Objective: Wernicke’s encephalopathy is caused by thiamine deficiency and occurs predominantly in alcohol-dependent individuals but also develops in those who are malnourished due to other reasons including medical and psychiatric disorders. This study examined the frequency rate and management of Wernicke’s encephalopathy in alcohol-dependent and non-alcohol-dependent patients admitted to a psychiatric hospital.
Methods: Data were retrospectively collected from electronic medical records of psychiatric inpatients admitted to a teaching hospital located in Texas between September 2013 and March 2014. The diagnostic criteria of Caine and colleagues and thiamine dosing strategies were used to identify cases of suboptimal management.
Results: A total of 486 charts were reviewed. Nine patients (1.85%) had clinical signs of Wernicke’s encephalopathy, and 36 (7%, n = 486) were at a high risk for developing the disorder. None of these patients received adequate doses of parenteral thiamine, and of those who were prescribed thiamine, the majority, including high-risk patients, were prescribed oral thiamine at the traditional dose of 100 mg/d.
Conclusions: The findings suggest that Wernicke’s encephalopathy is underdiagnosed and undertreated. Our study also highlights the need for clarifying diagnostic criteria, identifying the risk factors for thiamine deficiency, and improving awareness among physicians about diagnosis, prevention, and adequate treatment of Wernicke’s encephalopathy in alcohol-dependent and non-alcohol-dependent patients.

CME Background
Articles are selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME articles on a variety of topics throughout each volume. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the article, correctly answer the questions in the Posttest, and complete the Evaluation.
CME Objective
After studying this article, you should be able to:
‘ ¢ Identify patients with signs of Wernicke’s encephalopathy so that parenteral thiamine can be administered
Accreditation Statement
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Creditâ„¢. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Note: The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Creditâ„¢ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 1.0 hour of Category I credit for completing this program.
Date of Original Release/Review
This educational activity is eligible for AMA PRA Category 1 Creditâ„¢ through December 31, 2019. The latest review of this material was December 2017.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Larry Culpepper, MD, MPH, Editor in Chief, has been a consultant for Allergan, Ironshore, Lundbeck, Shire, and Sunovion; has been a stock shareholder of M-3 Information; and has received royalties from UpToDate and Oxford University Press. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears at the end of the article.
This CME activity is expired. For more CME activities, visit cme.psychiatrist.com.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
ABSTRACT
Objective: Wernicke’s encephalopathy is caused by thiamine deficiency and occurs predominantly in alcohol-dependent individuals but also develops in those who are malnourished due to other reasons including medical and psychiatric disorders. This study examined the frequency rate and management of Wernicke’s encephalopathy in alcohol-dependent and non-alcohol-dependent patients admitted to a psychiatric hospital.
Methods: Data were retrospectively collected from electronic medical records of psychiatric inpatients admitted to a teaching hospital located in Texas between September 2013 and March 2014. The diagnostic criteria of Caine and colleagues and thiamine dosing strategies were used to identify cases of suboptimal management.
Results: A total of 486 charts were reviewed. Nine patients (1.85%) had clinical signs of Wernicke’s encephalopathy, and 36 (7%, n = 486) were at a high risk for developing the disorder. None of these patients received adequate doses of parenteral thiamine, and of those who were prescribed thiamine, the majority, including high-risk patients, were prescribed oral thiamine at the traditional dose of 100 mg/d.
Conclusions: The findings suggest that Wernicke’s encephalopathy is underdiagnosed and undertreated. Our study also highlights the need for clarifying diagnostic criteria, identifying the risk factors for thiamine deficiency, and improving awareness among physicians about diagnosis, prevention, and adequate treatment of Wernicke’s encephalopathy in alcohol-dependent and non-alcohol-dependent patients.
Prim Care Companion CNS Disord 2017;19(6):17m02179
https://doi.org/10.4088/PCC.17m02179
© Copyright 2017 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, Texas Tech University Health Sciences Center, Lubbock, Texas
*Corresponding author: Medhat Guirguis, MD, Department of Psychiatry, Texas Tech University Health Sciences Center, 3601 4th St Stop 8103, Lubbock, TX 79430 ([email protected]).
Wernicke’s encephalopathy is a medical emergency1 resulting from thiamine deficiency.2,3 If left untreated, the disorder can lead to significant neurologic sequelae including Korsakoff syndrome or even death in up to 20% of cases.2 Wernicke’s encephalopathy is reversible if treated with adequate parenteral thiamine administered within the first 48-72 hours of the onset of symptoms.1,3
Wernicke’s encephalopathy is closely associated with alcohol use disorder and other psychiatric disorders including schizophrenia, mood disorders, and eating disorders.4-6 According to the National Survey on Drug Use and Health,7 in 2015 there were an estimated 9.8 million adults aged 18 or older with serious mental illness in the United States. Nearly 15.1 million adults aged 18 and older had alcohol use disorder, of whom 1.3 million received treatment at a specialized facility.7 An estimated 88,000 people die from alcohol-related causes annually, making alcohol the fourth leading preventable cause of death in the United States.8
The estimated prevalence of Wernicke’s encephalopathy has been calculated from a variety of retrospective autopsy studies.1,2,9-12 The prevalence rate of Wernicke’s encephalopathy is between 0.4% and 2.8%.4,9,10 The rates are slightly different worldwide, with higher rates in Australia (2.8%) and the United States (2.2%).1-4,9-12
Autopsy reports10,13,14 have shown that 75%-80% of Wernicke’s encephalopathy cases were not diagnosed before death. Particular patient populations have an increased prevalence rate compared to the general population. Autopsy reports10,13,14 show 12.5% of alcoholic, 10% of AIDS, and 6% of bone marrow transplant patients have Wernicke’s encephalopathy.
