Objective: Undiagnosed and/or undertreated Wernicke’s encephalopathy can result in permanent brain damage, long-term institutionalization, and death. The purpose of this article is to heighten clinical awareness of Wernicke’s encephalopathy and shed light on its diagnosis and treatment, which are often inconsistent due to unclear diagnostic criteria and limited practice guidelines. An update on the management of Wernicke’s encephalopathy is presented and several case reports and a quality improvement project from our hospital are described.
Data Sources: PubMed, the Cochrane Database of Systematic Reviews, and PsycINFO were searched for English-language articles published between January 1991 and January 2014 using combinations of the following keywords: Wernicke’s encephalopathy, diagnosis, treatment/guideline(s), and thiamine.
Study Selection: The automated search identified over 500 articles. A manual review of the related citations and reference lists from articles of interest was also conducted. The articles reviewed were chosen on the basis of author consensus and because they represented expert opinion or the highest quality of evidence available.
Results: Diagnostic criteria are reviewed in this article and should be used to diagnose Wernicke’s encephalopathy with high sensitivity and specificity. The European Federation of Neurologic Societies and the Royal College of Physicians issued national guidelines for the diagnosis, prevention, and treatment of Wernicke’s encephalopathy. No benchmark national guidelines for treating Wernicke’s encephalopathy exist in the United States.
Conclusions: Whenever Wernicke’s encephalopathy is suspected, treatment should be initiated immediately with intravenous thiamine because oral thiamine is inadequate for preventing permanent brain damage. An adequate dose of intravenous thiamine administrated in a timely manner is a safe and life-saving treatment for Wernicke’s encephalopathy that could preserve brain cells and function.
CME Background
Articles are selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME articles on a variety of topics throughout each volume. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the article, correctly
answer at least 70% of the questions in
the Posttest, and complete the Evaluation.
The Posttest and Evaluation are available at
http://www.cmeinstitute.com/activities/Pages/journal.aspx.
CME Objective
After studying this article, you should be able to:
• Recognize signs and symptoms of Wernicke’s encephalopathy so that appropriate treatment can be provided to prevent permanent brain damage or death
Accreditation Statement
The CME Institute of Physicians
Postgraduate Press, Inc., is
accredited by the Accreditation
Council for Continuing Medical
Education to provide continuing
medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Credit™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Note: The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Credit™ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 1.0 hour of Category I credit for completing this program.
Date of Original Release/Review
This educational activity is eligible for AMA PRA Category 1 Credit™ through June 30, 2017. The latest review of this material was May 2015.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Larry Culpepper, MD, MPH, Editor in Chief, has been a member of the speakers/advisory boards for AstraZeneca, Forest, Jazz, Lundbeck, Merck, Shire, Sunovion, and Takeda; has been a stock shareholder of M3 My Mood Monitor; and has received royalties from UpToDate and other financial or material support from Oxford University Press. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears with the article.
Wernicke’s Encephalopathy: Increasing Clinician Awareness
of This Serious, Enigmatic,
Yet Treatable Disease
ABSTRACT
Objective: Undiagnosed and/or undertreated Wernicke’s encephalopathy can result in permanent brain damage, long-term institutionalization, and death. The purpose of this article is to heighten clinical awareness of Wernicke’s encephalopathy and shed light on its diagnosis and treatment, which are often inconsistent due to unclear diagnostic criteria and limited practice guidelines. An update on the management of Wernicke’s encephalopathy is presented and several case reports and a quality improvement project from our hospital are described.
Data Sources: PubMed, the Cochrane Database of Systematic Reviews, and PsycINFO were searched for English-language articles published between January 1991 and January 2014 using combinations of the following keywords: Wernicke’s encephalopathy, diagnosis, treatment/guideline(s), and thiamine.
Study Selection: The automated search identified over 500 articles. A manual review of the related citations and reference lists from articles of interest was also conducted. The articles reviewed were chosen on the basis of author consensus and because they represented expert opinion or the highest quality of evidence available.
Results: Diagnostic criteria are reviewed in this article and should be used to diagnose Wernicke’s encephalopathy with high sensitivity and specificity. The European Federation of Neurologic Societies and the Royal College of Physicians issued national guidelines for the diagnosis, prevention, and treatment of Wernicke’s encephalopathy. No benchmark national guidelines for treating Wernicke’s encephalopathy exist in the United States.
Conclusions: Whenever Wernicke’s encephalopathy is suspected, treatment should be initiated immediately with intravenous thiamine because oral thiamine is inadequate for preventing permanent brain damage. An adequate dose of intravenous thiamine administrated in a timely manner is a safe and life-saving treatment for Wernicke’s encephalopathy that could preserve brain cells and function.
Prim Care Companion CNS Disord
2015;17(3):doi:10.4088/PCC.14r01738
© Copyright 2015 Physicians Postgraduate Press, Inc.
