Ketamine is a phencyclidine derivative with N-methyl-D-aspartate receptor antagonist properties used for decades as an anesthetic and analgesic. At subanesthetic doses, it produces dissociative and psychotropic effects, prompting inquiry into its utility for psychiatric conditions. Racemic ketamine, a mixture of equal parts R- and S-enantiomers, has been studied off-label for treatment-resistant depression and other psychiatric disorders.1 More recently, intranasal S-ketamine (esketamine) received US Food and Drug Administration approval for treatment-resistant depression.
Outside of clinical settings, ketamine has a long history of nonmedical use. Early surveys in the 1990s documented recreational use in clubs, concerts, and “rave” environments, driven by its dissociative and hallucinogenic properties. In the United States, population-based survey data indicate that recreational ketamine use, while still uncommon, has increased over the past decade. Analyses from the National Survey on Drug Use and Health show that past-year ketamine use among adults increased by 81% from 0.11% in 2015 to 0.20% in 2019 and subsequently increased by 40% from 0.20% in 2021 to 0.28% in 2022, suggesting growing population-level exposure.2 Adults reporting past-year ketamine use were initially concentrated in younger age groups, but more recent increases have been observed among adults aged 26–34 years, indicating a broadening demographic of recreational users.2,3 Complementing these survey findings, drug seizure records demonstrate a substantial increase in illicit ketamine availability in the United States between 2017 and 2022.4
Illicit ketamine is obtained through several channels. Historically, supplies have come from veterinary diversion, international trafficking, or local synthesis. Some users acquire ketamine through dark web marketplaces, purchased with cryptocurrency to maintain anonymity.5 More recently, concerns have arisen about telehealth prescribing platforms that provide ketamine with little or no formal evaluation for off-label psychiatric indications,6 with some patients escalating doses or diverting medication for nonmedical use. This evolving access underscores the overlap between therapeutic exposure and recreational misuse.
Recreational ketamine can be acquired in liquid or powder forms. Powdered ketamine is commonly insufflated. Liquid ketamine is injected intravenously or intramuscularly or, less commonly, ingested orally or used rectally.7,8 Orally ingested ketamine is rapidly metabolized to norketamine, producing more of a sedative and less of a psychedelic experience.7 Compared with oral use, intranasal or parenteral administration provides faster onset and greater intensity of dissociative effects. Street formulations vary in purity, and slang terms such as “Special K,” “Vitamin K,” or “Kit-Kat” reflect its reputation in recreational drug culture.
The reinforcing appeal of ketamine lies in its rapid onset of euphoria, increased sociability, dissociation, and altered perception of time and space.7,8 Its relatively short duration of action, low cost, and accessibility make it attractive to those seeking intense psychoactive experiences.9 Tolerance develops quickly, necessitating escalating doses to achieve similar effects,9 increasing the risks of psychiatric and physical complications.
Chronic, heavy use has been strongly linked to ketamine-induced uropathy (KIU),10,11 which typically presents with cystitis-like lower urinary tract symptoms (LUTS).12,13 KIU can progress to severe bladder and upper urinary tract injury if use continues.14,15
Despite increasing recognition in urologic literature, KIU remains underrecognized in psychiatric and primary care settings, leading to extensive evaluation for infection or interstitial cystitis, repeated empirical antibiotic treatment despite negative urine cultures, and disease progression.16,17 Without awareness of ketamine exposure, the underlying etiology may go undetected, delaying appropriate treatment and drug cessation. For primary care physicians, psychiatrists, and other mental health providers, consideration of recreational ketamine use is therefore essential when patients present with unexplained urinary complaints.
Here, we present 2 cases that illustrate the psychiatric and urologic consequences of KIU. These reports highlight the diagnostic challenges posed by KIU, the role of psychiatric comorbidity in ongoing use, and the need for multidisciplinary management.