CAUSES OF THIAMINE DEFICIENCY
The average healthy person needs 1.2 mg of thiamine daily,15 and the healthy body’s storage of thiamine is limited to 30-50 mg. This storage can be depleted within 4 weeks if associated with inadequate intake, increased loss, or decreased absorption.16,17 Neuropsychiatric symptoms start to appear when the level of thiamine drops below 20% of the optimal level.11,18
Thiamine deficiency occurs predominantly in those with alcohol dependence but increasingly occurs in individuals who are malnourished from other causes, including hyperemesis gravidarum, HIV, gastric bypass, and malignancy.1 Alcohol-dependent individuals are susceptible to Wernicke’s encephalopathy due to inadequate nutritional intake, and the presence of ethanol in the body decreases the absorption of thiamine from the gastrointestinal tract. As documented by the World Health Organization,19 due to biopsychosocial factors,4-6 psychiatric patients are known to have increased risk for many chronic diseases and malnutrition, which contribute to the risk of developing Wernicke’s encephalopathy.

- Wernicke’s encephalopathy should be part of the differential diagnosis of all psychiatric patients meeting any of the criteria of Caine and colleagues.
- Until further guidelines are available, clinicians may apply various thiamine dosing strategies to prevent and treat Wernicke’s encephalopathy.
MISCONCEPTIONS OF DIAGNOSING WERNICKE’ S ENCEPHALOPATHY
There are several reasons for the underdiagnosis of Wernicke’s encephalopathy. Historically, clinicians suspect Wernicke’s encephalopathy in alcohol-dependent patients with the classic triad: altered mental status, ocular abnormalities, and ataxia. However, only a small percentage of patients exhibit all 3 elements.20 Autopsy-based studies21 showed that 82% of patients had altered mental status, 29% had ocular abnormalities, 23% had loss of balance, 19% had none of the traditional signs, and only 16% manifest all 3 classic symptoms.
Early signs and symptoms of Wernicke’s encephalopathy are vague and nonspecific. Patients report nausea, vomiting, weight loss, and sometimes memory loss. Even after progression of the disease, most patients manifest only one symptom of the triad. Altered mental status is the most common symptom of late-stage Wernicke’s encephalopathy, and because altered mental status is a symptom of many other neurologic conditions, the diagnosis is easy to miss. Thus, the ambiguity of symptoms leads to a missed diagnosis of Wernicke’s encephalopathy in both early and late stages of the illness.12,21-23
Wernicke’s encephalopathy is still being defined in contemporary alcohol misuse. Therefore, it is often missed in non-alcohol-dependent patients.4 Concomitant medical and psychiatric conditions, particularly if the patient has no apparent history of nutritional deficiency, can dissuade a clinician from diagnosing Wernicke’s encephalopathy.24
In psychiatric patients, Wernicke’s encephalopathy can mimic major depressive disorder with psychotic or catatonic features, psychotic disorders, or dementia,25-27 or it can present with agitation, hallucinations, and behavioral disturbances, mimicking acute psychotic disorders.28,29
MISCONCEPTIONS OF TREATING WERNICKE’ S ENCEPHALOPATHY
To our knowledge, there are no standardized national guidelines in the United States on how to prescribe thiamine to treat or prevent Wernicke’s encephalopathy. Hospital protocol for treatment of the disorder is not always established using evidence-based knowledge. There are still hospitals and physicians that use the traditional recommendation of an oral dosage of thiamine 100 mg once per day to treat chronic alcohol use with altered mental status. This habitual dosage of 50-100 mg was estimated in 1950,24 and no randomized, controlled clinical trial has studied the effectiveness of this regimen to date.
A randomized, double-blind, multidose trial30 was conducted concerning the therapeutic benefits of thiamine in an alcohol-dependent sample with no clinical signs of Wernicke’s encephalopathy. Based on responses of improvement in mental state, results suggested that an intramuscular (IM) dose of thiamine ≥ 200 mg for 2 days is superior to smaller doses.3,30
Thomson and colleagues3 demonstrated that thiamine 500 mg should be infused 3 times per day for 2 to 3 days to prevent permanent damage to the the brain. If improvement is noticed, thiamine 250 mg intravenous (IV) or IM daily should be continued for another 5 days or until clinical improvement stops. Doses < 250 mg are insufficient in the prevention of irreversible brain damage. For prophylaxis in alcohol-dependent patients, thiamine 250 mg IM should be given daily for 3-5 days after hospital admission.3
Pharmacokinetics indicate that the half-life of thiamine is approximately 96 minutes or less. With a short half-life, multiple daily administrations may be necessary to replenish levels and allow for optimal blood-brain diffusion. However, previous studies24,31 did not evaluate whether the short half-life of thiamine reflected distribution in the predetermined and essential locations (ie, central nervous system). Thus, the optimal treatment regimen remains unknown but may be 2 to 3 times per day rather than the traditional once-daily dosing.32
Hypomagnesemia is common in alcohol-dependent patients and can contribute to the development of Wernicke’s encephalopathy, as thiamine needs magnesium as a cofactor.31 In the presence of hypomagnesemia, patients with suspected Wernicke’s encephalopathy may be unresponsive to parenteral thiamine. Thus, those who are at potential risk should have their magnesium level checked and corrected.33
METHODS
The study design was a retrospective electronic medical record review of patients aged 18-100 years admitted to an inpatient psychiatric facility between September 2013 and March 2014. The study took place at a nonprofit 32-bed acute care teaching hospital located in Lubbock, Texas. The hospital accepts patients with private insurance, Medicaid, or Medicare and indigents who are in crisis. The study was approved by the institutional review boards of Texas Tech University Health Sciences Center and Saint Joseph Health.
