Prim Care Companion CNS Disord 2021;23(3):20br02855
To cite: Gautam M, Patel S, Sablaban I. Intramuscular B52. Prim Care Companion CNS Disord. 2021;23(3):20br02855.
To share: https://doi.org/10.4088/PCC.20br02855
© Copyright 2021 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, Henry Ford Hospital, Detroit, Michigan
bDepartment of Psychiatry, Henry Ford Health System, Detroit, Michigan
*Corresponding author: Mohan Gautam, DO, MS, Department of Psychiatry, Henry Ford Hospital, 2799 West Grand Blvd, Detroit, MI 48202 ([email protected]).
As psychiatry residents, we necessarily encounter aggressive patients. Often, these patients respond to nonpharmacologic measures such as verbal de-escalation or to a less invasive pharmacologic measure such as an oral medication. Sometimes these measures fail, and a patient’s aggression is so severe that it necessitates use of an intramuscular agent. Across several training sites, we are often taught to order intramuscular (IM) haloperidol with IM lorazepam with IM diphenhydramine. This recommendation is so common that it even has its own name: B52 or Benadryl (diphenhydramine), haloperidol 5 mg, and lorazepam 2 mg.
This article stems from the widespread use of this cocktail. On the basis of many psychiatrists’ personal anecdotes, it seems to work well. But, does its teaching harm an approach to psychiatry that is evidence based?
Methods
We conducted a systematic literature review consistent with Preferred Reporting Items for Systematic Review Protocols guidelines. Inclusion criteria were adults with or without psychiatric conditions who were treated with an intramuscular agent for aggression. Only randomized controlled trials were included. We excluded children and adults with a primary neurologic disorder such as dementia. We excluded all studies that were not randomized controlled trials. PubMed was searched with the following MeSH terms: aggression AND randomized controlled trial. The initial search resulted in 1,481 articles. Duplicates were removed. A screening of the titles and abstracts with inclusion/exclusion criteria resulted in a total of 11 articles.1–11 Four articles were excluded because they analyzed medications not available in the United States (zuclupenthixol, levomepromazine, and flunitrazepam), resulting in 7 final articles for this review.1–4,7,8,11
Results
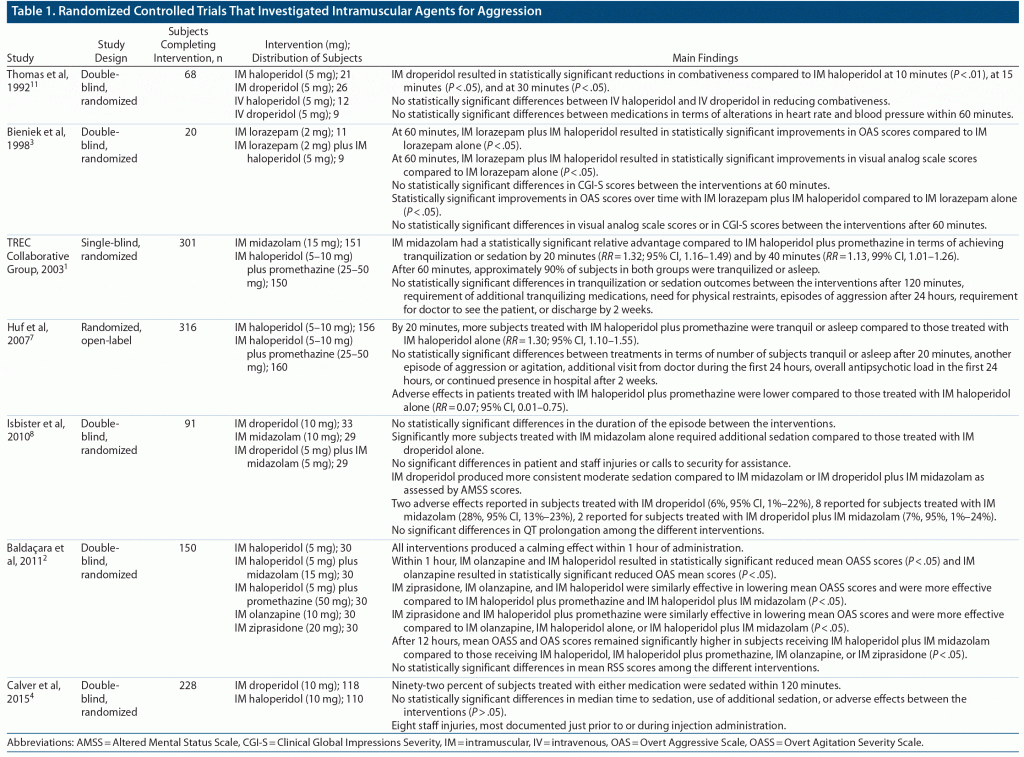
We were unable to identify any randomized controlled trials that have utilized intramuscular B52 for aggression. Table 1 summarizes the findings from randomized controlled trials that have investigated intramuscular agents for aggression. Intramuscular haloperidol, administered with or without midazolam, promethazine, or lorazepam demonstrates efficacy in the reduction of aggression. Other identified intramuscular agents that also demonstrate efficacy include lorazepam alone, midazolam alone, and droperidol with or without midazolam. Further, this review identifies available data on the use of second-generation antipsychotics as intramuscular agents for aggression in adults, primarily olanzapine and ziprasidone.