Submitted: October 15, 2014; accepted January 14, 2015.
Published online: May 21, 2015.
Corresponding author: Matthew Macaluso, DO, Department of Psychiatry and Behavioral Sciences, University of Kansas School of Medicine–Wichita, 1010 North Kansas, Wichita, KS 67214-3199 ([email protected]).
Wernicke’s encephalopathy is difficult to diagnose and often goes untreated or undertreated in clinical practice. This lack of detection and treatment is evidenced by the fact that Wernicke’s encephalopathy is first diagnosed postmortem in over 80% of cases.1,2 Even in those cases in which Wernicke’s encephalopathy is diagnosed clinically, there are multiple, often conflicting, guidelines suggesting how much thiamine should be given and via which route of administration. This review serves as a summary of the diagnostic and treatment considerations for Wernicke’s encephalopathy and will guide clinicians in understanding the evidence base in order to make informed treatment decisions.
METHOD
A literature review was conducted using PubMed, the Cochrane Database of Systematic Reviews, and PsycINFO electronic databases. We searched for English-language articles published between January 1991 and January 2014 using combinations of the following keywords: Wernicke’s encephalopathy, diagnosis, treatment/guideline(s), and thiamine. The automated search identified over 500 articles. We also conducted a manual review of the related citations and reference lists from articles of interest. The articles reviewed were chosen on the basis of author consensus and because they represent expert opinion or the highest quality of evidence available.
RESULTS
Definition and Etiology
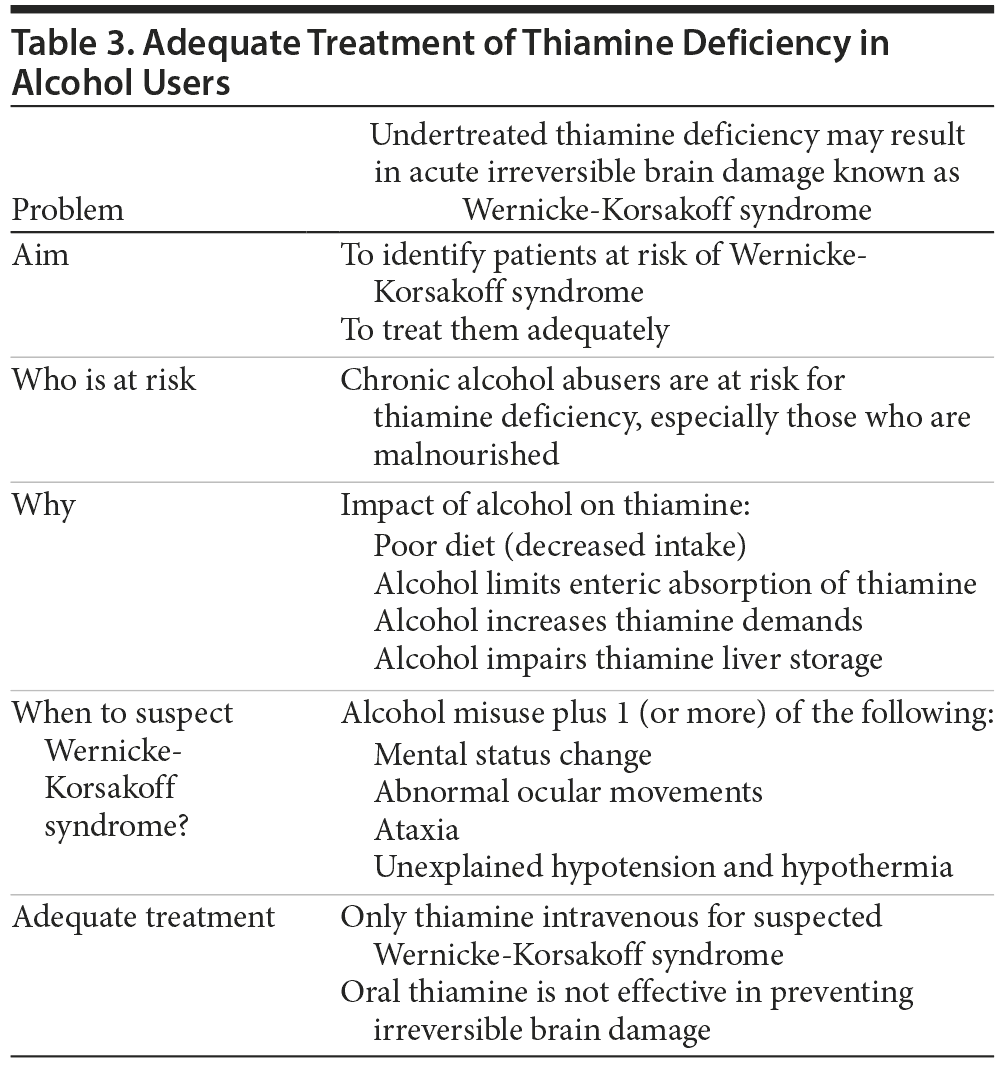
Wernicke’s encephalopathy is a neuropsychiatric emergency with high morbidity (84%) and mortality (up to 20%).3 The Wernicke’s encephalopathy syndrome of acute mental status change with associated ophthalmoplegia and ataxia was first reported in 1881 by the German neuropsychiatrist Carl Wernicke.3,4 In 1887, the Russian neuropsychiatrist Sergei Korsakoff described a syndrome of severe and persistent working memory impairment known as Korsakoff psychosis.2–4
Decades after Wernicke’s initial report, thiamine (vitamin B1) deficiency was discovered to be responsible for both Wernicke’s encephalopathy and Korsakoff psychosis: Wernicke’s encephalopathy is associated with acute thiamine deficiency, while Korsakoff psychosis is associated with chronic thiamine deficiency.1,4–7 Intuitively, urgent treatment with intravenous thiamine may reverse the early symptoms of Wernicke’s encephalopathy. This fact is evidenced by the fact that 84% of individuals who survive an acute episode of Wernicke’s encephalopathy without adequate treatment will go on to develop Korsakoff psychosis.3–5,7 These data led to the name Wernicke-Korsakoff syndrome.2,6