Data Collection and Procedure
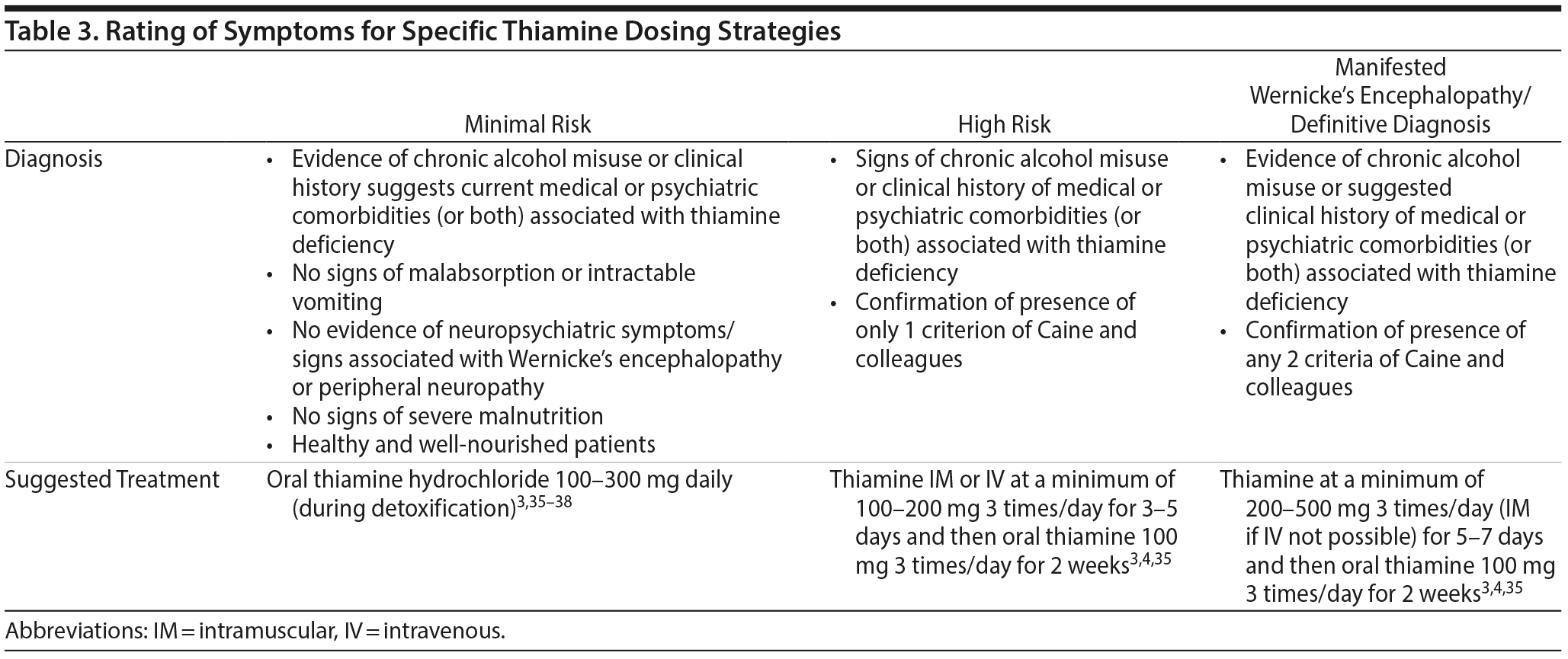
The criteria of Caine and colleagues23 were used in conjunction with medical and psychiatric risk factors4 to stratify patients into 2 distinct risk groups: alcohol dependent and non-alcohol dependent. We further classified each group into manifested Wernicke’s encephalopathy, high-risk, and minimal-risk subsets. Caine and colleagues’ criteria for Wernicke’s encephalopathy in alcohol dependence require at least 2 of the following 4 signs: dietary deficiencies, eye signs, cerebellar signs, and either an abnormal mental status or memory impairment. It is reasonable to apply the same diagnostic criteria to non-alcohol-dependent patients with medical and/or psychiatric disorders associated with thiamine deficiency.4 Patients who met 1 criterion in the presence of alcohol misuse and medical or psychiatric risk factors were considered high risk for Wernicke’s encephalopathy. Patients with no neuropsychiatric signs of Wernicke’s encephalopathy or specific signs of malnourishment but who had a clinical history indicating concurrent medical or psychiatric risk factors and the presence of nonspecific signs and symptoms of malnutrition were considered to be at minimal risk. The nonspecific signs and symptoms of malnutrition may include but are not limited to any of the following: reported loss of appetite, living alone or being homeless, insignificant weight loss, and nausea and vomiting.
The criteria of Caine and colleagues23 were validated retrospectively by autopsy studies2,4,14 and found to have 85% sensitivity and 100% specificity. Although these criteria allow the clinician to suspect Wernicke’s encephalopathy with a high degree of confidence, the full range of clinical presentation should be considered, including hypothermia, hypotension, coma, and neuropathy.4,23
Thiamine prescribing data (dosage amount, route, frequency schedule) were collected from the medical records. We also recorded if a patient received a banana bag consisting of 1 L of normal saline (sodium chloride 0.9%) with thiamine 100 mg, folic acid 1 mg, multivitamins for infusion, and magnesium sulfate 3 g IV infused over 4-8 hours in the emergency department. We analyzed whether the thiamine regimen correlated with the degree of risk for developing Wernicke’s encephalopathy. Blood magnesium levels and prescribing data were also collected.
