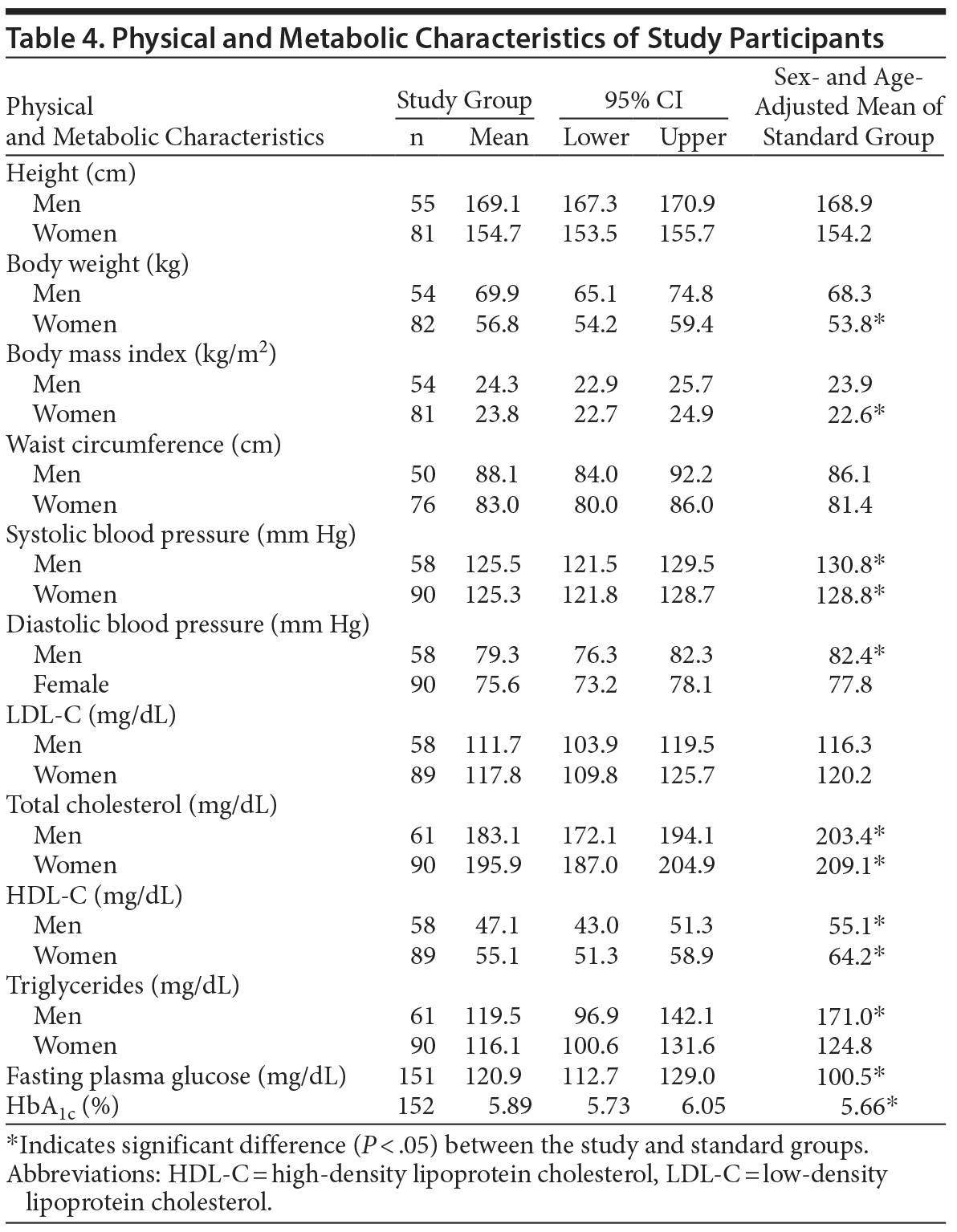
Table 4 shows physical characteristics of study patients. Body weight and BMI were significantly higher in female study group patients compared with the controls (P < .05). Waist circumference was not significantly different between the study group and the control group. Blood pressure was significantly lower (P < .05) in the study group except for diastolic blood pressure in women, suggesting that blood pressure was well controlled in the Kohnodai Hospital. Increased BMI often accompanies abnormal metabolism of lipid and glucose.
