Tapentadol, a dual-action synthetic opioid approved in 2008 for acute pain and in 2011 for chronic pain,1 offers advantages over tramadol, including higher μ-receptor affinity, stronger noradrenergic activity, and reduced cytochrome P450 2D6 dependence.2,3 Despite its initial reputation for low abuse potential, increasing reports from India describe intravenous misuse due to its short half-life and euphoric effects.4,5 Intranasal tapentadol, introduced in 2020 with proposed better bioavailability, lacks safety and abuse data.6 We present 2 cases of intranasal tapentadol dependence marked by rapid tolerance and significant physical and psychological harms.
Case 1
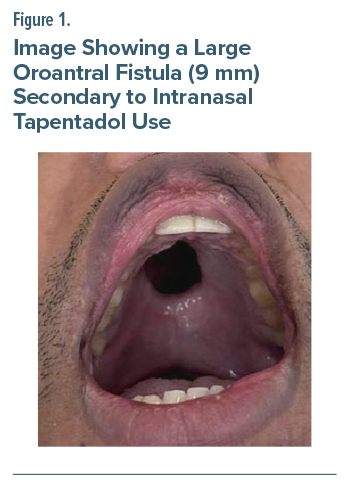
A 35-year-old man was prescribed intranasal tapentadol for lumbar disc pain 2 years ago. He escalated the dose rapidly from a few puffs daily to 15 puffs per session (22.5 mg/puff), 3 to 4 times a day, amounting to nearly 1–1.3 g of tapentadol daily (about 2 bottles). Despite initial nasal burning, he experienced a soothing and euphoric effect that reinforced continued use. He also felt relieved from his hectic work schedule and distressing thoughts about his mother’s recent demise. Within months, he developed nasal vasculitis and mucormycosis requiring extensive surgical debridement, leading to facial disfigurement, nasal collapse, and a large oroantral fistula (Figure 1). Persistent use despite these complications caused recurrent acute-on-chronic pancreatitis, severe weight loss, social withdrawal, and depressive symptoms marked by guilt, hopelessness, and passive death wishes. On evaluation, he was underweight and in moderate to severe opioid withdrawal. He was admitted for detoxification and managed with buprenorphine up to 8 mg/d, quetiapine 50 mg/d, and later escitalopram 10 mg/d, alongside psychotherapy and family psychoeducation. After stabilization, he achieved 3 months of abstinence, with marked improvement in mood, appetite, nasal health, and occupational functioning.
Case 2
A 30-year-old man with an internalizing temperament and anxious-avoidant traits presented with dependence on sedatives (clonazepam 2–3 mg/d and erratic use of chlordiazepoxide/zolpidem/ alprazolam) for the last 5 years. His family history was notable for anxiety disorders and benzodiazepine dependence in his parents. Medically, he had experienced recurrent episodes of pancreatitis, the last one occurring 6 months before presentation. He was introduced to intranasal tapentadol spray (22.5 mg/puff) for a bout of acute pancreatitis 2 years ago. He began with initial use of 1–2 puffs daily and discovered that beyond pain relief, the spray provided a subjective sense of calm, enhanced well-being, and improved self-confidence and facilitated smoother social interactions. This reinforcing effect prompted escalation, and within months, his consumption increased to 20–25 puffs daily (450–550 mg/d). His use fulfilled criteria for opioid use disorder according to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. He described withdrawal symptoms of rhinorrhea, tremulousness, anxiety, and diffuse body aches, which drove repeated use. He also reported mucosal crusting and development of a nasal septal perforation requiring surgical intervention. With abstinence from tapentadol, his baseline anxiety symptoms reemerged, dominated by morning autonomic arousal and somatic complaints. Multiple concurrent benzodiazepines were stopped, and diazepam 20 mg/d was introduced on a tapering schedule. Escitalopram up to 20 mg/d and pregabalin 300 mg/d were added for anxiety symptoms. Concurrently, psychotherapy sessions focused on anxiety management, coping skill enhancement, and relapse prevention strategies.
Discussion
Intranasal tapentadol, marketed for superior bioavailability and convenience, carries significant abuse potential. The cases described demonstrate rapid dose escalation driven by its euphoric effects and ease of access. Both patients developed severe nasal complications—vasculitis, mucormycosis, septal perforation, and oroantral fistula—requiring surgical intervention in line with previous reports6–8 alongside psychiatric and systemic harms such as worsening anxiety and recurrent pancreatitis. These findings underscore the drug’s potential for rapid dependence and serious local and systemic toxicity, warranting greater clinical caution and regulatory oversight.
Article Information
Published Online: February 26, 2026. https://doi.org/10.4088/PCC.25cr04114
© 2026 Physicians Postgraduate Press, Inc.
Prim Care Companion CNS Disord 2026;28(1):25cr04114
Submitted: October 17, 2025; accepted November 26, 2025.
To Cite: Gakkhar S, Jha A, Pazhanivel V, et al. Intranasal tapentadol dependence and emerging patterns of harm: a report of 2 cases. Prim Care Companion CNS Disord 2026;28(1):25cr04114.
Author Affiliations: Department of Psychiatry, National Institute of Mental Health and Neurosciences, Bengaluru, India (all authors).
Corresponding Author: Jayant Mahadevan, MD, DM, Department of Psychiatry, National Institute of Mental Health and Neurosciences (NIMHANS), Bengaluru, India ([email protected]).
Relevant Financial Relationships: None.
Funding/Support: None.
Patient Consent: Consent was received from the patients to publish the case reports, and information (including dates) has been de-identified to protect anonymity.
References (8)

- Zajączkowska R, Przewłocka B, Kocot-Kępska M, et al. Tapentadol – a representative of a new class of MOR-NRI analgesics. Pharmacol Rep. 2018;70(4):812–820. PubMed CrossRef
- Zavaleta-Monestel E, Anchía-Alfaro A, Villalobos-Madriz J, et al. Tapentadol: a comprehensive review of its role in pain management. Cureus. 2024;16(11):e74307. PubMed CrossRef
- Faria J, Barbosa J, Moreira R, et al. Comparative pharmacology and toxicology of tramadol and tapentadol. Eur J Pain. 2018;22(5):827–844. PubMed CrossRef
- Mukherjee D, Shukla L, Saha P, et al. Tapentadol abuse and dependence in India. Asian J Psychiatr. 2020;49:101978. PubMed CrossRef
- Shivaprakash P, Shukla L, Joshi S, et al. Tapentadol as a drug of abuse – a preliminary report. Indian J Psychiatry. 2025;67(2):256–259. PubMed CrossRef
- Mazhari MdKH, Chandra J, Sequeira JP. Tapentadol nasal spray is an effective way of pain management after tooth extraction: a randomized control clinical trial. J Pharm Res Int. 2023;35(8):9–14. CrossRef
- Kumar PNS, Edwin G, Suresh R, et al. A nose for trouble: tapentadol nasal spray use disorder associated nasal cartilage atrophy in a young male. Asian J Psychiatry. 2025;103:104346. PubMed CrossRef
- Swain SK, Parida PK, Saleem S. Longstanding use of tapentadol hydrochloride nasal spray resulting in palatal perforation − A case report. Med J Dr DY Patil Vidyapeeth. 2025;18(2):338. CrossRef
Enjoy this premium PDF as part of your membership benefits!