RESULTS
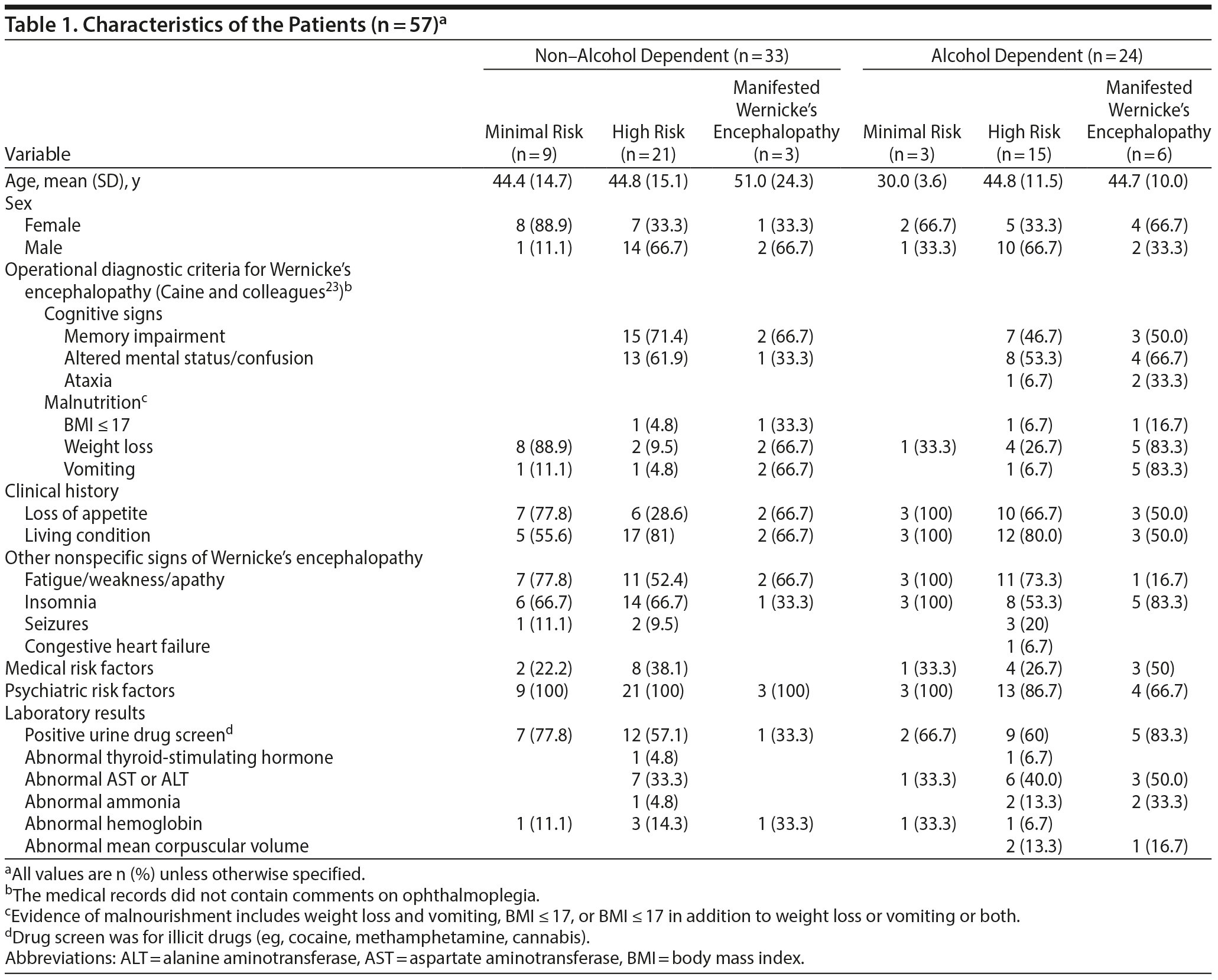
A total of 486 charts were reviewed. We excluded 429 patients (88%) who met none of the diagnostic criteria or who had a clinical history of malnourishment in the presence of medical or psychiatric risk factors for developing Wernicke’s encephalopathy. Fifty-seven subjects (12%) who met inclusion criteria were identified. Table 1 provides characteristics of the sample.
Of the alcohol-dependent group (42%, n = 24), subsets of patients were identified: 25% (n = 6) manifested Wernicke’s encephalopathy, 62.5% (n = 15) high risk, and 12.5% (n = 3) minimal risk. Of the non-alcohol-dependent group (58%, n = 33), subsets of patients were also identified: 9% (n = 3) manifested Wernicke’s encephalopathy, 64% (n = 21) high risk, and 27% (n = 9) minimal risk.
The estimated frequency rate of manifested Wernicke’s encephalopathy in the total patient sample was 1.85% (95% CI, 0.98-3.48): 1.23% (95% CI, 0.57-2.67) in the alcohol-dependent group and 0.62% (95% CI, 0.21-1.80) in the non-alcohol-dependent group. We also found the frequency rates of Wernicke’s encephalopathy to be 25% (95% CI, 12-44.89) and 9.1% (95% CI, 3.14-23.57) in the alcohol-dependent and non-alcohol-dependent categories, respectively.