Variations in Blood Levels of Lipid and Glucose
Table 4 shows serum blood levels of lipid and glucose. Patients had significantly lower serum levels of total cholesterol and HDL-C compared with the controls, in accordance with a high ratio of low HDL cholesterolemia. Serum LDL-C levels were not significantly different between patients and controls. Male patients had significantly lower triglycerides than the male controls, but female patients did not. Fasting plasma glucose and HbA1c were significantly higher in patients than controls. This finding is in accordance with a high ratio of diabetes. Diabetes and low HDL cholesterolemia are well known to enhance atherosclerosis.
Increased Silent Brain Infarction in Psychiatric Patients
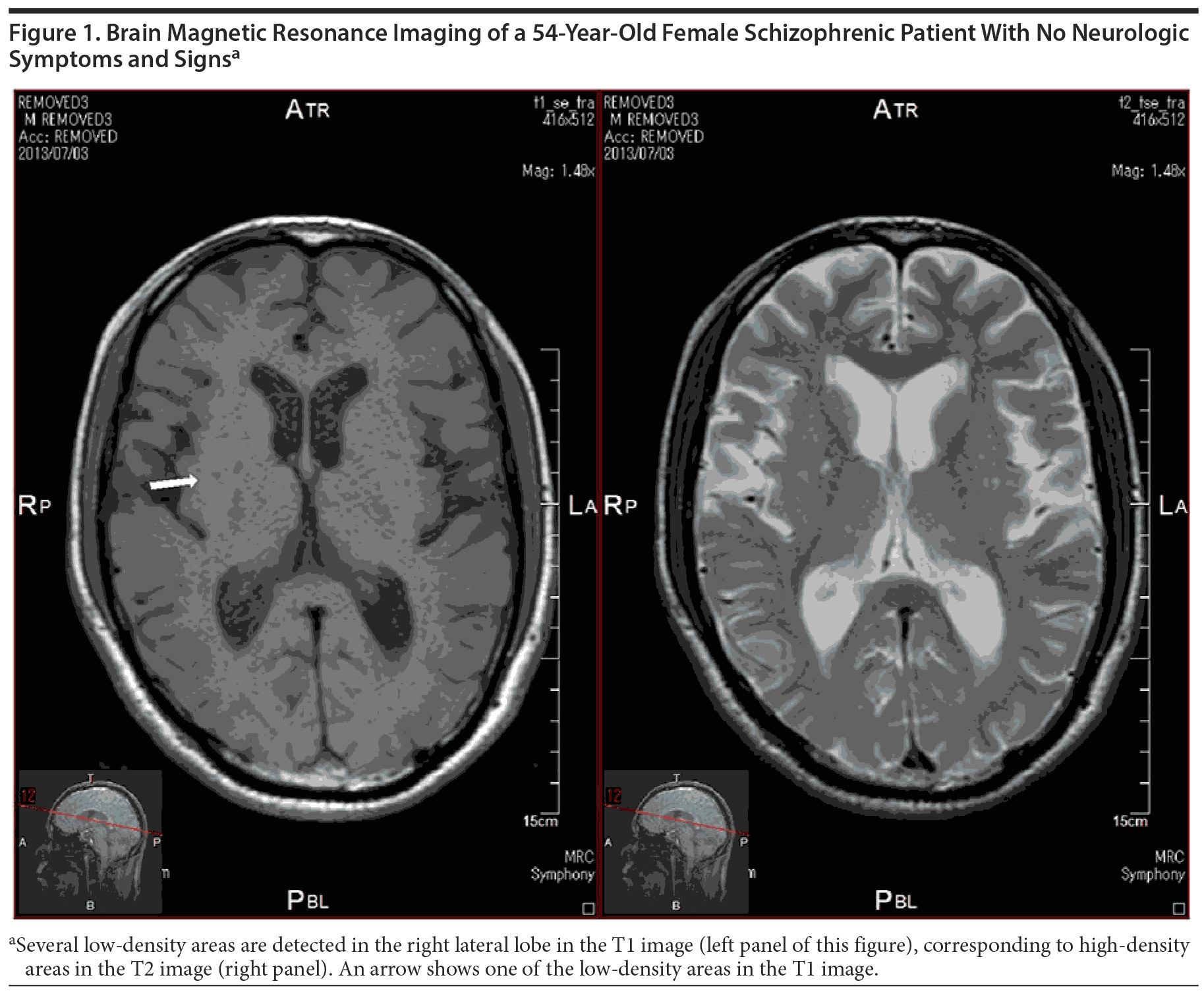
