Wernicke encephalopathy is a condition that classically presents with a triad of symptoms characterized by oculomotor dysfunction, cerebellar dysfunction, and delirium. The incidence of all 3 symptoms in patients is rare (≈17%), and many patients present with delirium alone.1 The altered cognition of Wernicke encephalopathy can progress to Wernicke-Korsakoff syndrome (WKS), with key neuropsychological features of anterograde amnesia and temporally graded retrograde amnesia.2 The clinical manifestation of antibody-mediated limbic encephalitis (ALE) is primarily defined by the subacute onset of short-term memory loss, seizures, confusion, and psychiatric symptoms, suggesting involvement of the limbic system.3 As both delirium and short-term memory impairment can be presenting symptoms in both conditions, the potential of misdiagnosis and delayed treatment can occur.1 We present the case of a patient recently treated for ALE, admitted to our facility for similar symptoms, and longitudinally diagnosed with WKS.
Case Report
Ms A was a 66-year-old white woman who presented to our emergency department in August 2019 with “altered mental status and short-term memory loss.” Three months earlier, she presented to an outside hospital with a similar chief complaint. At that time, cerebrospinal fluid (CSF) evaluation was remarkable for elevated protein and lymphocytic pleocytosis, although paraneoplastic panel results were unremarkable. Magnetic resonance imaging (MRI) of the brain demonstrated increased T2-weighted fluid-attenuated inversion recovery (FLAIR) involving bilateral temporal lobes, specifically moderately extensive abnormal increased T2 signal through the cortex and subjacent white matter of the anterior and anterior medial right temporal lobes, extending throughout the right hippocampus and contiguously into the right insular cortex. While some mild swelling of the anterior temporal lobe was possible, prominent atrophy appeared to be present. Atrophy was noted in the right mammillary bodies and anterior thalamic nuclei. She was diagnosed with ALE and treated with intravenous immunoglobulin (IVIg). Reportedly, her cognition returned to baseline level.
According to family and unbeknownst during the prior admission, Ms A had been misusing alcohol for 3 decades. Blood alcohol, urine drug screen, and thiamine levels were not assessed at admission despite repletion of the latter beginning on hospital day 1. When measured on hospital day 23, her thiamine level was within normal limits. Despite treatment, the patient remained confused, and we were consulted at that time.
On our evaluation, Ms A was oriented to person only, with vacillating mentation. The Confusion Assessment Method (CAM)4 for delirium was positive. As mentioned, MRI of the head was remarkable for extensive bilateral anterior/anteromedial temporal lobe disease, mainly gray matter. Otherwise, complete blood count, complete metabolic profile, C-reactive protein, and infectious workup results were unremarkable.
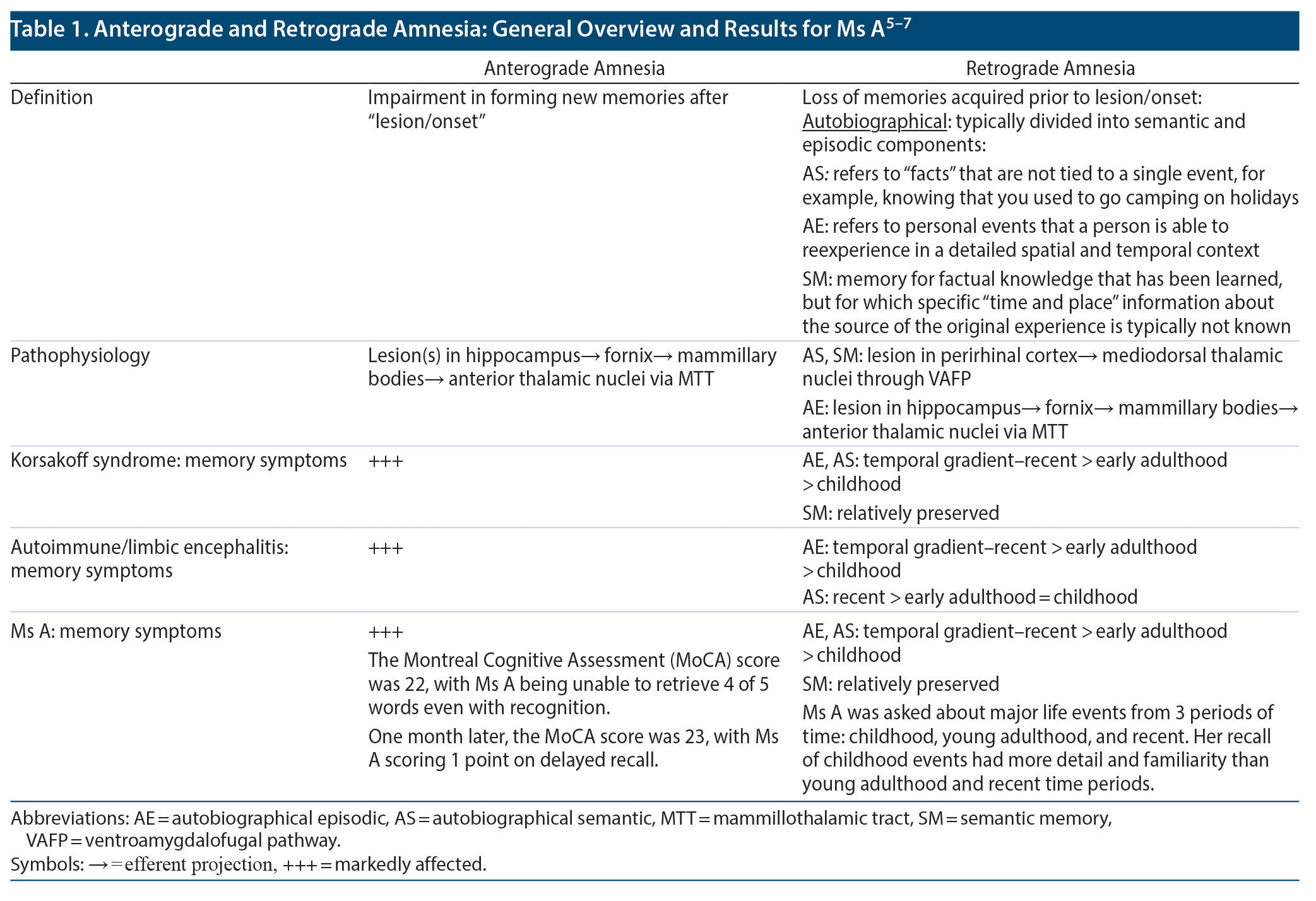
Ms A restarted a course of 5 IVIg treatments and then rituximab. Upon completion, the CAM was negative, although she had residual anterograde amnesia and retrograde amnesia. Table 1 shows the results of cognitive testing and follow-up.5-7 The patient was discharged on hospital day 40.