Thirty-three percent (n = 7) of manifested Wernicke’s encephalopathy and high-risk subsets (n = 21) in the alcohol-dependent group and 17% (n = 4) of those subsets (n = 24) in the non-alcohol-dependent group (had 1 of the following: weight loss and vomiting, BMI ≤ 17, or BMI ≤ 17 in addition to either weight loss or vomiting or both. Sixty-seven percent (n = 16) of the alcohol-dependent group and 45% (n = 15) of the non-alcohol-dependent group reported loss of appetite, and 75% (n = 18) and 73% (n = 24), respectively, reported living alone or being homeless.
In addition to the signs of malnourishment, characteristic findings of Caine’s traits in the manifested Wernicke’s encephalopathy and high-risk subsets of the alcohol-dependent and non-alcohol-dependent groups (n = 45) were confusion (n = 26, 58%), memory impairment without confusion (n = 15, 33%), and ataxia (n = 2, 6%).
Medical disorders in the alcohol-dependent group included diabetes (n = 4, 12.5%), HIV (n = 2, 8%), cancer (n = 2, 8%), dementia (n = 2, 8%), epilepsy (n = 2, 6%), and BMI ≥ 40 (n = 1, 4%). Medical disorders in the non-alcohol-dependent group included diabetes (n = 4, 12%), BMI ≥ 40 (n = 3, 9%), cancer (n = 2, 6%), epilepsy (n = 2, 6%), and chronic renal failure (n = 1, 3%).
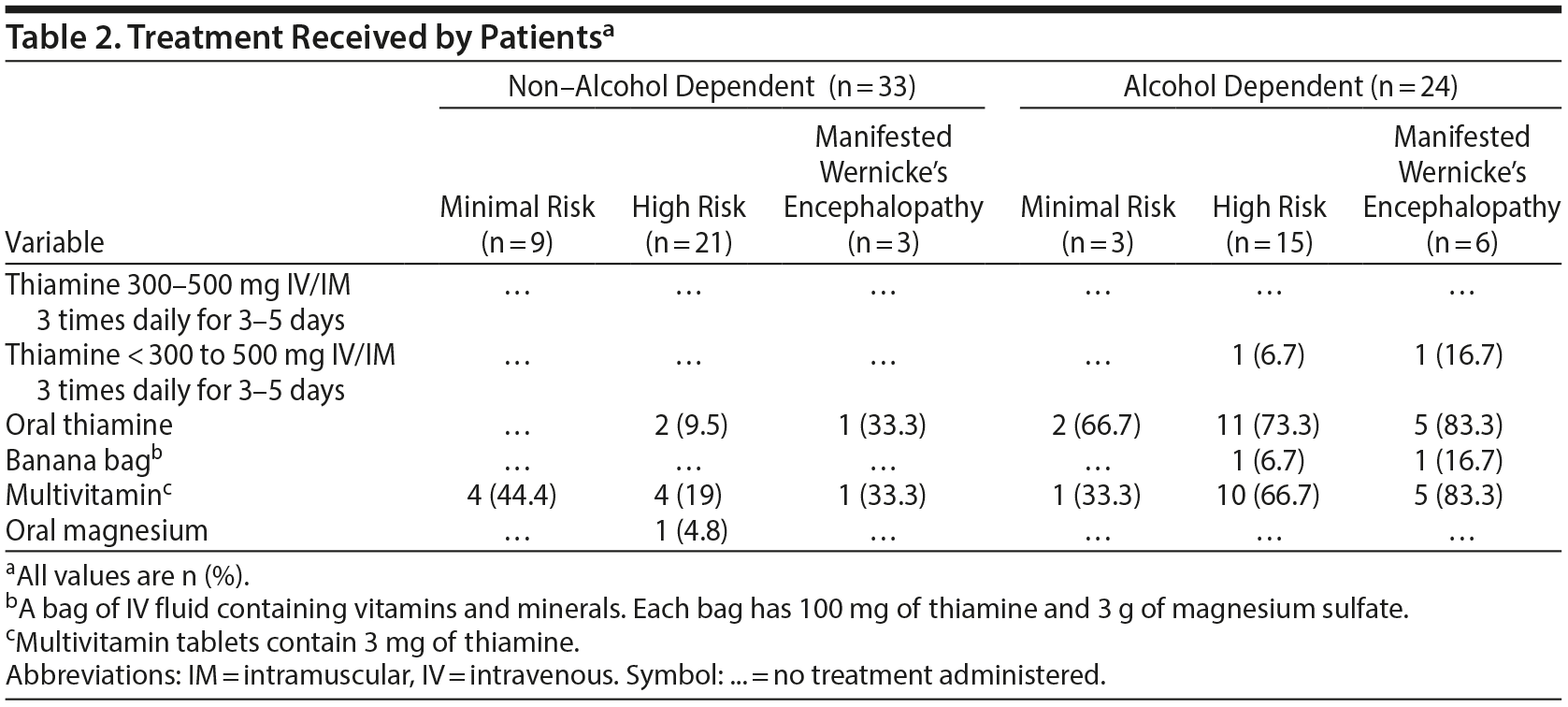
Concurrent psychiatric diagnoses in the alcohol-dependent patients included substance use disorders other than alcohol (n = 20, 87%), anxiety disorders (n = 10, 43%), depressive disorders (n = 9, 39%, bipolar disorders (n = 9, 39%), and psychotic disorders (n = 5, 22%). In the non-alcohol-dependent group, 52% (n = 17) had substance use disorders other than alcohol, 48% (n = 16) had depressive disorders, 45% (n = 15) had bipolar disorders, 33% (n = 11) had psychotic disorders, and 33% (n = 11) had anxiety disorders. Table 2 lists the treatments received by the patients.