A number of mechanisms can lead to acute thiamine deficiency, although alcohol abuse disorders remain the most common (up to 90%).1,6,7 The estimated prevalence of Wernicke-Korsakoff syndrome is up to 2.8% of the general population and approximately 12.5% of individuals who chronically abuse alcohol.1,4–6,8
Other causes of thiamine deficiency include gastrointestinal disorders and surgeries (ie, gastric bypass), acquired immune deficiency syndrome, hemodialysis, malignancies, and other systemic diseases.1,5,6 Thiamine deficiency can also result from infections, shock, prolonged nutritional deficiencies, or the administration of intravenous glucose prior to thiamine in malnourished or alcohol-dependent individuals.3,4,8,9
Pathophysiology
Thiamine is an essential cofactor for enzymes in the pentose phosphate and Krebs cycles, and the brain heavily relies on glucose metabolism and thiamine in order to function optimally.1,9 Clinical signs of thiamine deficiency present when brain thiamine levels drop to 20% below baseline.6 Thiamine deficiency lasting for over 4 days results in neurotoxic edema and a biochemical cascade of insufficient metabolism and oxidative stress. This process occurs on the subcellular level and, if not replenished in a timely fashion, can progress to neuronal necrosis and dysfunction of the blood-brain barrier.5,6,10
Certain brain structures are more vulnerable to damage resulting from thiamine deficiency including the thalamus, mamillary bodies, periaqueductal and paraventricular areas, locus ceruleus, cranial nerves nuclei, and reticular formation. In fact, the clinical presentation of Wernicke’s encephalopathy correlates with the areas of the brain that are affected, leading to a highly variable and nonspecific presentation including the following1,3,5,11:
- Mental status change including stupor and memory impairment correlate with the dorsomedial thalamus, mamillary bodies, and even cortical lesions (poor prognosis11).
- Wernicke-Korsakoff syndrome results from combined thalamo-hypothalamic lesions.
- Ocular signs are associated with lesions of the cranial nerves III and IV, the locus ceruleus, and periaqueductal gray area.
- Ataxia correlates with lesions in the superior vermis and cerebellar cortex.
- Hypothalamic involvement results in abnormal regulation of body temperature.