Case 1
A 33-year-old man was repeatedly evaluated by his primary care physician over a 2-year period for recurrent LUTS, including frequency, urgency, nocturia, and intermittent hematuria, which had become disruptive to daily functioning. Urinalysis revealed few white cells and mild proteinuria. Although urine cultures throughout the course of treatment were consistently negative, he was empirically treated with trimethoprim 100 mg twice/day and doxycycline 100 mg every 12 hours for presumed urinary tract infections and referred for urologic evaluation.
Computed tomography (CT) of the abdomen and pelvis revealed diffuse bladder wall thickening, possible endophytic lesions, and mild prostatomegaly, without hydronephrosis or renal masses. Cystoscopy demonstrated marked posterior bladder wall inflammation. He was initially treated with nonsteroidal anti-inflammatory drugs. Due to persisting symptoms, repeat cystoscopy with biopsy 1 month later revealed a contracted bladder and diffuse cystitis with mixed inflammatory infiltrate, prominently eosinophilic and neutrophilic.
When the urologist inquired about substance use, the patient disclosed intermittent recreational ketamine use since the COVID-19 pandemic. He was informed of the association between ketamine use and urinary symptoms and agreed to abstain. Symptomatic improvement was noted over the following month. He was referred to chemical dependency treatment, although follow-up was not confirmed.
Three months later, he presented to the hospital with worsening abdominal pain, dysuria, urgency, suprapubic discomfort, and fatigue due to nocturia. Episodes of incontinence increasingly led to social isolation. Psychiatric evaluation was requested for dysphoria related to the impact of his urinary symptoms on his functioning. Psychiatric interview unveiled long-standing ketamine use, with intermittent use of cocaine, cannabis, alcohol, and hallucinogens. Ketamine was his drug of choice due to low cost and accessibility. Due to tolerance, his use escalated quickly to 3 g/day. He acknowledged having been informed that his urinary symptoms were related to ketamine use. By his own admission, he had complete resolution of symptoms when he abstained from ketamine previously. He acknowledged that he relapsed into daily use in the month preceding hospitalization. He volunteered, “I really need to stop using,” but acknowledged that his attempts to abstain were limited.
Imaging revealed new-onset moderate bilateral hydronephrosis without obstruction. Laboratory studies demonstrated renal and hepatic function abnormalities (ie, aspartate transferase: 83 U/L, alanine transaminase: 180 U/L, alkaline phosphatase: 1,839 IU/L, and total bilirubin: 1.6 mg/dL), though imaging of the liver and biliary tree was unremarkable. He underwent infrared-guided bilateral nephrostomy tube placement, resulting in normalization of renal function. His dysphoria improved with symptom resolution.
Motivational interviewing techniques were employed to support commitment to abstinence. Supportive management included bladder decompression and counseling regarding persistent bladder dysfunction and risk of further renal compromise if ketamine use resumed. He was referred for urology and chemical dependency outpatient resources.
Case 2
A 30-year-old woman with a history of childhood trauma and posttraumatic stress disorder began using recreational ketamine following the death of her best friend as a means of coping with grief and dissociative symptoms. Over the next 3 years, her ketamine use escalated due to tolerance, reaching approximately 2–6 grams daily administered rectally.
During this period, she developed progressive LUTS—urinary frequency, urgency, dysuria, and suprapubic pain—for which she repeatedly sought medical attention. She often received empiric antibiotic therapy for presumed urinary tract infection, but her urine cultures were consistently negative. Prior CT imaging of the abdomen and pelvis demonstrated diffuse bladder wall thickening without hydronephrosis or renal masses, and she had been referred to urology but did not follow up.
Over the course of her illness, she experienced intermittent periods of abstinence from ketamine lasting approximately 2–5 months at a time. During these abstinence periods, she reported gradual but incomplete improvement in urinary frequency and bladder discomfort, with recurrence of symptoms following relapse. She further noted that with each subsequent return to ketamine use, urinary symptoms became more severe, and bladder pain emerged more rapidly than during earlier episodes. Her longest continuous period of abstinence was 5 months.
As her urinary symptoms worsened and became increasingly refractory, she began using opioids obtained from nonmedical sources to manage bladder pain. Her opioid use escalated, ultimately resulting in admission to an inpatient chemical-dependency rehabilitation program, where she was treated with buprenorphine.