DISCUSSION
Our study is the first in the United States to examine the frequency rate of Wernicke’s encephalopathy in psychiatric inpatients and report on the suboptimal management of the alcohol-dependent and non-alcohol-dependent groups at risk of developing the disorder. Our findings highlight the difficulties in recognizing, appropriately treating, and preventing Wernicke’s encephalopathy.
The rate of Wernicke’s encephalopathy was not statistically higher in alcohol-dependent compared to non-alcohol-dependent psychiatric patients (risk ratio = 2.75; 95% CI, 0.80-9.40; P = .146); however, this might be related to the low prevalence rate and small sample size. Our results seem to support the estimated prevalence of Wernicke’s encephalopathy calculated from previous retrospective autopsy studies.1-4,9-12
The findings of our retrospective chart review suggest increased risk of malnourishment and chronic medical illness associated with thiamine deficiency in the alcohol-dependent group. We also found the frequency rate of dementia is higher in the alcohol-dependent subsets, which could be a consequence of alcohol misuse and its improper management. Other medical conditions like HIV and cancer were found to be slightly higher in the alcohol-dependent patients. These findings possibly explain the higher rate of Wernicke’s encephalopathy in the alcohol-dependent subsets. We suggest that a detailed dietary history and risk factors for and signs of malnutrition should be acquired for patients with a mental health condition. The level of suspicion for Wernicke’s encephalopathy should be high in all clinical conditions that could cause thiamine deficiency.4 It could be helpful for psychiatric history and physical forms to contain a designated screening section for detailed dietary history and signs of malnutrition.
Substance use disorders were more prevalent among the alcoholic-dependent group, while psychotic disorders, bipolar disorders, and depressive disorders were more common in the non-alcohol-dependent group. These results may explain why clinicians often miss the diagnosis of Wernicke’s encephalopathy in psychiatric patients, as the illness can mimic or coexist with those mental disorders.
Altered mental status followed by impaired memory and ataxia were common in the high-risk and manifested Wernicke’s encephalopathy subsets. Therefore, Wernicke’s encephalopathy should be included in the differential diagnosis of all psychiatric patients presenting with altered mental status, memory impairment, or ataxia.3,12,26,31,34
Wernicke’s encephalopathy may also be missed in psychiatric patients because physical and neurologic examinations did not contain comments on ophthalmoplegia. Therefore, it is crucial to improve the diagnostic procedure by conducting comprehensive physical and neurologic examinations.
Since Caine and colleagues’ diagnostic criteria have a high sensitivity and specificity,23 we strongly recommend using those standards as the guideline for diagnosing Wernicke’s encephalopathy in both alcohol-dependent and non-alcohol-dependent patients.
After examining the different thiamine dosing strategies, we found that the majority of those with Wernicke’s encephalopathy or at high risk (76%, n = 21) in the alcohol-dependent group were prescribed traditional oral dosing of 100 mg oral daily. In the non-alcohol-dependent group, a small percentage (9%) received oral thiamine 100 mg daily. None of the groups received high dosages of parenteral thiamine; high dose refers to the minimum single dose ≥ 200 mg of thiamine IM or IV recommended by the European guidelines.4
Perhaps the most surprising finding was that many of the patients in the manifested Wernicke’s encephalopathy and high-risk subsets in both groups (57%, n = 45) did not receive thiamine at all. The emergency department records revealed a small percentage (10%, n = 21) of the manifested Wernicke’s encephalopathy and high-risk patients received an IV banana bag once, and the magnesium level was checked in only 1 patient. Early identification and management of high-risk patients at the time of admission to the emergency department is crucial to prevent and treat Wernicke’s encephalopathy.
A limitation of our study is that we could not assess the outcome for those patients with Wernicke’s encephalopathy or at high risk who did not receive thiamine or those who received traditional dosages of thiamine.3,4 It is difficult to gather information about patients once they have left the hospital. In future studies, it would be necessary to follow up once the patient has left the hospital.
In the emergency department, Wernicke’s encephalopathy is often underdiagnosed and undertreated, particularly in psychiatric patients.3 We believe that patients with Wernicke’s encephalopathy or at high risk should be admitted to general medical floors to receive high doses of IV thiamine and have their vital signs and mental status monitored.
There was no correlation between the severity of the risk for developing Wernicke’s encephalopathy and the thiamine dosing regimen, suggesting inadequacy of treatment. Furthermore, many European authors suggest that the traditional dosing of oral thiamine 100 mg daily is unreliable, particularly in high-risk patients,3,4,34 due to the short half-life and low bioavailability of thiamine.