Diagnostic Approaches
The initial diagnostic criteria for Wernicke’s encephalopathy described by Wernicke in 1881 required the presence of all 3 of the following symptoms: ophthalmoplegia, ataxia, and mental status change. This full, classic triad is present in only about 10%–16% of cases of Wernicke’s encephalopathy, which may explain the large percentage of cases that are first diagnosed postmortem (80%–90%).1,7,9 Retrospective studies have shown that mental status change is the most prevalent presenting symptom (82%), followed by ocular signs (29%) and a variety of gait disturbances (23%).2,5,7 Mental status change can manifest in a number of ways including memory impairment, agitated delirium, or coma.3,6 Ophthalmoplegia may present as nystagmus, papilledema, or gaze palsies.2,12
In order to diagnose Wernicke’s encephalopathy with high specificity and sensitivity (85%–100%), Caine et al13 proposed new diagnostic criteria that included dietary deficiencies in addition to the classic triad (Table 1). These criteria were adopted by the European Federation of Neurologic Societies (EFNS).8 The Royal College of Physicians (RCP) recommends presumptively diagnosing Wernicke’s encephalopathy in patients who present with a history of alcohol abuse and any 1 of the following unexplained symptoms: mental status change, memory or gait problems, ophthalmoplegia, hypothermia, or hypotension (Table 1).10

At present, there are no biomarkers that can be used to confirm the diagnosis or quantify its severity. It is established that plasma thiamine levels do not correlate with the diagnosis of Wernicke’s encephalopathy.1,4,6,10 Conventional magnetic resonance imaging has 53% sensitivity and 93% specificity and, thus, may be of use in supporting a clinical diagnosis of Wernicke’s encephalopathy, but cannot be used to exclude it.4,6,8 Magnetic resonance imaging findings are highly variable, and 58% of cases show lesions in the mamillary bodies, thalamus, and periaqueductal areas, although other brain regions may be implicated as well.5,11
Pharmacokinetics, Pharmacodynamics,
and Safety of Thiamine
Orally administered thiamine is absorbed in the small intestine by a rate-limited, saturable process of active transport that reaches a plateau of 4.5 mg after an oral dose of 30 mg when administered in healthy volunteers.1,6 In individuals who abuse alcohol, thiamine’s gastrointestinal absorption may be decreased by up to 70%. Moreover, orally administered thiamine in alcohol-dependent individuals does not increase thiamine concentration in the cerebrospinal fluid and may not be sufficient in most cases of acute thiamine deficiency. Parenteral thiamine is both actively and passively transported across the blood-brain barrier.6 As a result, acute thiamine deficiency associated with Wernicke’s encephalopathy may be quickly corrected with high doses of parenteral thiamine. Because parenteral thiamine has a short half-life (about 96 minutes), multiple doses may be required in order to achieve the maximal concentration.1,4,9
Thiamine has a favorable safety profile.4,7,9 There are few side effects, and administration is rarely associated with anaphylaxis (1:5,000,000 administrations).6,10 Multiple sources recommend administering intravenous thiamine slowly (over 10–30 minutes) and diluted in 50–100 mL of normal saline to further minimize risk of anaphylaxis.7,10,12
Treatment of Wernicke’s Encephalopathy:
Conflicting Approaches
On the basis of our review of the literature and practice guidelines, the timely administration of adequately dosed parenteral thiamine is the most effective treatment for Wernicke’s encephalopathy.4,12 This approach was initially suggested by Victor and colleagues3 in the 1950s and involved giving parenteral thiamine at a dose of 100 mg daily.4 The use of oral thiamine in managing acute Wernicke’s encephalopathy has been criticized in the literature as inadequate, and case reports exist of new-onset Wernicke’s encephalopathy in individuals despite administration of high-dose oral thiamine. In part, oral thiamine may provide inadequate treatment for Wernicke’s encephalopathy due to poor absorption in alcohol-dependent or malnourished patients.1,7–9
The exact dose of parenteral thiamine required to adequately treat Wernicke’s encephalopathy is unknown.4–7,14 Limited data exist in this area because conducting randomized, controlled trials would be difficult or even unethical given the known risk of permanent impairment that can result from improper treatment.
A Cochrane review in 201314 found only 1 controlled clinical trial comparing the efficacy of different thiamine dosages in treating alcohol-dependent individuals. This review concluded that current data on the treatment of Wernicke’s encephalopathy remain inconclusive.14 Ambrose and colleagues15 found that parenteral thiamine doses of 200 mg/d better restored neurologic functioning when compared with daily dosages of 100 mg or less. The remainder of the existing data are based solely on case reports. Thomson and Marshall16 debate on the efficiency of intramuscular versus intravenous thiamine administration. They conclude that only intravenous thiamine should be used to adequately treat Wernicke’s encephalopathy.16 Many authors criticize thiamine doses of less than 200 mg/d as insufficient and recommend using 500-mg doses of thiamine dosed 3 times per day for adequate treatment of Wernicke’s encephalopathy.4–7,10,12,16 Others suggest dosing intravenous thiamine as high as 1 g per dose in patients not adequately responding to lower thiamine doses in order to achieve a high blood to brain ratio of thiamine and to allow for passive thiamine diffusion to occur.9,15,17
No guidelines that specify thiamine dosage or route of administration when treating individuals with Wernicke’s encephalopathy have been endorsed by national organizations in the United States, and most treatment approaches are empiric.6,7,14,18 The thiamine hydrochloride package insert19 recommends administering an initial 100-mg dosage of thiamine intravenously followed by intramuscular doses of 50–100 mg of thiamine daily until the patient is able to return to a normal diet (Table 2). The RCP (United Kingdom) and EFNS issued national guidelines for the treatment of Wernicke’s encephalopathy.8,10 Both protocols suggest the same frequency (3 times daily) and route of administration (intravenous) with recommended thiamine doses of 500 mg (RCP) and 200 mg (EFNS) (Table 2). The UK National Institute for Health and Clinical Excellence notes that British treatment guidelines for Wernicke’s encephalopathy (RCP and British National Formulary) are established by expert consensus.16,20 Michael E. Charness, MD,12 the National Institutes of Health expert on alcohol abuse and alcoholism, recommends that clinicians use thiamine doses of 500 mg with a route and frequency of administration comparable to RCP’s protocol.