Figure 1 shows typical SBI in a 54-year-old schizophrenic female patient with no neurologic symptoms and signs. We can detect several low-density areas in the T1 image and corresponding high-density areas in the T2 image of the right lateral lobe.
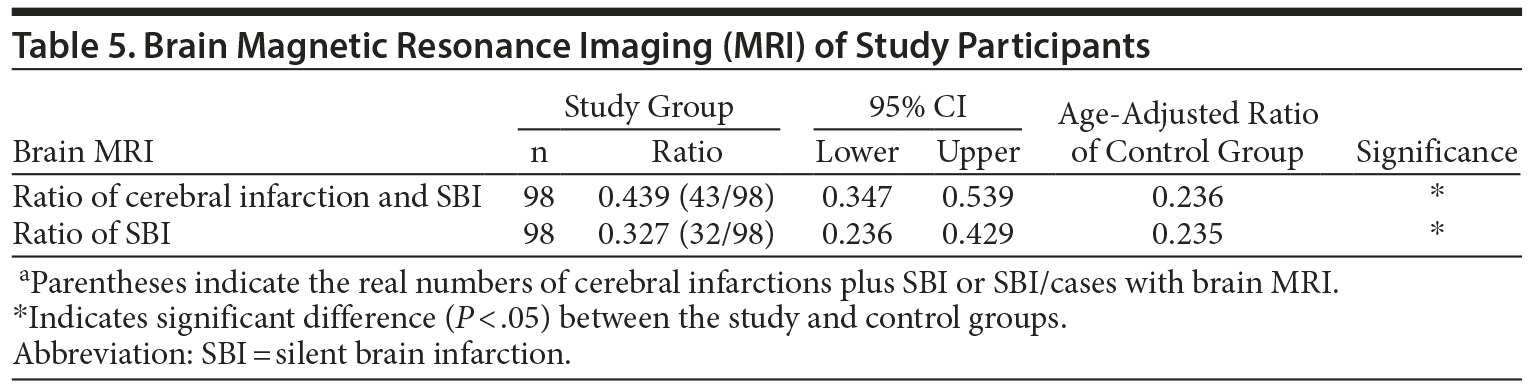
Eleven patients were diagnosed with cerebral infarction by the presence of neurologic symptoms and signs corresponding to brain imaging. Ninety-eight of 152 patients agreed to undergo brain MRI (Table 5). Age-adjusted ratios of SBI plus cerebral infarction and SBI in the healthy control group were 0.236 and 0.235, respectively. The SBI ratio of the Japanese healthy controls is 0.076 in 40–49 years, 0.116 in 50–59 years, 0.313 in 60–69 years, and 0.542 in > 70 years as described by Yoshida et al.25 The 95% CIs of SBI plus cerebral infarction and SBI in the study group were 0.347–0.539 and 0.236–0.429, respectively, indicating that these ratios were significantly higher in psychiatric patients.