CONCLUSION
Psychiatric patients who manifest signs of Wernicke’s encephalopathy should be admitted to general medical floors. Clinicians should obtain a detailed history including the pattern of alcohol use, other substance use, and dietary habits and screen for medical and psychiatric comorbidities associated with thiamine deficiency. Comprehensive physical and neurologic examinations should be performed, looking for signs of malnutrition and checking for the presence of hypothermia and hypotension. It is reasonable and recommended to use the criteria of Caine and colleagues to diagnose Wernicke’s encephalopathy in alcohol-dependent and non-alcohol-dependent patients.3,4,12,31,34,35
Parenteral thiamine should be administered to all high-risk subjects (Table 3). To decrease the risk of anaphylactic reaction, thiamine should be infused in 100-mL normal saline over 15-30 minutes,3 or an intradermal skin test should be administered before IV/IM thiamine infusion. Also, ensure that the serum magnesium level is adequate and always give thiamine IV or IM before any carbohydrate.4
This study reflects the misconceptions with regard to diagnosis and prevention of Wernicke’s encephalopathy in alcohol-dependent and non-alcohol-dependent patients. At the individual level, the results should help emergency department physicians, medical residents, and inpatient clinicians assess their daily practices with regard to early identification and treatment of suspected Wernicke’s encephalopathy. At the institutional level, this study should serve as a reminder to hospital administrators to review diagnostic procedures and thiamine prescribing practices.
Our findings are limited to one psychiatric inpatient facility and may not be generalizable to other patient populations, including other psychiatric hospitals and ambulatory patients in the United States. It would be useful to have a follow-up study to examine the percentage of mental health hospitals that have local guidelines and whether these guidelines support prescribing traditional doses of oral thiamine or high doses of parenteral thiamine.
Submitted: June 12, 2017; accepted September 13, 2017.
Published online: December 28, 2017.
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this article.
Financial disclosure: Drs Guirguis, Manning, Chavez, Nelson, Baronia, Bobba, Ahmed, and Ajufo have no personal affiliations or financial relationships with any commercial interest to disclose relative to the article.
Funding/support: None.
Acknowledgments: The authors thank Terry McMahon, MD; Miguel Roberto Villarreal, MD; and Chuck Giles, PhD, for reviewing the manuscript; Eneko Larumbe, PhD, for designing the study and providing statistical analysis; and Vivek Rachamallu, MD; Ram Aynampudi, MD; Joshua Chang, MD; and Rose Izuchi, MD, for data collection. All of the above individuals are affiliated with Texas Tech University Health Sciences Center, Lubbock, Texas, and have no conflicts of interest to declare.
REFERENCES
1. Thomson AD, Guerrini I, Marshall EJ. Wernicke encephalopathy: the role of thiamine, nutrition issues in gastroenterology, series #75. Pract Gastroenterol. 2009;23:21-30.
2. Harper CG, Giles M, Finlay-Jones R. Clinical signs in the Wernicke-Korsakoff complex: a retrospective analysis of 131 cases diagnosed at necropsy. J Neurol Neurosurg Psychiatry. 1986;49(4):341-345. PubMed doi:10.1136/jnnp.49.4.341
3. Thomson AD, Cook CC, Touquet R, et al; Royal College of Physicians, London. The Royal College of Physicians report on alcohol: guidelines for managing Wernicke’s encephalopathy in the accident and Emergency Department. Alcohol Alcohol. 2002;37(6):513-521. PubMed doi:10.1093/alcalc/37.6.513
4. Galvin R, Br×¥then G, Ivashynka A, et al; EFNS. EFNS guidelines for diagnosis, therapy and prevention of Wernicke encephalopathy. Eur J Neurol. 2010;17(12):1408-1418. PubMed doi:10.1111/j.1468-1331.2010.03153.x
5. Casanova MF. Wernicke’s disease and schizophrenia: a case report and review of the literature. Int J Psychiatry Med. 1996;26(3):319-328. PubMed doi:10.2190/GD76-1UP3-TR18-YPYH
6. Epstein RS. Wernicke’s encephalopathy following lithium-induced diarrhea. Am J Psychiatry. 1989;146(6):806-807. PubMed doi:10.1176/ajp.146.6.806a
7. National Institute of Mental Health website. https://www.nimh.nih.gov/health/statistics/prevalence/serious-mental-illness-smi-among-us-adults.shtml. Accessed June 1, 2017.
8. National Institute on Alcohol Abuse and Alcoholism website. https://www.niaaa.nih.gov/alcohol-health/overview-alcohol-consumption/alcohol-facts-and-statistics Accessed June 1, 2017.
9. Charness ME, Simon RP, Greenberg DA. Ethanol and the nervous system. N Engl J Med. 1989;321(7):442-454. PubMed doi:10.1056/NEJM198908173210706
10. Torvik A, Lindboe CF, Rogde S. Brain lesions in alcoholics. a neuropathological study with clinical correlations. J Neurol Sci. 1982;56(2-3):233-248. PubMed doi:10.1016/0022-510X(82)90145-9
11. Harper CG, Sheedy DL, Lara AI, et al. Prevalence of Wernicke-Korsakoff syndrome in Australia: has thiamine fortification made a difference? Med J Aust. 1998;168(11):542-545. PubMed
12. Sechi G, Serra A. Wernicke’s encephalopathy: new clinical settings and recent advances in diagnosis and management. Lancet Neurol. 2007;6(5):442-455. PubMed doi:10.1016/S1474-4422(07)70104-7
13. Boldorini R, Vago L, Lechi A, et al. Wernicke’s encephalopathy: occurrence and pathological aspects in a series of 400 AIDS patients. Acta Biomed Ateneo Parm. 1992;63:43-49. PubMed
14. Bleggi-Torres LF, de Medeiros BC, Werner B, et al. Neuropathological findings after bone marrow transplantation: an autopsy study of 180 cases. Bone Marrow Transplant. 2000;25(3):301-307. PubMed doi:10.1038/sj.bmt.1702140
15. Hoyumpa AM Jr. Mechanisms of thiamin deficiency in chronic alcoholism. Am J Clin Nutr. 1980;33(12):2750-2761. PubMed
16. Tanphaichitr V. Shils Thiamine ME, Olson JA, et al, eds. Modern Nutrition in Health and Disease. 9th ed. Baltimore, MD: Williams and Wilkins; 1999:381-389.