Another issue is the selective management of patients at “high risk” for Wernicke’s encephalopathy, which include those alcohol-dependent individuals who are malnourished, have severe liver damage, present in acute alcohol withdrawal, or are hospitalized for any acute trauma, head injuries, or acute illness.10,20 Both RCP and EFNS recommend that at-risk patients be treated with high doses of parenteral thiamine (Table 2).8,10
CLINICAL VIGNETTES
Our consultation liaison psychiatry practice encountered low awareness of the diagnostic criteria and treatment of Wernicke’s encephalopathy among hospitalist physicians. We selected cases most likely to be encountered by hospitalists on a daily basis, with the goal of highlighting specific key points with each case.
Case 1: Oral Versus Intravenous Thiamine
Mr A, a 67-year-old man with past medical history including alcohol dependence and gastrointestinal bleeding, presented with suicidal ideation, confusion, and ataxia. He was dysarthric and endorsed visual hallucinations and paranoid delusions at presentation to the hospital. He had an elevated blood pressure level and reduced body mass index (BMI). Laboratory workup was generally unremarkable and showed normal liver function tests and a nondetectable blood alcohol concentration. Thiamine was initiated at a dosage of 100 mg daily, administered intravenously. However, the hospital pharmacy automatically substituted oral thiamine in place of the intravenous thiamine because Mr A was taking all other medications by mouth.
Mr A’s mental status and ataxia did not improve with orally administered thiamine. After the thiamine route of delivery was changed from oral to intravenous administration, his clinical status improved rapidly, including reduction of cerebellar signs and improved mental status. Intravenous thiamine was continued until clinical improvement plateaued, at which point Mr A was switched to an oral thiamine preparation. He had a nearly full recovery to his baseline level of mental status, although was noted to confabulate at the time of discharge, suggesting the possibility of mild to moderate Korsakoff psychosis. Key points are as follows:
- It can be difficult to differentiate Wernicke’s encephalopathy from alcohol intoxication or withdrawal. However, the potentially serious consequences of undiagnosed and untreated Wernicke’s encephalopathy should lower the threshold of clinical suspicion.2,4
- Wernicke’s encephalopathy should always be treated with intravenous, rather than oral thiamine. Oral thiamine is not adequately absorbed in alcohol-dependent or malnourished patients and is never adequate for the treatment of Wernicke’s encephalopathy.1,7–9
- Ophthalmoplegia is generally the first symptom to resolve and often reverses within hours to days of starting treatment. Improvement of gait disturbance and mental status change may take 1 or more weeks to resolve.3
Case 2: Wernicke’s Encephalopathy
Presenting as Postoperative Delirium
Ms B, a 57-year-old woman, was transferred to our hospital from another facility due to ongoing confusion, agitation, and irritability after undergoing spinal surgery 7–10 days prior. Her past medical history included chronic-obstructive pulmonary disease, hypertension, degenerative disc disease, hepatitis C, and alcohol dependence. At arrival, Ms B was lethargic and disoriented to time, place, and situation. Her gait and mobility were impaired due to a feeling of “dizziness,” which was presumably related to the spinal surgery. Benzodiazepines were ordered on an as-needed basis for alcohol withdrawal per Clinical Institute Withdrawal Assessment for Alcohol protocol.21 Nonetheless, confusion and periods of agitation persisted. Ms B had been functioning well before spinal surgery and was living independently without assistance. No known past psychiatric history was elicited; however, she had a history of alcohol dependence and last consumed alcohol the night before the surgery. Ms B’s blood glucose levels and metabolic panel were unremarkable, and it appeared that she had not received any thiamine at the hospital that conducted her spinal surgery.
Because of Ms B’s history of alcohol dependence, mental status change, and ataxia, she met the diagnostic criteria of Caine and colleagues13 for Wernicke’s encephalopathy. We initiated intravenous thiamine at a dosage of 500 mg every 8 hours. On the second day after starting thiamine, Ms B’s dizziness and mental status improved, and she eventually regained full orientation with resolution of her irritability and disorganized thought processes. Key points are as follows:
- Ms B was at high risk for Wernicke’s encephalopathy because of her history of alcohol dependence and because she recently stopped using alcohol for planned surgery (alcohol withdrawal increases the brain’s thiamine demands).
- Intravenous thiamine is the treatment of choice for patients with multiple risk factors for developing Wernicke’s encephalopathy including malnourishment, acute alcohol withdrawal, trauma, or recent head injury.1,7–10,22
- It would be beneficial to complete a thorough screening for the risk of complicated alcohol withdrawal prior to any surgical procedure, including outpatient surgery.