DISCUSSION
The present study shows that psychiatric inpatients have increased SBI accompanied with high prevalence of diabetes and low HDL cholesterolemia. However, dyslipidemia, especially LDL-C and triglycerides, is different in each study. Tschoner et al27 and Murashita et al28 describe high serum levels of LDL-C and triglycerides in psychiatric patients. There are reports of low serum LDL-C and triglycerides in schizophrenic patients.29 These differences in LDL-C and triglyceride levels may be dependent on the situations of patients. Patients took different antipsychotics and had varied lifestyles, factors which were difficult to control in outpatients. Patients had no significant difference in LDL-C in both sexes in this study. Male patients showed significant low serum triglyceride levels, but female patients had no significant difference in serum triglycerides compared with controls. The level of serum triglycerides is greatly influenced by diet and alcohol.30 Triglyceride metabolisms may have changed in psychiatric patients with hospital admission through proper diet and exercise during their stay. Furthermore, the serum triglyceride level of the male Japanese standard was 171.0 mg/dL, higher than that of women (124.8 mg/dL). These findings are due to the decrease of serum triglyceride level in male patients.
There are many surveys on the causes of low HDL cholesterolemia.17,28 Lifestyle-related conditions such as diabetes, obesity, shortage of exercise, and smoking are causes of low HDL-C.31 Liver cirrhosis, malnutrition, and administration of probucol (antidyslipidemic agent) are also known to induce low HDL cholesterolemia,32 but this is not the case in our study because few patients had liver cirrhosis (1 patient) and malnutrition (1 patient had chronic obstructive pulmonary disease and colon carcinoma), and probucol was not used (data not shown). Serum HDL-C in patients with diabetes (total of 28 patients, 12 male and 16 female) is 50.1 mg/dL. Sex-adjusted HDL-C is 51.9 mg/dL in nondiabetic patients, indicating that approximately 2 mg/dL of serum HDL-C is decreased by diabetes. Obesity may not be the cause of low HDL cholesterolemia in male patients because BMI in male patients was not significantly different from the Japanese standard in our study. It is reported that smoking decreases serum HDL-C by 3.5 mg/dL and walking 6,000 steps/d increases HDL-C by 3 mg/dL.33,34 According to The National Health and Nutrition Survey in Japan 2011, 20.1% of the general Japanese population are smokers compared to 33.3% of patients in this study. Also, male patients were 8.0 mg/dL lower in HDL-C than the Japanese controls, and female patients were 9.1 mg/dL lower in our study. These findings suggest that it is difficult to explain low HDL cholesterolemia in psychiatric patients by lifestyle-related conditions only. Serum total cholesterol was lower in psychiatric patients compared with the Japanese controls. Serum total cholesterol level is calculated by LDL-C, HDL-C, and triglycerides. Psychiatric patients had low serum HDL-C in both sexes, almost the same levels of LDL-C, and low serum triglycerides in men, indicating that serum total cholesterol was low in psychiatric patients.
It is reported that antipsychotic users have an increased risk of cardiac mortality and all-cause mortality.35,36 In general, atypical antipsychotics have less neurologic side effects (extrapyramidal symptoms) but greater metabolic effects compared with typical antipsychotics.37 Several clinical studies showed that atypical antipsychotics, especially olanzapine and clozapine, had increased risks of diabetes and dyslipidemia, which are dependent on insulin resistance accompanied with weight gain or increase of visceral fat.4,17 However, there are some cases that have shown increased blood glucose and dyslipidemia without weight gain. Houseknecht et al38 reported that atypical antipsychotics directly induced whole-body insulin resistance in animal models, in accordance with diabetes and dyslipidemia without weight gain. Over 70% of patients used atypical antipsychotics in our study (Table 3). It is probable that atypical antipsychotics play an important role in the high rate of diabetes and dyslipidemia.