17. Thomson AD, Marshall EJ. The natural history and pathophysiology of Wernicke’s Encephalopathy and Korsakoff’s Psychosis. Alcohol Alcohol. 2006;41(2):151-158. PubMed doi:10.1093/alcalc/agh249
18. Thomson AD. Mechanisms of vitamin deficiency in chronic alcohol misusers and the development of the Wernicke-Korsakoff syndrome. Alcohol Alcohol suppl. 2000;35(1):2-7. PubMed
19. World Health Organization website. http://www.who.int/mental_health/mhgap/risks_to_mental_health_EN_27_08_12.pdf. Accessed June 1, 2017.
20. Isenberg-Grzeda E, Kutner HE, Nicolson SE. Wernicke-Korsakoff-syndrome: under-recognized and under-treated. Psychosomatics. 2012;53(6):507-516. PubMed doi:10.1016/j.psym.2012.04.008
21. Victor M. The Wernicke-Korsakoff syndrome. In Vinken PJ, Bruyn GW, eds. Handbook of Clinical Neurology. Vol 28, Part II. Amsterdam, The Netherlands: North-Holland Publishing; 1976:243-270.
22. Shils ME, Olson JA, Shike M, et al, eds. Modern Nutrition in Health and Disease. 9th ed. Baltimore, MD: Williams and Wilkins; 1999:381-389.
23. Caine D, Halliday GM, Kril JJ, et al. Operational criteria for the classification of chronic alcoholics: identification of Wernicke’s encephalopathy. J Neurol Neurosurg Psychiatry. 1997;62(1):51-60. PubMed doi:10.1136/jnnp.62.1.51
24. Donnino MW, Vega J, Miller J, et al. Myths and misconceptions of Wernicke’s encephalopathy: what every emergency physician should know. Ann Emerg Med. 2007;50(6):715-721. PubMed doi:10.1016/j.annemergmed.2007.02.007
25. Charness ME, Aminoff MJ, Wilterdink JL. Overview of the chronic neurologic complications of alcohol. http://www.uptodate.com/contents/overview-of-the-chronic-neurologic-complications-of-alcohol. Accessed October 31, 2017.
26. So YT, Aminoff MJ, Wilterdink JL. Wernicke encephalopathy. https://www.uptodate.com/contents/wernicke-encephalopathy. Accessed October 31, 2017
27. BMJ Best Practice. Wernicke’s encephalopathy. http://bestpractice.bmj.com/best-practice/monograph/405/treatment/evidence/references/1017.html. Accessed October 31, 2017
28. Jiang W, Gagliardi JP, Raj YP, et al. Acute psychotic disorder after gastric bypass surgery: differential diagnosis and treatment. Am J Psychiatry. 2006;163(1):15-19. PubMed doi:10.1176/appi.ajp.163.1.15
29. Worden RW, Allen HM. Wernicke’s encephalopathy after gastric bypass that masqueraded as acute psychosis: a case report. Curr Surg. 2006;63(2):114-116. PubMed doi:10.1016/j.cursur.2005.06.004
30. Ambrose ML, Bowden SC, Whelan G. Thiamin treatment and working memory function of alcohol-dependent people: preliminary findings. Alcohol Clin Exp Res. 2001;25(1):112-116. PubMed doi:10.1111/j.1530-0277.2001.tb02134.x
31. Cook CCH, Hallwood PM, Thomson AD. B Vitamin deficiency and neuropsychiatric syndromes in alcohol misuse. Alcohol Alcohol. 1998;33(4):317-336. PubMed doi:10.1093/oxfordjournals.alcalc.a008400
32. Bonucchi J, Hassan I, Policeni B, et al. Thyrotoxicosis associated Wernicke’s encephalopathy. J Gen Intern Med. 2008;23(1):106-109. PubMed doi:10.1007/s11606-007-0438-333.
33. Zieve L. Influence of magnesium deficiency on the utilization of thiamine. Ann N Y Acad Sci. 1969;162(2):732-743. PubMed doi:10.1111/j.1749-6632.1969.tb13005.x
34. Isenberg-Grzeda E, Chabon B, Nicolson SE. Prescribing thiamine to inpatients with alcohol use disorders: how well are we doing? J Addict Med. 2014;8(1):1-5. PubMed doi:10.1097/01.ADM.0000435320.72857.c8
35. Latt N, Dore G. Thiamine in the treatment of Wernicke encephalopathy in patients with alcohol use disorders. Intern Med J. 2014;44(9):911-915. PubMed doi:10.1111/imj.12522
36. Morgan MY, Ritson B. Alcohol and Health. Medical Students’ Handbook. London, England: Medical Council on Alcoholism; 1998.
37. Therapeutic Guidelines. Acute alcohol withdrawal. http://onlinelibrary.wiley.com/doi/10.1111/imj.12522/full. Accessed December 1, 2017
38. Lingford-Hughes AR, Welch S, Peters L, et al; British Association for Psychopharmacology, Expert Reviewers Group. BAP updated guidelines: evidence-based guidelines for the pharmacological management of substance abuse, harmful use, addiction and comorbidity: recommendations from BAP. J Psychopharmacol. 2012;26(7):899-952. PubMed doi:10.1177/0269881112444324