While in rehabilitation, she again complained of LUTS. Urinalysis revealed mild hematuria, and she was initially treated with nitrofurantoin for a presumed urinary infection; this was discontinued after cultures remained negative. Repeat CT imaging of the abdomen and pelvis revealed bladder wall thickening, mucosal enhancement, perivesical inflammation, and reduced bladder capacity.
At this time, she disclosed the extent and pattern of her prior ketamine use, leading clinicians to suspect KIU as the cause of her chronic urinary symptoms. She was referred for urologic evaluation, including cystoscopy and biopsy, and was provided resources for ongoing outpatient substance use treatment and trauma-focused psychotherapy following discharge from the rehabilitation program.
Discussion
These 2 cases illustrate several important clinical themes in KIU. Both patients experienced recurrent urinary symptoms in the absence of infection, with improvement during periods of ketamine abstinence and relapse upon resumption, reinforcing a causal relationship between ketamine exposure and urinary tract injury. The cases also demonstrate the spectrum of severity ranging from debilitating LUTS prompting opioid misuse (Case 2) to progression to hydronephrosis and renal impairment requiring nephrostomy (Case 1). In both cases, diagnostic delay contributed to prolonged symptoms, underscoring the importance of explicitly inquiring about ketamine use in patients with atypical urinary complaints.
Epidemiologic work suggests that approximately 25% of recreational ketamine users develop urinary symptoms, with risk strongly associated with dose and frequency of use.18 The pathophysiology of KIU remains incompletely understood. Proposed mechanisms include prolonged exposure of the urothelium to ketamine metabolites, particularly norketamine,19 producing direct urothelial toxicity, microvascular injury, neurogenic inflammation, and immune dysregulation.8,19,20 Chronic injury may lead to persistent inflammation and fibrosis, causing bladder wall thickening, reduced capacity, vesicoureteral reflux, hydronephrosis, and ultimately renal dysfunction.8,14,15 Ketamine-induced hydronephrosis is a strong, independent predictor of renal function decline and eventual end-stage renal disease, underscoring the urgency of early recognition and cessation.21 Discontinuation of ketamine use remains the only consistently effective strategy to promote symptom resolution and prevent irreversible renal injury,22 although urologic and pain management strategies23 are often needed to address persistent symptoms.
Chronic ketamine use has also been associated with hepatobiliary injury, including elevated transaminases and cholestatic liver enzyme patterns (Case 1), and, in some cases, biliary tract abnormalities.24,25 Concurrent hepatobiliary and urinary tract toxicity may reflect shared metabolic or inflammatory pathways. Elevated cholestatic liver enzymes, ie, alkaline phosphatase, have been correlated with KIU severity and worse renal outcomes, particularly among patients with hydronephrosis.21
An important clinical feature highlighted by these cases is the psychiatric and behavioral context in which KIU develops. Co-occurring psychiatric conditions, such as trauma-related distress, grief, and depression, may delay recognition and complicate cessation. Additionally, patients may fail to recognize the association between ketamine use and urinary symptoms. Moreover, because ketamine has analgesic properties, some individuals may increase use to self-manage suprapubic pain, paradoxically worsening underlying injury.