Coronary heart disease is the main cause of death in psychiatric patients in the United States and Western Europe.1,2 However, only 3 patients had a past history of coronary heart disease in our study. Smith et al3 reported that cardiovascular diseases are likely to be underrecognized and undertreated in people with schizophrenia. It is possible that symptoms related with coronary heart disease were masked in psychiatric patients in our study. Enhanced ratio of SBI is an important finding for future care of psychiatric patients because SBI increases the risk of subsequent cerebral infarction, dementia, and cognition decline.21,22 Cerebral infarction is known to be related with hypertension, diabetes, low HDL-C, and smoking.39–42 However, there is no clear evidence on the relation between SBI and low HDL-C.43 Indeed, this study does not show a significant difference of HDL-C between patients with and without SBI (52.4 mg/dL in HDL-C with SBI and 47.4 mg/dL in HDL-C without SBI). Most SBIs were lacunar infarction, which is closely related with hypertension but very little with cholesterol metabolism.43,44 In this study, systolic blood pressure was significantly low in patients with schizophrenia compared with the control group. The relationship between low HDL-C, diabetes, smoking, and SBI remains to be elucidated.
This current study also has some limitations. First, it was a cross-sectional study. It is necessary to perform a prospective study to clarify the cause-effect relationship between serum HDL-C, diabetes, smoking, and SBI in brain imaging. Second, patients in this study were inpatients. It is possible that lipid profiles in hospitalized patients, especially LDL-C and triglycerides, are different from profiles in outpatients. Third, the number of patients is too small to classify our results under each psychotic disorder, such as schizophrenia, bipolar disorders, depressive disorders, and anxiety disorders. Fourth, it is difficult to explain the mechanism of increased SBI in the study group, because it contains several kinds of psychotic disorders that have various backgrounds with mental and physical treatments. Finally, we need to check other atherosclerotic markers in psychotic patients for prevention of atherosclerosis.
In conclusion, psychiatric patients in this study had increased ratio of SBI and SBI plus cerebral infarction compared with the Japanese controls. Patients also showed high ratios of diabetes and low HDL cholesterolemia, mainly by the use of antipsychotics.
Drug names: aripiprazole (Abilify), clozapine (Clozaril, FazaClo, and others), ezetimibe (Zetia, Vytorin, and others), haloperidol (Haldol and others), lamotrigine (Lamictal and others), lithium (Lithobid and others), mirtazapine (Remeron and others), olanzapine (Zyprexa), paliperidone (Invega), quetiapine (Seroquel), risperidone (Risperdal and others).
Author affiliations: Department of Clinical Medicine (Drs Kanzaki and Mimori) and Department of Biostatistics (Dr Takizawa), Faculty of Pharmacy, Chiba Institute of Science, Choshi City; Department of Psychiatry (Drs Uju, Sekine, Ishii, Yoshimi, Yasui, Yasukawa, Sato, Okamoto, Hisaoka, Miura, Kusanishi, Murakami, Nakano, Mizuta, Hayakawa, and Tsukada) and Department of Internal Medicine (Dr Mishima), Kohnodai Hospital, National Center for Global Health and Medicine, Ichikawa City; and Amine Pharma Research Institute, Innovation Plaza at Chiba University, Chiba City (Dr Igarashi), Japan.
Potential conflicts of interest: None reported.
Funding/support: This study was funded by the Foundation of St Luke’s Life Science Institute, Tokyo, Japan.
Role of the sponsor: The Foundation of St Luke’s Life Science Institute contributed to the design and interpretation of this study.
Acknowledgments: We thank Yuichi Sugaya, MD, PhD, Department of Neurosurgery, Asahi Hospital, Asahi City, Chiba, Japan, for help in diagnosing silent brain infarction by brain magnetic resonance imaging.
Dr Sugaya reports no conflicts of interest related to the subject of this article.
