Abstract
Objective: To evaluate if ultra–low-dose mirtazapine (3.75 mg) improves insomnia without next-day effects.
Methods: This retrospective study evaluated data collected from September 5, 2024, to March 7, 2025, from an outpatient setting consisting of veterans with insomnia who were treated with ultra–low-dose mirtazapine. The Insomnia Severity Index (ISI) was administered during the first appointment and at each subsequent visit with the respective psychiatrist to monitor insomnia symptoms. Summary statistics were used to compare ISI scores at baseline and 1–3 months after starting treatment.
Results: Considering all veterans evaluated (N = 53), 47% showed a meaningful decrease in ISI value (greater than 7 points). Patients who completed treatment showed a constant or decreased ISI score (mean [SD] change: 11.3 [6.46]).
Conclusion: Ultra–low-dose mirtazapine may improve symptoms and ISI values for chronic insomnia.
Prim Care Companion CNS Disord 2026;28(2):25m04074
Author affiliations are listed at the end of this article.
A chronic insomnia disorder is generally characterized by a persistent difficulty with sleep initiation or maintenance, associated with a perceived daytime impairment or concern/ dissatisfaction with sleep occurring for at least 3 nights per week and for a period of at least 3 months.1,2 Criteria include patient or caregiver report of difficulties with initiating sleep, maintaining sleep, waking up earlier than desired, resistance to going to bed on an appropriate schedule, or difficulty sleeping without caregiver intervention. The difficulty is accompanied by a daytime consequence such as fatigue/malaise; impaired attention, concentration, or memory; impaired social, occupational, or academic performance; mood disturbance; daytime sleepiness; behavior problems; reduced motivation, energy, or initiative; proneness for error or accidents; or a concern or dissatisfaction with sleep.1,2 These symptoms occur despite adequate opportunity and circumstances for sleep and are not better explained by an alternative diagnosis or medication.1,2
The gold standard treatment of insomnia, cognitive-behavioral therapy for insomnia (CBTi), has been shown to shorten sleep latency and reduce wake after sleep onset by an average of 19 minutes and 26 minutes, respectively,3 and also to improve total sleep time and sleep efficiency by 8 minutes and 10%, respectively.3 Treating insomnia can be challenging given that CBTi is not recommended or contraindicated in certain patients secondary to its sleep restriction component.4 These cases may include epilepsy, bipolar disorders, and those at high risk for falls. Sleep restriction can also exacerbate symptoms for those with untreated disorders of excessive sleepiness and with parasomnias.4
Low doses of mirtazapine are commonly prescribed as an alternative treatment when there is a need for sleep medication that can be used for several months,5 with higher dosages used to treat depression.6,7 The typical dose range for depression is 15–45 mg in the evening. Lower mirtazapine doses (7.5–15 mg) tend to be prescribed to promote sleep due to concern about excessive activation at higher doses. Studies have demonstrated that low-dose mirtazapine, 7.5 mg, exerts significant effects on sleep duration and sleep architecture by reducing wakefulness after sleep onset and increasing total sleep time.8 Literature also suggests that 30 mg improves sleep efficiency while decreasing the number of awakenings.9
The objective of this study was to systematically evaluate the efficacy and tolerability of low-dose mirtazapine, 3.75 mg, in individuals with chronic insomnia in a Veterans Affairs psychiatric practice.
METHODS
This retrospective chart review evaluated data collected from September 5, 2024, to March 7, 2025, in an outpatient setting consisting of veterans aged 31–81 years who were prescribed an ultra-low dose of mirtazapine due to the lack of efficacy of traditional sleep aids (N = 53, 49 males and 4 females). The primary study outcome was insomnia severity as measured by the validated Insomnia Severity Index (ISI)10,11 collected at baseline and 1–3 months after initiating therapy. Data were analyzed using summary statistics (means, SDs, and proportions). The mean ISI score for each visit, the mean change in ISI score for patients who completed treatment, the proportion of patients who reported clinically significant improvement (ISI score more than 7 points lower than baseline), and the proportion of patients who achieved recovery (ISI score of 7 or lower) were calculated. Means and SDs were calculated for the 1-to 3-month visit based on patients who completed treatment and had a valid ISI score. Reasons for discontinuation and side effects were reported.