Although recreational, long-term, high-dose use is frequently associated with a range of serious urinary complications, therapeutic ketamine administered under medical supervision appears to carry minimal risk based on large-scale provider surveys.1,26 To date, there is only one confirmed published case, to our knowledge, of cystitis attributed to therapeutic ketamine administration for depression.27 This distinction is clinically important to avoid undue reluctance to employ therapeutic ketamine in appropriate psychiatric contexts. Nevertheless, ongoing monitoring for urinary symptoms and attention to hydration are prudent during maintenance treatment or dose escalation.28
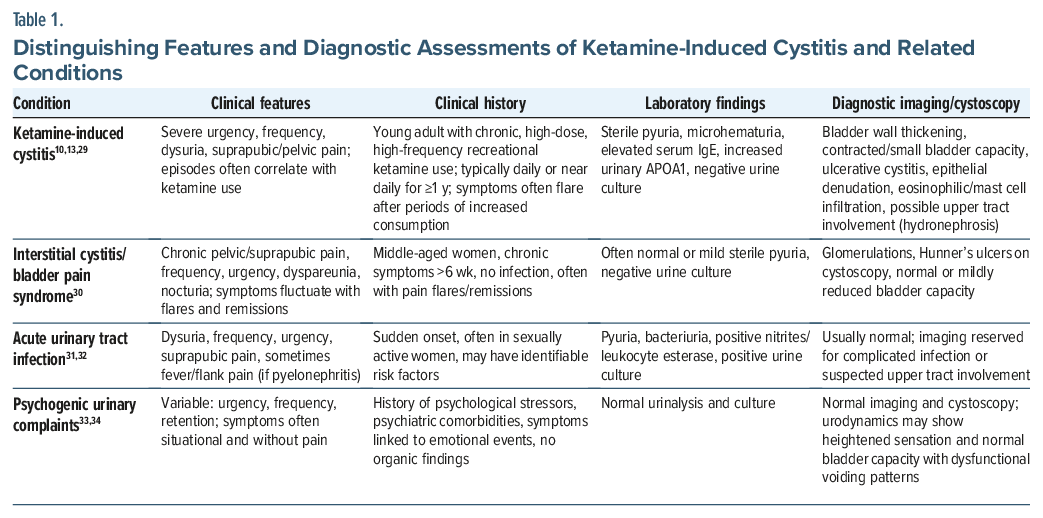
The clinical presentation of early KIU can overlap with interstitial cystitis/bladder pain, urinary tract infection, or psychogenic urinary complaints, making differential diagnosis essential (Table 1). Features such as younger age, frequent ketamine use, sterile pyuria, episodic symptom flares temporally linked to use, and rapid progression to reduced bladder capacity or upper tract involvement should raise suspicion for KIU.
Management of KIU is inherently multidisciplinary.35 Primary care clinicians play a key role in early recognition and longitudinal monitoring, urologists assess bladder and upper tract involvement and manage complications, and addiction specialists provide person-centered treatment supporting sustained ketamine abstinence. Given the association between ketamine use and co-occurring mood or anxiety disorders, integrated psychiatric and psychological care, including cognitive-behavioral therapy and other therapeutic modalities, is essential to address drivers of ongoing use and reduce relapse risk.36–38
Article Information
Published Online: March 19, 2026. https://doi.org/10.4088/PCC.25br04121
© 2026 Physicians Postgraduate Press, Inc.
Prim Care Companion CNS Disord 2026;28(2):25br04121
Submitted: October 28, 2025; accepted December 16, 2025.
To Cite: Leo RJ, Blaes SL, Jaffri MA, et al. Recreational ketamine-induced uropathy: recognition and management in primary care and psychiatric practice. Prim Care Companion CNS Disord 2026;28(2):25br04121.
Author Affiliations: Department of Psychiatry, Jacobs School of Medicine and Biomedical Sciences, State University of New York at Buffalo, Buffalo, New York (Leo, Blaes, Jaffri, Fleming); State University of New York at Niagara and Northern Erie Clinical Services, Erie County Medical Center, Buffalo, New York (Galvano).
Corresponding Author: Raphael J. Leo, MA, MD, Erie County Medical Center, 462 Grider St, Buffalo, New York 14215 ([email protected]).
Financial Disclosure: None.
Funding/Support: None.
Patient Consent: Consent was received from the patients to publish the case reports, and information was de-identified to protect patient anonymity.
ORCID: Raphael J. Leo: https://orcid.org/0000-0003-1373-3230
Clinical Points
- Ketamine-induced uropathy should be suspected in younger patients with persistent or episodic lower urinary tract symptoms and negative urine cultures, particularly when symptoms fluctuate with substance use.
- Frequency and cumulative exposure to recreational ketamine are key determinants of bladder and upper urinary tract injury, with relapse often leading to rapid symptom recurrence or progression.
