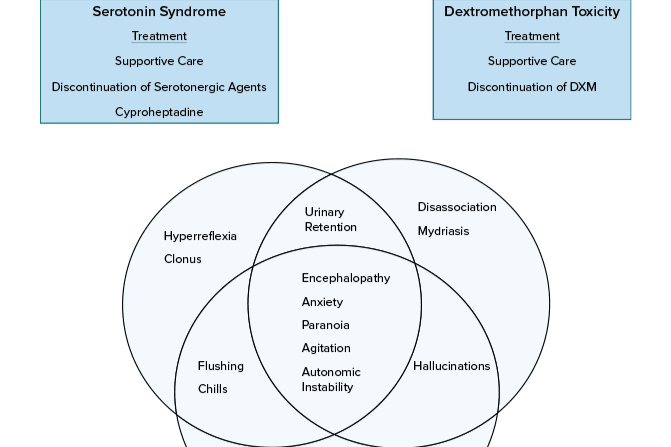
Because this piece does not have an abstract, we have provided for your benefit the first 3 sentences of the full text.
There have been several reports regarding the development of gynecomastia in patients on selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors, but to our knowledge, this is the first reported case in a patient taking citalopram. Our literature search included PubMed and Ovid databases and the keywords SSRI, citalopram, gynecomastia, and hyperprolactinemia. The exact mechanisms are still unclear, but SSRI use has been associated with hyperprolactinemia and perturbations of the hypothalamic-pituitary-testicular (HPT) axis.

Gynecomastia Associated With Citalopram
There have been several reports1-3 regarding the development of gynecomastia in patients on selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors, but to our knowledge, this is the first reported case in a patient taking citalopram. Our literature search included PubMed and Ovid databases and the keywords SSRI, citalopram, gynecomastia, and hyperprolactinemia. The exact mechanisms are still unclear, but SSRI use has been associated with hyperprolactinemia4,5 and perturbations of the hypothalamic-pituitary-testicular (HPT) axis.6
Case Report
A 44-year-old man with a history of hypertension, alcohol use disorder, and generalized anxiety disorder (GAD) was referred to the psychiatry service for management of his GAD due to suspected citalopram-induced gynecomastia. In September 2017, the patient presented increasing anxiety during his hospitalization for treatment of alcohol-induced pancreatitis—after which he was discharged home with a prescription for hydroxyzine. At a follow-up appointment with his primary care provider, he was started on citalopram 10 mg daily, which was initially tolerated well and thus the dosage was optimized to 20 mg daily.
After 5 months of treatment with citalopram 20 mg, he presented in February 2018 to his primary care provider complaining of right nipple pain with a palpable mass, decreased libido, and erectile dysfunction. There was no report of any testicular symptoms, prior sexual dysfunction, or family history of breast cancer. The patient discontinued alcohol use after the hospitalization for pancreatitis, and his medication regimen consisted only of citalopram 20 mg daily, which had been unchanged for several months. Examination revealed several 1-cm mobile, palpable masses in both breasts but no other abnormalities.
Workup consisted of thyroid-stimulating hormone level, liver function tests, and testosterone level, which were all within normal limits, and mammogram, which showed bilateral gynecomastia with no findings concerning for a malignancy. At the initial psychiatry appointment, citalopram was discontinued and escitalopram was started at 10 mg daily due to it being the only SSRI without gynecomastia listed as a side effect in its package insert. At the 1-month follow-up, he reported that his gynecomastia resolved, breast pain decreased, and libido increased.