- Wernicke’s encephalopathy may present as postoperative delirium and should be considered if the cause of delirium is unclear.23,24
- Delirium or coma may be the only signs of Wernicke’s encephalopathy.23,25 Mental status change is the most prevalent symptom of Wernicke’s encephalopathy (82%),2,4,12 and 30.6% of patients diagnosed postmortem presented clinically with coma alone.25
- Hospitalists should have a high index of suspicion for Wernicke’s encephalopathy, especially if delirium or coma of unknown etiology coincides with dietary deficiency, alcohol abuse, or decreased body mass index.2 Intravenous thiamine should be started immediately in such cases.1,4 In severe cases, higher doses of thiamine may be needed to quickly restore brain thiamine content. Many authors suggest that patients with severe Wernicke’s encephalopathy should receive dosages of 500 mg of intravenous thiamine 3 times per day.10,12,16
HOSPITAL QUALITY IMPROVEMENT PROJECT: ADEQUATE TREATMENT OF
THIAMINE DEFICIENCY IN ALCOHOL USERS
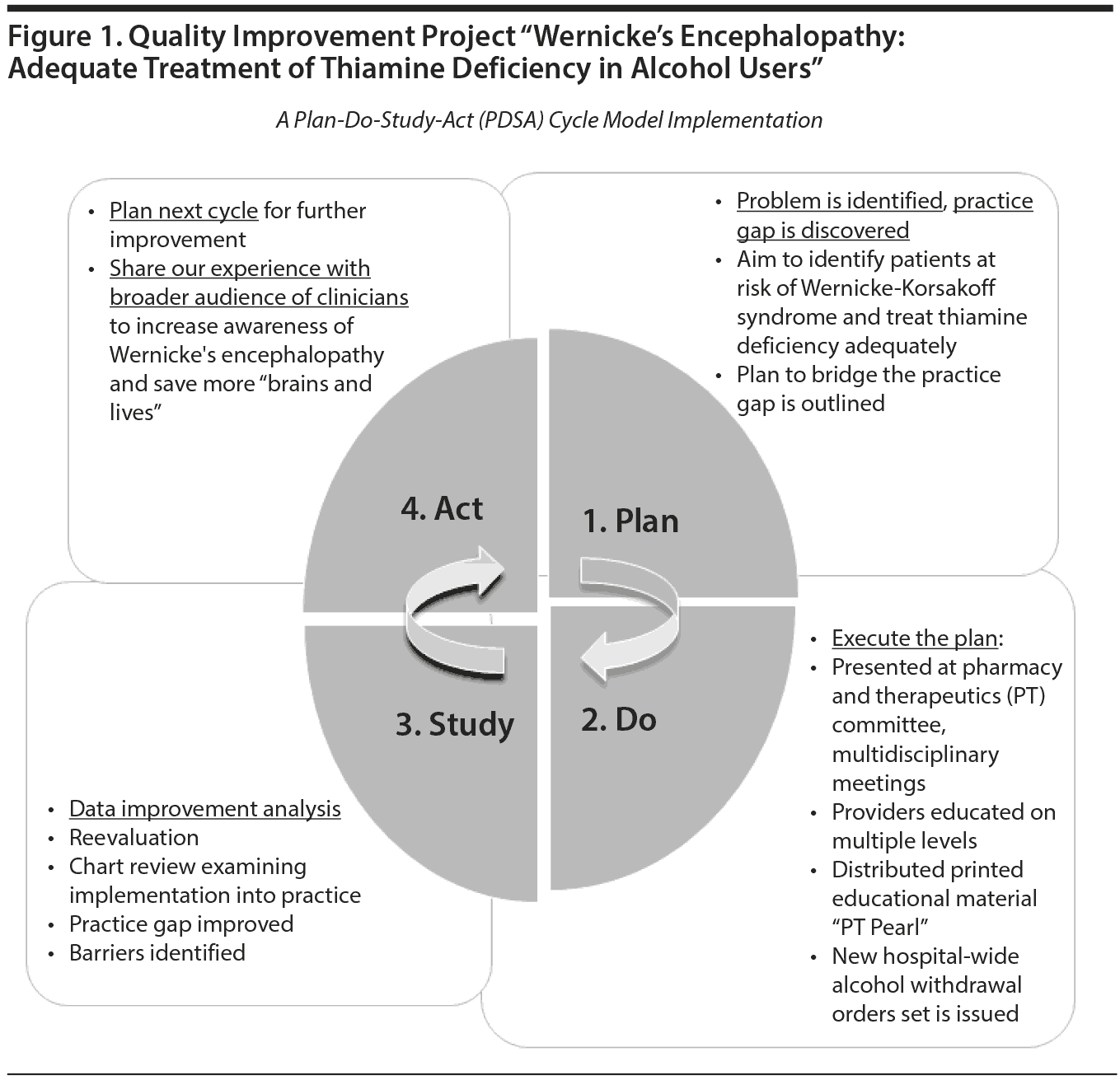
We completed a quality improvement project intended to update prescribers and pharmacists regarding the diagnosis and treatment of Wernicke’s encephalopathy. The goal of describing this quality improvement project as part of this article is to share our hospital’s experience and add to the dialogue regarding best practices for managing Wernicke’s encephalopathy.