- Early recognition, explicit inquiry about ketamine use, and sustained cessation supported by multidisciplinary care are essential to preventing irreversible urologic and renal complications.
References (38)

- Wilkinson ST, Katz RB, Toprak M, et al. Acute and longer-term outcomes using ketamine as a clinical treatment at the yale psychiatric hospital. J Clin Psychiatry. 2018;79(4):17m11731. PubMed CrossRef
- Yang KH, Kepner W, Cleland CM, et al. Trends and characteristics in ketamine use among US adults with and without depression, 2015-2022. J Affect Disord. 2025;373:345–352. PubMed CrossRef
- Yockey RA. Past-year ketamine use: evidence from a United States population, 2015-2019. J Psychoactive Drugs. 2023;55(2):134–140. PubMed CrossRef
- Palamar JJ, Wilkinson ST, Carr TH, et al. Trends in illicit ketamine seizures in the US from 2017 to 2022. JAMA Psychiatry. 2023;80(7):750–751. PubMed CrossRef
- U.S. Department of Justice. National Drug Intelligence Center: National Drug Threat Assessment. 2010. https://www.justice.gov/archive/ndic/pubs10/10255/10255p.pdf
- Fallik D. Dysregulated: the patchwork policies of ketamine therapy. Undark Mag. 2025. https://undark.org/2025/06/04/ketamine-therapy-regulation/. Accessed October 19, 2025.
- Morgan CJ, Curran HV. Independent scientific committee on drugs. Ketamine use: a review. Addiction. 2012;107(1):27–38. PubMed CrossRef
- Myers FA, Jr, Bluth MH, Cheung WW. Ketamine: a cause of urinary tract dysfunction. Clin Lab Med. 2016;36(4):721–744. PubMed CrossRef
- Sassano-Higgins S, Baron D, Juarez G, et al. A review of ketamine abuse and diversion. Depress Anxiety. 2016;33(8):718–727. PubMed CrossRef
- Castellani D, Pirola GM, Gubbiotti M, et al. What urologists need to know about ketamine-induced uropathy: a systematic review. Neurourol Urodyn. 2020;39(4):1049–1062. PubMed CrossRef
- Chan EOT, Chan VWS, Tang TST, et al. Systematic review and meta-analysis of ketamine-associated uropathy. Hong Kong Med J. 2022;28(6):466–474. PubMed CrossRef
- Pal R, Balt S, Erowid E, et al. Ketamine is associated with lower urinary tract signs and symptoms. Drug Alcohol Depend. 2013;132(1–2):189–194. PubMed CrossRef
- Shahani R, Streutker C, Dickson B, et al. Ketamine-associated ulcerative cystitis: a new clinical entity. Urology. 2007;69(5):810–812. PubMed CrossRef
- Yee CH, Teoh JY, Lai PT, et al. The risk of upper urinary tract involvement in patients with ketamine-associated uropathy. Int Neurourol J. 2017;21(2):128–132. PubMed CrossRef
- Zheng Z, Li Z, Yuan J, et al. Ketamine-associated upper urinary tract dysfunction: what we know from current literature. Asian J Urol. 2025;12(1):33–42. PubMed CrossRef
- Lai Y, Wu S, Ni L, et al. Ketamine-associated urinary tract dysfunction: an underrecognized clinical entity. Urol Int. 2012;89(1):93–96. PubMed CrossRef
- Wood D, Cottrell A, Baker SC, et al. Recreational ketamine: from pleasure to pain. BJU Int. 2011;107(12):1881–1884. PubMed CrossRef
- Winstock AR, Mitcheson L, Gillatt DA, et al. The prevalence and natural history of urinary symptoms among recreational ketamine users. BJU Int. 2012;110(11):1762–1766. PubMed CrossRef
- Lin JW, Lin YC, Liu JM, et al. Norketamine, the main metabolite of ketamine, induces mitochondria-dependent and ER stress-triggered apoptotic death in urothelial cells via a Ca2+-regulated ERK1/2-activating pathway. Int J Mol Sci. 2022;23(9):4666. PubMed CrossRef
- Xie X, Liang J, Huang R, et al. Molecular pathways underlying tissue injuries in the bladder with ketamine cystitis. FASEB J. 2021;35(7):e21703. PubMed CrossRef
- Ou SH, Wu LY, Chen HY, et al. Risk of renal function decline in patients with ketamine-associated uropathy. Int J Environ Res Public Health. 2020;17(19):7260. PubMed CrossRef
- Malik NM, Kumar H, Perera KK. Ketamine-induced bladder syndrome: recognition as a key differential diagnosis and the need to curb recreational use. Indian J Case Rep. 2025;11(6):254–256.