While details of Ms A’s prior hospitalization were scant, there was no history of ictal episodes or behavioral changes. Thus, it would appear that the patient’s primary symptoms for both prior and current admission were delirium followed by short-term memory deficits. Prior CSF evaluation was remarkable for elevated protein (elevated in ~50% of LE patients)8 and lymphocytic pleocytosis despite the paraneoplastic panel not identifying an antigenic target. Interestingly, it has been reported that in alcohol use disorder, ~6% of patients have a slightly elevated CSF protein (up to 75 mg/dL), while 2.3% have a moderately elevated level (up to 100 mg/dL).9 Our patient’s CSF protein level was 73.1 mg/dL.
MRI of the head at this admission was interpreted as improved but nonspecific temporal lobe disease, compatible with ALE. Most patients with ALE have unilateral or bilateral increased T2/FLAIR signal in the medial temporal lobes without contrast enhancement or abnormal diffusion-weighted images.10 Alternatively, diencephalon disease (mammillary body and thalamus) also described in the MRI of Ms A is more consistent with WKS. Neuroimaging in both ALE and KS has been reviewed elsewhere,11 although, in summary, the former has findings of unilateral/bilateral T2/FLAIR signals in the medial temporal lobe, while the latter has principle findings of atrophy in thalami, mammillary bodies, and the frontal cortex.12
Discussion
While WKS could explain Ms A’s symptoms, her cognition was reported by her family to have returned to baseline after IVIg treatments during the initial hospitalization, and improvement during our hospitalization after little change was noted with thiamine supplementation. First, it is possible that Ms A may have been experiencing the effects of both ALE and WKS. For instance, neuroimaging findings of anteromedial temporal and diencephalon disease support ALE and WKS, respectively. Second, as there was no objective evaluation of the patient’s cognition, such as the Mini-Mental State Examination, the reports by family were their subjective opinion. Additionally, Ms A’s presentation with delirium could have been that of Wernicke encephalopathy. Notably, even with thiamine supplementation, mental status changes and acute encephalopathy often gradually recede in Wernicke encephalopathy.13 Thus our patient’s cognitive improvement with IVIg could have been coincident with the predicted response course of Wernicke encephalopathy with thiamine. Finally, prolonged and excessive use of alcohol may lead to structural and functional brain damage, leading to alcohol-related dementia. The DSM-5 classifies this diagnosis as alcohol-induced major/mild neurocognitive disorder. Ms A presented at age 66 years with a 30-year history of alcohol use disorder. Thus, alcohol-related dementia also should be included in her differential diagnosis.14
Therefore, as our patient demonstrates, if ALE is not manifest in its full form, the symptoms are often not specific. Furthermore, most oligosymptomatic courses show a wide overlap with established neuropsychiatric conditions,15 although specific parts of our case that point toward the diagnosis of WKS include preservation of semantic memory, lack of behavioral disturbance, negative paraneoplastic panel, and lack of identifiable tumor. Therefore, combined with similarities in imaging and even CSF protein, KS should be considered in the differential diagnosis of ALE.6,7
Published online: October 8, 2020.