REFERENCES
1. Hennekens CH, Hennekens AR, Hollar D, et al. Schizophrenia and increased risks of cardiovascular disease. Am Heart J. 2005;150(6):1115–1121. doi:10.1016/j.ahj.2005.02.007 PubMed
2. Crump C, Winkleby MA, Sundquist K, et al. Comorbidities and mortality in persons with schizophrenia: a Swedish national cohort study. Am J Psychiatry. 2013;170(3):324–333. doi:10.1176/appi.ajp.2012.12050599 PubMed
3. Smith DJ, Langan J, McLean G, et al. Schizophrenia is associated with excess multiple physical-health comorbidities but low levels of recorded cardiovascular disease in primary care: cross-sectional study. BMJ Open. 2013;3(4):e2808. doi:10.1136/bmjopen-2013-002808 PubMed
4. Newcomer JW, Ratner RE, Eriksson JW, et al. A 24-week, multicenter, open-label, randomized study to compare changes in glucose metabolism in patients with schizophrenia receiving treatment with olanzapine, quetiapine, or risperidone. J Clin Psychiatry. 2009;70(4):487–499. doi:10.4088/JCP.08m04132 PubMed
5. Okumura Y, Ito H, Kobayashi M, et al. Prevalence of diabetes and antipsychotic prescription patterns in patients with schizophrenia: a nationwide retrospective cohort study. Schizophr Res. 2010;119(1–3):145–152. doi:10.1016/j.schres.2010.02.1061 PubMed
6. Akhtar S, Kelly C, Gallagher A, et al. Newer antipsychotic agents,
carbohydrate metabolism and cardiovascular risk. Br J Diabetes Vasc Dis. 2004;4(5):303–309. doi:10.1177/14746514040040050301
7. Hu FB, Manson JE, Stampfer MJ, et al. Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. N Engl J Med. 2001;345(11):790–797. doi:10.1056/NEJMoa010492 PubMed
8. Hu FB, Sigal RJ, Rich-Edwards JW, et al. Walking compared with vigorous physical activity and risk of type 2 diabetes in women: a prospective study. JAMA. 1999;282(15):1433–1439. doi:10.1001/jama.282.15.1433 PubMed
9. Uchimoto S, Tsumura K, Hayashi T, et al. Impact of cigarette smoking on the incidence of Type 2 diabetes mellitus in middle-aged Japanese men: the Osaka Health Survey. Diabet Med. 1999;16(11):951–955. doi:10.1046/j.1464-5491.1999.00173.x PubMed
10. Watanabe J, Suzuki Y, Sugai T, et al. The lipid profiles in Japanese patients with schizophrenia treated with antipsychotic agents. Gen Hosp Psychiatry. 2012;34(5):525–528. doi:10.1016/j.genhosppsych.2012.04.002 PubMed
11. Sasaki J, Kumagae G, Sata T, et al. Decreased concentration of high-density lipoprotein cholesterol in schizophrenic patients treated with phenothiazines. Atherosclerosis. 1984;51(2–3):163–169. doi:10.1016/0021-9150(84)90164-3 PubMed
12. Sugawara N, Yasui-Furukori N, Sato Y, et al. Prevalence of metabolic syndrome among patients with schizophrenia in Japan. Schizophr Res. 2010;123(2–3):244–250. doi:10.1016/j.schres.2010.08.030 PubMed
13. Davidson S, Judd F, Jolley D, et al. Cardiovascular risk factors for people with mental illness. Aust N Z J Psychiatry. 2001;35(2):196–202. doi:10.1046/j.1440-1614.2001.00877.x PubMed
14. Prah P, Petersen I, Nazareth I, et al. National changes in oral antipsychotic treatment for people with schizophrenia in primary care between 1998 and 2007 in the United Kingdom. Pharmacoepidemiol Drug Saf. 2012;21(2):161–169. doi:10.1002/pds.2213 PubMed
15. Yang M, Barner JC, Lawson KA, et al. Antipsychotic medication utilization trends among Texas veterans: 1997–2002. Ann Pharmacother. 2008;42(9):1229–1238. doi:10.1345/aph.1L155 PubMed
16. Uno J, Tanifuji H, Shibata Y, et al. Survey of prescriptions in inpatients with schizophrenia in Japan: a nationwide multiple-center survey on prescriptions in 2008. Jpn J Clin Psychopharmacol. 2012;15:1231–1240.