Our consultation liaison psychiatry group encountered gaps in local practice in the form of low clinician awareness of Wernicke’s encephalopathy and lack of knowledge of diagnostic criteria and appropriate treatment. We used a plan-do-study-act (PDSA) cycle model26 to implement a quality improvement project titled “Wernicke’s Encephalopathy: Adequate Treatment of Thiamine Deficiency in Alcohol Users” (Figure 1). Our team developed the “plan,” which aimed to bridge the knowledge gap and improve health care quality. The “do” part of this project involved presenting the problem and reviewing the literature and clinical cases at our interdepartmental Morbidity and Mortality Conference. We also presented the same to our hospital’s pharmacy and therapeutics committee and distributed printed educational material to clinicians (Table 3). This work was incorporated into a new standing order set within our hospital system for alcohol withdrawal that now includes intravenous thiamine as an option. The “study” part of the PDSA cycle involved reevaluating the success of these interventions with a multidisciplinary chart review. We concluded that the proposed change was successfully adopted within our hospital system with resultant narrowing of the practice gap.
CONCLUSION
Wernicke’s encephalopathy is not a rare disease and can occur in any malnourished patient, not solely in those who are alcohol dependent. Inadequate treatment of patients at risk for Wernicke’s encephalopathy can cause permanent brain damage, long-term institutionalization, and death; all of which could be prevented if appropriate treatment is implemented in a timely fashion. The serious consequences of untreated Wernicke’s encephalopathy warrant a high index of suspicion and low threshold for treatment. If Wernicke’s encephalopathy is suspected, it is critical to initiate immediate treatment with intravenous thiamine administered at least 3 times daily. Thiamine by mouth is inadequate and ineffective for preventing permanent brain damage. Timely administration of adequately dosed parenteral thiamine is a “brain-saving,” safe, and inexpensive treatment for Wernicke’s encephalopathy. Clinicians should consider the diagnostic criteria that were reviewed in this article and treatment with intravenous thiamine in high-risk populations.
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration–approved labeling has been presented in this article.
Author affiliations: Department of Psychiatry and Behavioral Sciences, University of Kansas School of Medicine–Wichita (Drs Flynn, Macaluso, and D’Empaire) and Via Christi Hospitals (Dr Troutman), Wichita, Kansas.
Financial disclosure: Dr Macaluso has conducted clinical trials research as a principal investigator over the last 12 months for AbbVie, Alkermes, AssureRx, Eisai, Forum, and Naurex. All clinical trial and study contracts were with and payments made to the Kansas University Medical Center Research Institute, a research institute affiliated with Kansas University School of Medicine–Wichita. Drs Flynn, D’Empaire, and Troutman have no personal affiliations or financial relationships with any commercial interest to disclose relative to the article.
Funding/support: None reported.
REFERENCES
1. Thomson AD. Mechanisms of vitamin deficiency in chronic alcohol misusers and the development of the Wernicke-Korsakoff syndrome. Alcohol Alcohol suppl. 2000;35(1):2–7. PubMed
2. Harper CG, Giles M, Finlay-Jones R. Clinical signs in the Wernicke-Korsakoff complex: a retrospective analysis of 131 cases diagnosed at necropsy. J Neurol Neurosurg Psychiatry. 1986;49(4):341–345. doi:10.1136/jnnp.49.4.341 PubMed
3. Victor M, Adams R, Collins G. The Wernicke-Korsakoff Syndrome and Related Neurologic Disorders Due to Alcoholism and Malnutrition. Philadelphia, PA: F.A. Davis; 1989.