Potential conflicts of interest: Dr Spiegel is on the speakers’ bureau for Allergen, Alkermes, Otsuka, and IntraCellular but has no conflict of interest in preparation of this manuscript. Drs O’ Connell, Stocker, and Slater and Ms Spiegel report no conflicts of interest related to the subject of this report.
Funding/support: None.
Previous presentation: This manuscript was accepted as an abstract at the 2020 Annual Meeting of the American Psychiatric Association; April 25-29; Philadelphia, Pennsylvania.
Patient consent: The patient and her family verbally consented to publish this case report, and information has been de-identified to protect anonymity.
aDepartment of Psychiatry and Behavioral Sciences, Eastern Virginia Medical School, Norfolk, Virginia
*Corresponding author: David R. Spiegel, MD, Department of Psychiatry and Behavior Sciences, Eastern Virginia Medical School, 825 Fairfax Ave, Norfolk, VA 23507 ([email protected]).
Prim Care Companion CNS Disord 2020;22(5):20l02693
To cite: Spiegel DR, O’ Connell K, Stocker G, et al. A case of Wernicke-Korsakoff syndrome initially diagnosed as autoimmune limbic encephalitis: differential diagnosis of delirium and short-term memory deficits. Prim Care Companion CNS Disord. 2020;22(5):20l02693.
To share: https://doi.org/10.4088/PCC.20l02693
© Copyright 2020 Physicians Postgraduate Press, Inc.
REFERENCES
1.Chandrakumar A, Bhardwaj A, ‘ t Jong GW. Review of thiamine deficiency disorders: Wernicke encephalopathy and Korsakoff psychosis. J Basic Clin Physiol Pharmacol. 2018;30(2):153-162. PubMed CrossRef
2.Isenberg-Grzeda E, Kutner HE, Nicolson SE. Wernicke-Korsakoff-syndrome: under-recognized and under-treated. Psychosomatics. 2012;53(6):507-516. PubMed CrossRef
3.Asztely F, Kumlien E. The diagnosis and treatment of limbic encephalitis. Acta Neurol Scand. 2012;126(6):365-375. PubMed CrossRef
4.Inouye SK, van Dyck CH, Alessi CA, et al. Clarifying confusion: the Confusion Assessment Method: a new method for detection of delirium. Ann Intern Med. 1990;113(12):941-948. PubMed CrossRef
5.Alarcon R, Nalpas B, Pelletier S, et al. MoCA as a screening tool of neuropsychological deficits in alcohol-dependent patients. Alcohol Clin Exp Res. 2015;39(6):1042-1048. PubMed CrossRef
6.Rensen YCM, Kessels RPC, Migo EM, et al. Personal semantic and episodic autobiographical memories in Korsakoff syndrome: a comparison of interview methods. J Clin Exp Neuropsychol. 2017;39(6):534-546. PubMed CrossRef
7.Allen RJ. Classic and recent advances in understanding amnesia. F1000 Res. 2018;7:331. PubMed CrossRef
8.Lascano AM, Vargas MI, Lalive PH. Diagnostic tools for immune causes of encephalitis. Clin Microbiol Infect. 2019;25(4):431-436. PubMed CrossRef
9.Jurado R, Walker HK. Cerebrospinal fluid. In: Walker HK, Hall WD, Hurst JW, eds. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd ed. Boston, MA: Butterworths; 1990.
10.Armangue T, Leypoldt F, Dalmau J. Autoimmune encephalitis as differential diagnosis of infectious encephalitis. Curr Opin Neurol. 2014;27(3):361-368. PubMed CrossRef
11.Sechi G, Serra A. Wernicke’s encephalopathy: new clinical settings and recent advances in diagnosis and management. Lancet Neurol. 2007;6(5):442-455. PubMed CrossRef
12.Kopelman MD. What does a comparison of the alcoholic Korsakoff syndrome and thalamic infarction tell us about thalamic amnesia? Neurosci Biobehav Rev. 2015;54:46-56. PubMed CrossRef
13.Sinha S, Kataria A, Kolla BP, et al. Wernicke encephalopathy-clinical pearls. Mayo Clin Proc. 2019;94(6):1065-1072. PubMed CrossRef
14.Sachdeva A, Chandra M, Choudhary M, et al. Alcohol-related dementia and neurocognitive impairment: a review study. Int J High Risk Behav Addict. 2016;5(3):e27976. PubMed CrossRef
15.Steiner J, Pr×¼ss H, Köhler S, et al. Autoimmune encephalitis with psychosis: warning signs, step-by-step diagnostics and treatment. World J Biol Psychiatry. 2020;21(4):241-254. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!