17. Meyer JM, Koro CE. The effects of antipsychotic therapy on serum lipids: a comprehensive review. Schizophr Res. 2004;70(1):1–17. doi:10.1016/j.schres.2004.01.014 PubMed
18. Saku M, Tokudome S, Ikeda M, et al. Mortality in psychiatric patients, with a specific focus on cancer mortality associated with schizophrenia. Int J Epidemiol. 1995;24(2):366–372. doi:10.1093/ije/24.2.366 PubMed
19. Lin HC, Hsiao FH, Pfeiffer S, et al. An increased risk of stroke among young schizophrenia patients. Schizophr Res. 2008;101(1-3):234–241. doi:10.1016/j.schres.2007.12.485 PubMed
20. Zhang X, Patel A, Horibe H, et al; Asia Pacific Cohort Studies Collaboration. Cholesterol, coronary heart disease, and stroke in the Asia Pacific region. Int J Epidemiol. 2003;32(4):563–572. doi:10.1093/ije/dyg106 PubMed
21. Vermeer SE, Hollander M, van Dijk EJ, et al; Rotterdam Scan Study. Silent brain infarcts and white matter lesions increase stroke risk in the general population: the Rotterdam Scan Study. Stroke. 2003;34(5):1126–1129. doi:10.1161/01.STR.0000068408.82115.D2 PubMed
22. Vermeer SE, Longstreth WT Jr, Koudstaal PJ. Silent brain infarcts: a systematic review. Lancet Neurol. 2007;6(7):611–619. doi:10.1016/S1474-4422(07)70170-9 PubMed
23. Matsuzawa Y. Metabolic syndrome: definition and diagnostic criteria in Japan. J Atheroscler Thromb. 2005;12(6):301. doi:10.5551/jat.12.301 PubMed
24. Yoshida M, Tomitori H, Machi Y, et al. Acrolein, IL-6 and CRP as markers of silent brain infarction. Atherosclerosis. 2009;203(2):557–562. doi:10.1016/j.atherosclerosis.2008.07.022 PubMed
25. Yoshida M, Higashi K, Kobayashi E, et al. Correlation between images of silent brain infarction, carotid atherosclerosis and white matter hyperintensity, and plasma levels of acrolein, IL-6 and CRP. Atherosclerosis. 2010;211(2):475–479. doi:10.1016/j.atherosclerosis.2010.03.031 PubMed
26. The National Health and Nutrition Survey in Japan. Cancer Control and Health Promotion Division, Health Service Bureau, Ministry of Health, Labor and Welfare. Tokyo, Japan; 2011.