4. Donnino MW, Vega J, Miller J, et al. Myths and misconceptions of Wernicke’s encephalopathy: what every emergency physician should know. Ann Emerg Med. 2007;50(6):715–721. doi:10.1016/j.annemergmed.2007.02.007 PubMed
5. Sechi G, Serra A. Wernicke’s encephalopathy: new clinical settings and recent advances in diagnosis and management. Lancet Neurol. 2007;6(5):442–455. doi:10.1016/S1474-4422(07)70104-7 PubMed
6. Isenberg-Grzeda E, Kutner HE, Nicolson SE. Wernicke-Korsakoff syndrome: underrecognized and undertreated. Psychosomatics. 2012;53(6):507–516. doi:10.1016/j.psym.2012.04.008 PubMed
7. Cook CC. Prevention and treatment of Wernicke-Korsakoff syndrome. Alcohol Alcohol suppl. 2000;35(1):19–20. PubMed
8. Galvin R, Bråthen G, Ivashynka A, et al; EFNS. EFNS guidelines for diagnosis, therapy and prevention of Wernicke encephalopathy. Eur J Neurol. 2010;17(12):1408–1418. doi:10.1111/j.1468-1331.2010.03153.x PubMed
9. Cook CC, Hallwood PM, Thomson AD. B Vitamin deficiency and neuropsychiatric syndromes in alcohol misuse. Alcohol Alcohol. 1998;33(4):317–336. doi:10.1093/oxfordjournals.alcalc.a008400 PubMed
10. Thomson AD, Cook CCH, Touquet R, et al; Royal College of Physicians, London. The Royal College of Physicians Report on Alcohol: guidelines for managing Wernicke’s encephalopathy in the accident and emergency department. Alcohol Alcohol. 2002;37(6):513–521. doi:10.1093/alcalc/37.6.513 PubMed
11. Zuccoli G, Pipitone N. Neuroimaging findings in acute Wernicke’s encephalopathy: review of the literature. AJR Am J Roentgenol. 2009;192(2):501–508. doi:10.2214/AJR.07.3959 PubMed
12. Charness ME. Alcohol. In: Noseworthy J, ed. Neurological Therapeutics Principles and Practice. London, UK: Martin Dunitz; 2006:1677–1685.
13. Caine D, Halliday GM, Kril JJ, et al. Operational criteria for the classification of chronic alcoholics: identification of Wernicke’s encephalopathy. J Neurol Neurosurg Psychiatry. 1997;62(1):51–60. doi:10.1136/jnnp.62.1.51 PubMed
14. Day E, Bentham PW, Callaghan R, et al. Thiamine for prevention and treatment of Wernicke-Korsakoff syndrome in people who abuse alcohol. Cochrane Database Syst Rev. 2013;7(7) PubMed
15. Ambrose ML, Bowden SC, Whelan G. Thiamin treatment and working memory function of alcohol-dependent people: preliminary findings. Alcohol Clin Exp Res. 2001;25(1):112–116. doi:10.1111/j.1530-0277.2001.tb02134.x PubMed
16. Thomson AD, Marshall EJ. BNF recommendations for the treatment of Wernicke’s encephalopathy: lost in translation? Alcohol Alcohol. 2013;48(4):514–515. doi:10.1093/alcalc/agt034 PubMed
17. Nakada T, Knight RT. Alcohol and the central nervous system. Med Clin North Am. 1984;68(1):121–131. PubMed
18. US Department of Health & Human Services. Agency for Healthcare Research and Quality. National Guideline Clearinghouse. Wernicke’s encephalopathy. http://www.guideline.gov/search/search.aspx?term=wernicke%27s+encephalopathy. Accessed October 14, 2014.
19. Thiamine (thiamine hydrochloride) injection, solution. [package insert]. Schaumburg, IL: APP Pharmaceuticals, LLC; 2012.
20. National Institute for Health and Clinical Excellence. Alcohol-Use
Disorders: Diagnosis, Assessment and Management of Harmful Drinking
and Alcohol Dependence Clinical Guideline 115. London, UK: NICE; 2011.
21. Sullivan JT, Sykora K, Schneiderman J, et al. Assessment of alcohol withdrawal: the revised Clinical Institute Withdrawal Assessment for Alcohol scale (CIWA-Ar). Br J Addict. 1989;84(11):1353–1357. doi:10.1111/j.1360-0443.1989.tb00737.x PubMed
22. Blackmore C, Ouellet JF, Niven D, et al. Prevention of delirium in trauma patients: are we giving thiamine prophylaxis a fair chance? Can J Surg. 2014;57(2):78–81. doi:10.1503/cjs.006413 PubMed
23. Fricchione GL, Nejad SH, Esses JA, et al. Postoperative delirium. Am J Psychiatry. 2008;165(7):803–812. doi:10.1176/appi.ajp.2008.08020181 PubMed
24. Onishi H, Sugimasa Y, Kawanishi C, et al. Wernicke encephalopathy presented in the form of postoperative delirium in a patient with hepatocellular carcinoma and liver cirrhosis: a case report and review of the literature. Palliat Support Care. 2005;3(4):337–340. doi:10.1017/S1478951505050510 PubMed
25. Lana-Peixoto MA, Dos Santos EC, Pittella JE. Coma and death in unrecognized Wernicke’s encephalopathy: an autopsy study. Arq Neuropsiquiatr. 1992;50(3):329–333. doi:10.1590/S0004-282X1992000300012 PubMed
26. Taylor MJ, McNicholas C, Nicolay C, et al. Systematic review of the application of the plan-do-study-act method to improve quality in health care. BMJ Qual Saf. 2014;23(4):290–298. doi:10.1136/bmjqs-2013-001862 PubMed
Enjoy this premium PDF as part of your membership benefits!