RESULTS
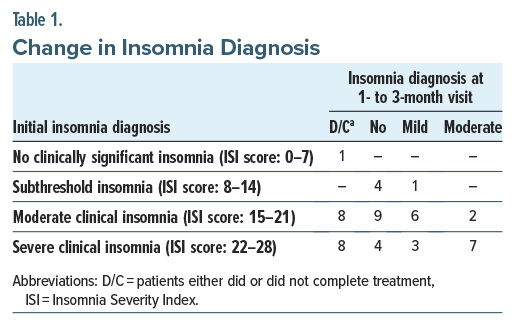
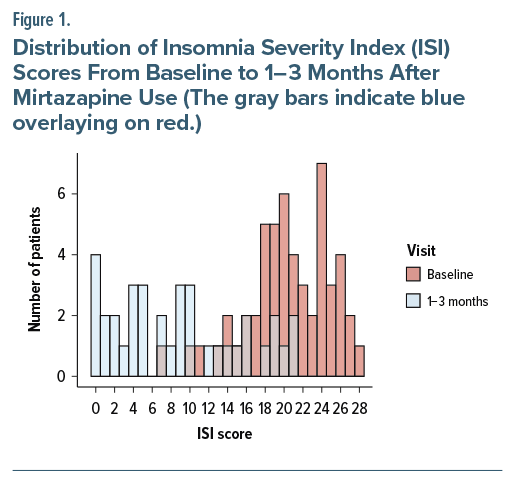
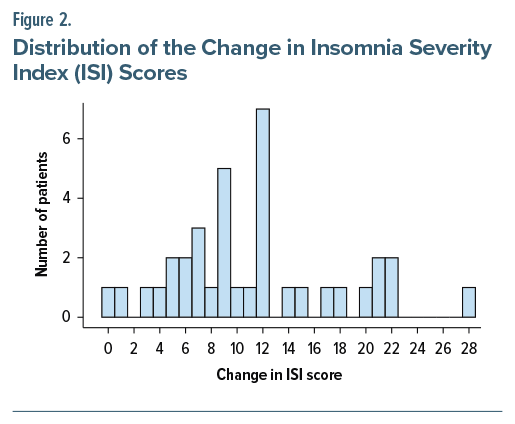
Considering all veterans evaluated, 47% showed a clinically significant decrease in ISI score (greater than 7 points), while 32% of patients achieved recovery (ISI score of 7 or lower). Table 1 illustrates the change in insomnia diagnosis. The mean ISI score was lower for the 1-to 3-month visit compared to the baseline visit (8.9 [6.76] vs. 20.4 [4.58]). Figure 1 illustrates a shift in the distribution of ISI scores from baseline to 1–3 months after mirtazapine usage. ISI scores for the 1-to 3-month visit are from patients who completed treatment. For veterans who completed treatment, all showed a constant or decreased ISI score 1–3 months after treatment (11.3 [6.46]). Figure 2 demonstrates the distribution of the change in ISI scores.
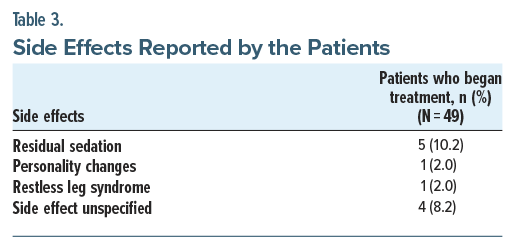
It is important to note that 17 of the 53 veterans evaluated did not complete treatment. Table 2 provides a summary of the discontinuations. Tolerability of mirtazapine 3.75 mg was assessed through the proportion of patients who experienced side effects. Table 3 presents the type of side effect, along with the proportion of those who experienced it. Summary statistics by visit are provided in the Supplementary Material.
Subjectively, individuals noted an increase in total sleep time. Similar increases in total sleep time were reported for low-dose esmirtazapine in industry-funded trials.12-15
DISCUSSION
This retrospective study in 53 patients with insomnia disorder demonstrated a clinically relevant effect of low-dose mirtazapine (3.75 mg) on insomnia severity from 1 to 3 months. This is the first study, to our knowledge, investigating 3.75 mg of mirtazapine on insomnia severity for the short term.