- Kalsi SS, Wood DM, Dargan PI. The epidemiology and patterns of acute and chronic toxicity associated with recreational ketamine use. Emerg Health Threats J. 2011;4:7107. PubMed CrossRef
- Chun J, Kotha S, Nayagam J, et al. Ketamine cholangiopathy: clinical features and liver biopsy findings. Histopathology. 2025;87(1):130–137. PubMed CrossRef
- Wong GL, Tam YH, Ng CF, et al. Liver injury is common among chronic abusers of ketamine. Clin Gastroenterol Hepatol. 2014;12(10):1759–1762. PubMed CrossRef
- Kerr-Gaffney J, Tröger A, Caulfield A, et al. Urological symptoms following ketamine treatment for psychiatric disorders: a systematic review. J Psychopharmacol. 2025:2698811251350267.
- Chang M, Juruena MF, Young AH. Ketamine cystitis following ketamine therapy for treatment-resistant depression -case report. BMC Psychiatry. 2024;24(1):9. PubMed CrossRef
- Andrade C. Ketamine-associated uropathy during therapeutic and nontherapeutic use: prevalence, clinical features, mechanisms, and strategies for risk reduction. J Clin Psychiatry. 2025;86(4):25f16083. PubMed CrossRef
- Jhang JF, Hsu YH, Kuo HC. Possible pathophysiology of ketamine-related cystitis and associated treatment strategies. Int J Urol. 2015;22(9):816–825. PubMed CrossRef
- Clemens JQ, Erickson DR, Varela NP, et al. Diagnosis and treatment of interstitial cystitis/bladder pain syndrome. J Urol. 2022;208(1):34–42. PubMed CrossRef
- Chu CM, Lowder JL. Diagnosis and treatment of urinary tract infections across age groups. Am J Obstet Gynecol. 2018;219(1):40–51. PubMed CrossRef
- Hooton TM. Clinical practice. Uncomplicated urinary tract infection. N Engl J Med. 2012;366(11):1028–1037. PubMed CrossRef
- Panicker JN, Selai C, Herve F, et al. Psychological comorbidities and functional neurological disorders in women with idiopathic urinary retention: international Consultation on Incontinence Research Society (ICI-RS) 2019. Neurourol Urodyn. 2020;39(suppl 3):S60–S69. PubMed CrossRef
- Sakakibara R, Uchiyama T, Awa Y, et al. Psychogenic urinary dysfunction: a uro-neurological assessment. Neurourol Urodyn. 2007;26(4):518–524. PubMed CrossRef
- Robles-Martínez M, Abad AC, Pérez-Rodríguez V, et al. Delayed urinary symptoms induced by ketamine. J Psychoactive Drugs. 2018;50(2):129–132. PubMed CrossRef
- Kutscher E, Greene RE. Ketamine cystitis: an underrecognized cause of dysuria. J Gen Intern Med. 2022;37(5):1286–1289. PubMed CrossRef
- Abdelrahman A, Belal M. Rare but relevant: ketamine-induced cystitis -an in-depth review for addiction medicine. Addiction. 2025;120(8):1689–1693. PubMed CrossRef
- Anderson DJ, Zhou J, Cao D, et al. Ketamine-induced cystitis: a comprehensive review of the urologic effects of this psychoactive drug. Health Psychol Res. 2022;10(3):38247. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!