Discussion
There are many practices in psychiatry that do not necessarily warrant need for the quality of evidence afforded by randomized controlled trials. Intramuscular B52 may well be one such practice. We do not provide evidence, if such evidence even exists, that the addition of intramuscular diphenhydramine exposes patients to undue harm.
But we do question the utility of adding diphenhydramine to an intramuscular cocktail of medications that are already supported by data from randomized controlled trials. Perhaps diphenhydramine is added to prophylactically treat for acute dystonia? If so, its explicit use for this purpose should be clear.
Published online: May 20, 2021.
Potential conflicts of interest: None.
Funding/support: None.
References (11)

- TREC Collaborative Group. Rapid tranquillisation for agitated patients in emergency psychiatric rooms: a randomised trial of midazolam versus haloperidol plus promethazine. BMJ. 2003;327(7417):708–713. PubMed CrossRef
- Baldaçara L, Sanches M, Cordeiro DC, et al. Rapid tranquilization for agitated patients in emergency psychiatric rooms: a randomized trial of olanzapine, ziprasidone, haloperidol plus promethazine, haloperidol plus midazolam and haloperidol alone. Br J Psychiatry. 2011;33(1):30–39. PubMed CrossRef
- Bieniek SA, Ownby RL, Penalver A, et al. A double-blind study of lorazepam versus the combination of haloperidol and lorazepam in managing agitation. Pharmacotherapy. 1998;18(1):57–62. PubMed CrossRef
- Calver L, Drinkwater V, Gupta R, et al. Droperidol v. haloperidol for sedation of aggressive behaviour in acute mental health: randomised controlled trial. Br J Psychiatry. 2015;206(3):223–228. PubMed CrossRef
- Dorevitch A, Katz N, Zemishlany Z, et al. Intramuscular flunitrazepam versus intramuscular haloperidol in the emergency treatment of aggressive psychotic behavior. Am J Psychiatry. 1999;156(1):142–144. PubMed CrossRef
- Fuglum E, Schillinger A, Andersen JB, et al. Zuclopenthixol and haloperidol/levomepromazine in the treatment of elderly patients with symptoms of aggressiveness and agitation: a double-blind, multi-centre study. Pharmatherapeutica. 1989;5(5):285–291. PubMed
- Huf G, Coutinho ES, Adams CE; TREC Collaborative Group. Rapid tranquillisation in psychiatric emergency settings in Brazil: pragmatic randomised controlled trial of intramuscular haloperidol versus intramuscular haloperidol plus promethazine. BMJ. 2007;335(7625):869. PubMed CrossRef
- Isbister GK, Calver LA, Page CB, et al. Randomized controlled trial of intramuscular droperidol versus midazolam for violence and acute behavioral disturbance: the DORM study. Ann Emerg Med. 2010;56(4):392–401.e1. PubMed CrossRef
- Lavania S, Praharaj SK, Bains HS, et al. Efficacy and safety of levosulpiride versus haloperidol injection in patients with acute psychosis: a randomized double-blind study. Clin Neuropharmacol. 2016;39(4):197–200. PubMed CrossRef
- Taymeeyapradit U, Kuasirikul S. Comparative study of the effectiveness of zuclopenthixol acetate and haloperidol in acutely disturbed psychotic patients. J Med Assoc Thai. 2002;85(12):1301–1308. PubMed
- Thomas H Jr, Schwartz E, Petrilli R. Droperidol versus haloperidol for chemical restraint of agitated and combative patients. Ann Emerg Med. 1992;21(4):407–413. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!