Our data do not support the long-term effectiveness of mirtazapine but serve as a foundation for future studies. Additional evidence regarding mirtazapine as a monotherapy in the treatment of insomnia will be needed, as it was not included in a 2018 systematic review of antidepressants used for insomnia in adults.16 It is also not reviewed in the American Academy of Sleep Medicine practice guideline on chronic insomnia.17
However, in individuals who do not experience efficacy from other insomnia medications (eg trazodone, amitriptyline, doxepin), low-dose mirtazapine can be considered. It may also be a safer approach compared to soporific medications that have dependence concerns such as benzodiazepines.17
In a double-blinded, placebo-controlled study by Aslan et al,18 the effect of acute administration of mirtazapine 30 mg on polysomnography outcomes was investigated in young healthy adults. Compared with placebo, mirtazapine was shown to improve sleep efficiency, decrease the frequency and duration of nocturnal awakenings, and increase the duration of slow-wave sleep (SWS). SWS is thought to facilitate glymphatic system functioning, a process responsible for the clearance of protein waste from the brain. The disruption of glymphatic function is thought to contribute to the progression of neurodegenerative dementias.19 Future studies utilizing mirtazapine to reduce sleep fragmentation and increase SWS duration are needed to assess its possible role as a protective factor in diminishing the risk of developing dementia.
While this is a retrospective study, the mechanisms of action, clinical successes or failures, may result from individual differences in the etiology of insomnia, neurotransmitter receptor variances, or effectiveness in the functions of sleep on wake systems such as through glymphatic clearance during sleep.
Considering all patients who completed therapy, 47% of veterans showed a meaningful decrease in ISI value (greater than 7 points). Ultra–low-dose mirtazapine may produce an improvement in symptoms and ISI values for chronic insomnia with less possibility of residual sedation.
Limitations
The generalizability of our findings is circumscribed by the study design. This was a retrospective, small-sample study of veterans, most of whom were male. The greatest limitation in our study is the absence of a control group. Nonetheless, our study has several strengths. We had a substantial number of patients. Insomnia severity was confirmed through a reliable and valid instrument to quantify perceived insomnia severity.20 Despite these limitations, the finding that participants who completed treatment demonstrated both efficacy and tolerability from low-dose mirtazapine is noteworthy.
CONCLUSION
Our findings have both research and clinical implications. They provide support for randomized, controlled, prospective trials. They also should remind clinicians to consider low-dose mirtazapine for treatment-resistant insomnia in those with persistent insomnia symptoms despite adequate medication trials. Such symptoms may be quite treatment responsive.
Article Information
Published Online: March 10, 2026. https://doi.org/10.4088/PCC.25m04074
© 2026 Physicians Postgraduate Press, Inc.
Submitted: September 7, 2025; accepted December 3, 2025.
To Cite: Berbara R, Gandotra K, Zink E, et al. Efficacy and tolerability of ultra–low-dose mirtazapine in adult chronic insomnia. Prim Care Companion CNS Disord 2026;28(2): 25m04074.
Author Affiliations: Division of Pulmonary, Critical Care and Sleep Medicine, University Hospitals, Case Western Reserve University, Cleveland, Ohio (Gandotra, EL Ghoul, Strohl); Psychiatry Service, Louis Stokes VA Medical Center, Cleveland, Ohio (Gandotra, Kumari, Vaghela, Rubai, Shaver, Jaskiw); Department of Psychiatry, Case Western Reserve University, Cleveland, Ohio (Gandotra, Jaskiw); Geriatric Research Education and Clinical Center (GRECC), Louis Stokes VA Medical Center, Cleveland, Ohio (Zink, Wilson); Division of Pulmonary, Critical Care, and Sleep Medicine, Louis Stokes VA Medical Center, Cleveland, Ohio (Gandotra, EL Ghoul, Strohl); National PBM Clinical Pharmacy Program Manager, VHA Pharmacy Benefits Management Services, Hines, Illinois (Fuller); Department of Sleep Medicine, VA Ann Arbor Healthcare System, Ann Arbor, Michigan (Berbara).
Corresponding Author: Rony Berbara, MD, VA Ann Arbor Healthcare System, 2215 Fuller Rd, Ann Arbor, Michigan 48105 ([email protected]).
Financial Disclosure: None.
Funding/Support: None.
Previous Presentation: Presented in poster form at the AASM Sleep National Meeting; June 9, 2025; Seattle, Washington.
Acknowledgments: The authors thank staff from the Geriatric Research Education and Clinical Center (GRECC) for helping formulate and perform the statistical analysis section of the manuscript.
Supplementary Material: Available at Psychiatrist.com.
Clinical Points
- Higher dosages of mirtazapine are primarily prescribed to patients with depression and insomnia.
- Mirtazapine is a strong H1 receptor antagonist, providing sedative and hypnotic effects.
- Ultra–low-dose mirtazapine may be used as monotherapy for insomnia and can be an alternative in patients for whom cognitive-behavioral therapy for insomnia is contraindicated.
References (20)

- American Academy of Sleep Medicine. The International Classification of Sleep Disorders, Diagnostic and Coding Manual. 3 ed. American Academy of Sleep Medicine; 2023.
