Personality disorders encompass a range of enduring patterns of behavior, cognition, and inner experience that deviate from cultural norms. The Journal of Clinical Psychiatry delves into the complexities of these disorders, from borderline and antisocial to avoidant and obsessive-compulsive personality disorders. Our expert editors curate content that offers a comprehensive understanding of the etiology, diagnosis, and treatment options. As these disorders often intersect with other mental health conditions, our journal emphasizes a holistic approach, ensuring clinicians are equipped with the knowledge to provide optimal care.
Personality Disorders
Recently published articles about Personality Disorders
Case Report
Risperidone Long-Acting Injection Reduced Emergency Visits in a Patient With Comorbid Autism Spectrum and Borderline Personality Disorders
May 1, 2025
This case demonstrates that LAI risperidone may support emotional and interpersonal improvements in a patient with both borderline personality disorder and autism spectrum disorder.
Original Research
Bridge Symptoms Between Borderline Personality and PTSD
September 9, 2024
Self-aggression, feelings of emptiness, dissociation, and feelings of detachment were found to be bridge symptoms between borderline personality disorder and PTSD. Targeting these might allow better prevention and management...
Recent JCP Articles on Personality Disorders
Meta-Analysis
Mechanisms of Treatment of Bipolar Disorder and Borderline Personality Disorder: Brain Imaging Research
March 27, 2023
Different brain regions were changed with psychotherapy versus medication. The findings may help with predicting treatment efficacy and identifying treatment biomarkers in these disorders.
Recent PCC Articles on Personality Disorders
Case Report
Comorbid Persistent Genital Arousal Disorder and Borderline Personality Disorder
January 10, 2023
This case highlights the challenges that patients with both persistent genital arousal disorder and borderline personality disorder face, as well as the multidisciplinary approach needed to provide evidence-based care.
Featured Personality Disorders Research
Original Research
ECT for Depressed Patients With and Without BPD
January 19, 2021
Borderline personality disorder (BPD) is thought to lead to poor response to electroconvulsive therapy (ECT) in depressed patients. This study assessed symptom-severity outcomes among depressed patients with and without...
Original Research
Psychoeducation for Borderline Personality Disorder Patients
December 31, 2019
Most evidence-based treatments for borderline personality disorder (BPD) are prohibitively lengthy and intensive. This study explores a psychoeducational group intervention as a possible treatment alternative for this underserved patient...
Original Research
Borderline Personality Disorder and Eating Disorders: The Role of Emotion Regulation
July 3, 2024
Deficits in emotion regulation significantly predicted borderline personality disorder (BPD). Although eating disorders were not significantly predicted by emotion regulation deficits, they did predict BPD diagnoses.
Original Research
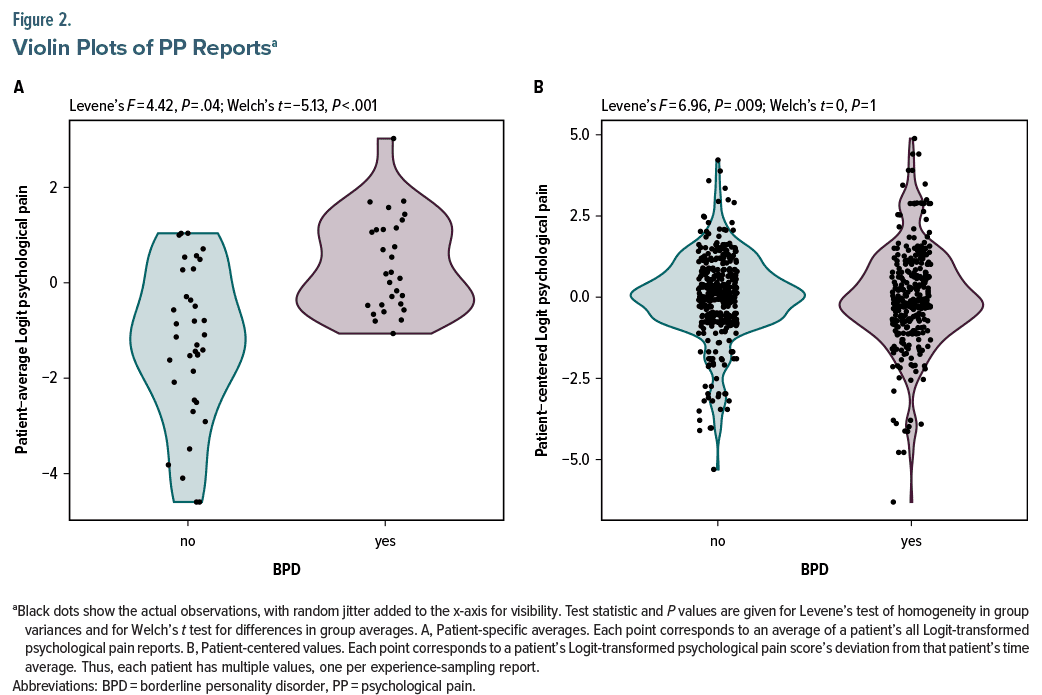
Psychological Pain as a Risk Factor for Suicidal Ideation
April 8, 2024
In inpatients with depression, psychological pain is a risk factor for suicidal ideation. Those with borderline personality disorder may experience more prominent and temporally unstable psychological pain, likely underlying...