Discussion
Although it is not exactly clear how gynecomastia is related to SSRI use, there are several proposed theories that implicate follicle-stimulating hormone (FSH), luteinizing hormone (LH), estrogen, testosterone, and prolactin. Estrogen has been credited with stimulating development of ductal tissue, and progesterone has been shown to be responsible for stimulating alveolar development, while testosterone plays an antagonistic role to both estrogen and progesterone, and gynecomastia has been correlated to an increase in the estrogen-to-testosterone ratio.7-9 A study by Safarinejad6 evaluated the effect of SSRIs on the HPT axis in depressed adult males and found that subjects taking SSRIs exhibited lower levels of LH, FSH, and testosterone but had no significant difference in estradiol levels. They found prolactin levels were elevated in patients taking SSRIs, and those reporting sexual side effects had a greater degree of hormonal derangements.6 Multiple studies have demonstrated the association between SSRIs and hyperprolactinemia, which has been linked to gynecomastia and other sexual dysfunction symptoms.3,4,10
Thus, we might theorize that our patient developed gynecomastia and sexual dysfunction from citalopram from a combination HPT axis disruption, increase in estrogen-to-testosterone ratio, and elevation in prolactin. A confounder to consider is that the patient also had concurrent alcohol use disorder. It is unclear why the patient developed gynecomastia with citalopram use but not with escitalopram and thus warrants further investigation. The correlation can be quantified using the Naranjo Adverse Drug Reaction Probability Scale,11 which gives a score of 4 (notably losing a point due to his alcohol use history alone being a potential cause of the symptoms) and thus classified gynecomastia as a possible adverse drug reaction. Ultimately, the development of gynecomastia in relation to SSRI use is multifactorial; however, the side effects improved when citalopram was discontinued and escitalopram was started in its place.
Published online: December 12, 2019.
Potential conflicts of interest: None.
Funding/support: None.
Patient consent: Consent was received from the patient to publish the case report, and information has been de-identified to protect anonymity.
REFERENCES
1.Nuttall FQ, Warrier RS, Gannon MC. Gynecomastia and drugs: a critical evaluation of the literature. Eur J Clin Pharmacol. 2015;71(5):569-578. PubMed CrossRef
2.Karakurt F, Kargili A, Uz B, et al. Venlafaxine-induced gynecomastia in a young patient: a case report. Clin Neuropharmacol. 2009;32(1):51-52. PubMed
3.Kaufman KR, Podolsky D, Greenman D, et al. Antidepressant-selective gynecomastia. Ann Pharmacother. 2013;47(1):e6. PubMed CrossRef
4.Kim S, Park Y-M. Serum prolactin and macroprolactin levels among outpatients with major depressive disorder following the administration of selective serotonin-reuptake inhibitors: a cross-sectional pilot study. PLoS One. 2013;8(12):e82749. PubMed CrossRef
5.Reeves KW, Okereke OI, Qian J, et al. Antidepressant use and circulating prolactin levels. Cancer Causes Control. 2016;27(7):853-861. PubMed CrossRef
6.Safarinejad MR. Evaluation of endocrine profile and hypothalamic-pituitary-testis axis in selective serotonin reuptake inhibitor-induced male sexual dysfunction. J Clin Psychopharmacol. 2008;28(4):418-423. PubMed CrossRef
7.Johnson RE, Murad MH. Gynecomastia: pathophysiology, evaluation, and management. Mayo Clin Proc. 2009;84(11):1010-1015. PubMed CrossRef
8.Ladizinski B, Lee KC, Nutan FN, et al. Gynecomastia: etiologies, clinical presentations, diagnosis, and management. South Med J. 2014;107(1):44-49. PubMed CrossRef
9.Niewoehner CB, Schorer AE. Gynaecomastia and breast cancer in men. BMJ. 2008;336(7646):709-713. PubMed CrossRef
10.Torre DL, Falorni A. Pharmacological causes of hyperprolactinemia. Ther Clin Risk Manag. 2007;3(5):929-951. PubMed
11.Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30(2):239-245. PubMed CrossRef
aCollege of Osteopathic Medicine, Marian University, Indianapolis, Indiana
bGallahue Mental Health Services, Community Health Network, Indianapolis, Indiana
*Corresponding author: Peter G. Karalis, MD, Gallahue Mental Health Services, Community Health Network, 7150 Clearvista Dr, Indianapolis, IN 46256 ([email protected]).
Prim Care Companion CNS Disord 2019;21(6):19l02456
To cite: Ji Ok S, Karalis PG, Daas M, et al. Gynecomastia associated with citalopram. Prim Care Companion CNS Disord. 2019;21(6):19l02456.
To share: https://doi.org/10.4088/PCC.19l02456
© Copyright 2019 Physicians Postgraduate Press, Inc.
Enjoy this premium PDF as part of your membership benefits!