- American Psychiatric Publishing. Diagnostic and Statistical Manual of Mental Disorders: DSM-5, 5th ed, text rev. American Psychiatric Publishing, Inc; 2022.
- Trauer JM, Qian MY, Doyle JS, et al. Cognitive behavioral therapy for chronic insomnia: a systematic review and meta-analysis. Ann Intern Med. 2015;163(3):191–204. PubMed CrossRef
- Smith MT, Perlis ML. Who is a candidate for cognitive-behavioral therapy for insomnia? Health Psychol. 2006;25(1):15–19. PubMed CrossRef
- Savarese M, Carnicelli M, Cardinali V, et al. Subjective hypnotic efficacy of Trazodone and Mirtazapine in patients with chronic insomnia: a retrospective, comparative study. Arch Ital Biol. 2015;153(2-3):231–238. PubMed CrossRef
- Cipriani A, Furukawa TA, Salanti G, et al. Comparative efficacy and acceptability of 12 new-generation antidepressants: a multiple-treatments meta-analysis. Lancet. 2009;373(9665):746–758. PubMed CrossRef
- Furukawa TA, Cipriani A, Cowen PJ, et al. Optimal dose of selective serotonin reuptake inhibitors, venlafaxine, and mirtazapine in major depression: a systematic review and dose-response meta-analysis. Lancet Psychiatry. 2019;6(7):601–609. PubMed CrossRef
- Karsten J, Hagenauw LA, Kamphuis J, et al. Low doses of mirtazapine or quetiapine for transient insomnia: a randomised, double-blind, cross-over, placebo-controlled trial. J Psychopharmacol. 2017;31(3):327–337. PubMed CrossRef
- Aslan S, Isik E, Cosar B. The effects of mirtazapine on sleep: a placebo controlled, double-blind study in young healthy volunteers. Sleep. 2002;25(6):666–668. PubMed CrossRef
- Morin CM, Belleville G, Bélanger L, et al. The Insomnia Severity Index: psychometric indicators to detect insomnia cases and evaluate treatment response. Sleep. 2011;34(5):601–608. PubMed CrossRef
- Bastien CH, Vallières A, Morin CM. Validation of the Insomnia Severity Index as an outcome measure for insomnia research. Sleep Med. 2001;2(4):297–307. PubMed CrossRef
- Ivgy-May N, Hajak G, van Osta G, et al. Efficacy and safety of esmirtazapine in adult outpatients with chronic primary insomnia: a randomized, double-blind placebo-controlled study andopen-label extension. J Clin Sleep Med. 2020;16(9):1455–1467. PubMed CrossRef
- Ivgy-May N, Roth T, Ruwe F, et al. Esmirtazapine in non-elderly adult patients with primary insomnia: efficacy and safety from a 2-week randomized outpatient trial. Sleep Med. 2015;16(7):831–837. PubMed CrossRef
- Ivgy-May N, Ruwe F, Krystal A, et al. Esmirtazapine in non-elderly adult patients with primary insomnia: efficacy and safety from a randomized, 6-week sleep laboratory trial. Sleep Med. 2015;16(7):838–844. PubMed CrossRef
- Ruwe F, P IJ-B, Roth T, et al. A phase 2 randomized dose-finding study with esmirtazapine in patients with primary insomnia. J Clin Psychopharmacol. 2016;36(5):457–464. PubMed CrossRef
- Everitt H, Baldwin DS, Stuart B, et al. Antidepressants for insomnia in adults. Cochrane Database Syst Rev. 2018;5(5):CD010753. PubMed CrossRef
- Sateia MJ, Buysse DJ, Krystal AD, et al. Clinical Practice Guideline for the Pharmacologic Treatment of Chronic Insomnia in Adults: an American Academy of Sleep Medicine Clinical Practice Guideline. J Clin Sleep Med. 2017;13(2):307–349. PubMed CrossRef
- Aslan S, Isik E, Cosar B. The effects of mirtazapine on sleep: a placebo controlled, double-blind study in young healthy volunteers. Sleep. 2002;25(6):677–679. PubMed CrossRef
- Nedergaard M, Goldman SA. Glymphatic failure as a final common pathway to dementia. Science. 2020;370(6512):50–56. PubMed CrossRef
- Longo LP, Johnson B. Addiction: Part I. Benzodiazepines-side effects, abuse risk and alternatives. Am Fam Physician. 2000;61(7):2121–2128. PubMed
Enjoy this premium PDF as part of your membership benefits!