27. Tschoner A, Engl J, Rettenbacher M, et al. Effects of six second generation antipsychotics on body weight and metabolism: risk assessment and results from a prospective study. Pharmacopsychiatry. 2009;42(1):29–34. doi:10.1055/s-0028-1100425 PubMed
28. Murashita M, Inoue T, Kusumi I, et al. Glucose and lipid metabolism of long-term risperidone monotherapy in patients with schizophrenia. Psychiatry Clin Neurosci. 2007;61(1):54–58. doi:10.1111/j.1440-1819.2007.01610.x PubMed
29. Kingsbury SJ, Fayek M, Trufasiu D, et al. The apparent effects of ziprasidone on plasma lipids and glucose. J Clin Psychiatry. 2001;62(5):347–349. doi:10.4088/JCP.v62n0507 PubMed
30. Verdy M, Gattereau A. Ethanol, lipase activity, and serum lipid level. Am J Clin Nutr. 1967;20(9):997–1003. PubMed
31. Barter P. HDL-C: role as a risk modifier. Atheroscler suppl. 2011;12(3):267–270. doi:10.1016/S1567-5688(11)70885-6 PubMed
32. McPherson R, Hogue M, Milne RW, et al. Increase in plasma cholesteryl ester transfer protein during probucol treatment: relation to changes in high-density lipoprotein composition. Arterioscler Thromb. 1991;11(3):476–481. doi:10.1161/01.ATV.11.3.476 PubMed
33. Hata Y, Nakajima K. Life-style and serum lipids and lipoproteins. J Atheroscler Thromb. 2000;7(4):177–197. doi:10.5551/jat1994.7.177 PubMed
34. Garrison RJ, Kannel WB, Feinleib M, et al. Cigarette smoking and HDL cholesterol: the Framingham Offspring Study. Atherosclerosis. 1978;30(1):17–25. doi:10.1016/0021-9150(78)90149-1 PubMed
35. Ray WA, Chung CP, Murray KT, et al. Atypical antipsychotic drugs and the risk of sudden cardiac death. N Engl J Med. 2009;360(3):225–235. doi:10.1056/NEJMoa0806994 PubMed
36. Gill SS, Bronskill SE, Normand S-L, et al. Antipsychotic drug use and mortality in older adults with dementia. Ann Intern Med. 2007;146(11):775–786. doi:10.7326/0003-4819-146-11-200706050-00006 PubMed
37. Crossley NA, Constante M, McGuire P, et al. Efficacy of atypical v. typical antipsychotics in the treatment of early psychosis: meta-analysis. Br J Psychiatry. 2010;196(6):434–439. doi:10.1192/bjp.bp.109.066217 PubMed
38. Houseknecht KL, Robertson AS, Zavadoski W, et al. Acute effects of atypical antipsychotics on whole-body insulin resistance in rats: implications for adverse metabolic effects. Neuropsychopharmacology. 2007;32(2):289–297. doi:10.1038/sj.npp.1301209 PubMed
39. Arima H, Tanizaki Y, Yonemoto K, et al. Impact of blood pressure levels on different types of stroke: the Hisayama Study. J Hypertens. 2009;27(12):2437–2443. doi:10.1097/HJH.0b013e328330e882 PubMed
40. Doi Y, Ninomiya T, Hata J, et al. Impact of glucose tolerance status on development of ischemic stroke and coronary heart disease in a general Japanese population: the Hisayama Study. Stroke. 2010;41(2):203–209. doi:10.1161/STROKEAHA.109.564708 PubMed
41. Lindenstrøm E, Boysen G, Nyboe J. Influence of total cholesterol, high density lipoprotein cholesterol, and triglycerides on risk of cerebrovascular disease: the Copenhagen City Heart Study. BMJ. 1994;309(6946):11–15. doi:10.1136/bmj.309.6946.11 PubMed
42. Hata J, Kiyohara Y. Epidemiology of stroke and coronary artery disease in Asia. Circ J. 2013;77(8):1923–1932. doi:10.1253/circj.CJ-13-0786 PubMed
43. Das RR, Seshadri S, Beiser AS, et al. Prevalence and correlates of silent cerebral infarcts in the Framingham Offspring Study. Stroke. 2008;39(11):2929–2935. doi:10.1161/STROKEAHA.108.516575 PubMed
44. Takashima Y, Miwa Y, Mori T, et al. Sex differences in the risk profile and male predominance in silent brain infarction in community-dwelling elderly subjects: the Sefuri Brain MRI Study. Hypertens Res. 2010;33(7):748–752. doi:10.1038/hr.2010.69 PubMed
Enjoy this premium PDF as part of your membership benefits!


