Pulmonary embolism is a potentially fatal condition that requires rapid diagnosis through clinical scoring, biomarkers, and radiological evaluation. D-dimer testing typically has a high negative predictive value in ruling out thromboembolism; however, false-negative results may occur, particularly with early or subsegmental emboli.1,2 Clozapine is a second-generation antipsychotic widely used in treatment-resistant schizophrenia and schizoaffective disorder. Its adverse effects include agranulocytosis, myocarditis, metabolic disturbances, and thromboembolic complications. Clozapine-associated pulmonary embolism is rare, dose independent, and often early in onset.3 Although the mechanistic pathways remain incompletely understood, published case reports and pharmacovigilance analyses continue to play an essential role in characterizing risk patterns.2,4 This report describes an unusual and diagnostically challenging presentation of a patient who developed pulmonary embolism while receiving clozapine, despite negative D-dimer levels and low clinical probability scores.
Case Report
A 48-year-old woman with schizoaffective disorder was admitted for evaluation for electroconvulsive therapy (ECT). She had previously undergone ECT with a partial response. For the preceding 3 months, she had been receiving clozapine. On admission, her psychiatric symptoms included a fixed delusion of pregnancy despite postmenopausal status and repeatedly negative β-hCG tests. No mood episode or other psychotic features were present.
Her medications included clozapine 450 mg/day, valproate 1,250 mg/day, and aripiprazole 30 mg/day. Ten days after admission, aripiprazole was switched to the long-acting injection to improve adherence, and clozapine was increased to 500 mg/day due to persistent delusional content.
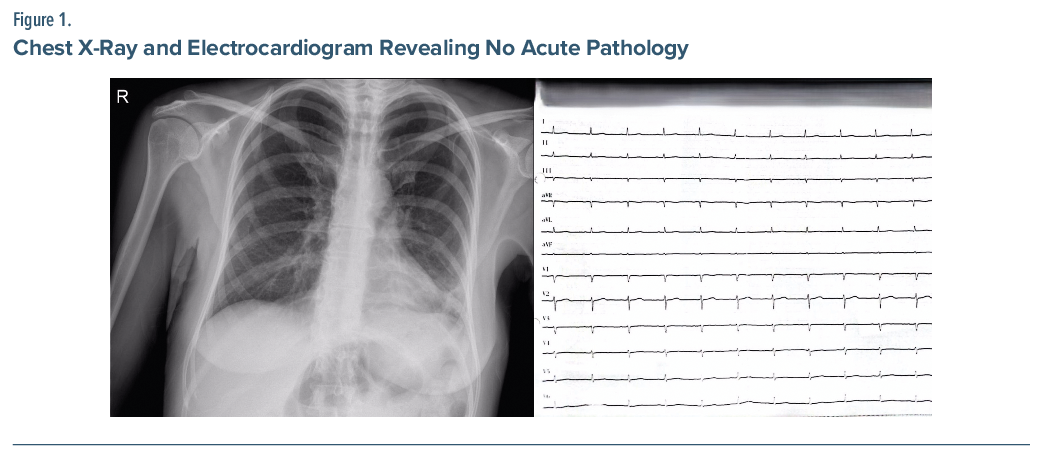
On hospital day 18, she developed sudden dyspnea and chest pain. Vital signs showed blood pressure of 90/60 mm Hg, oxygen saturation of 90%, heart rate of 78 bpm, and normal temperature. Electrocardiogram and chest X-ray revealed no acute pathology (Figure 1).
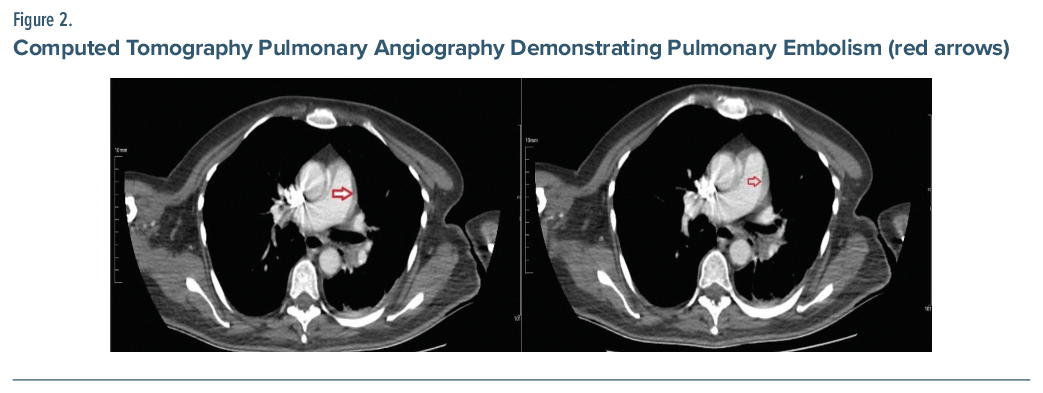
Laboratory results included white blood cell count: 7.83 × 103/µL, C-reactive protein: 71.5 mg/L, and D-dimer: 285.1 μg/mL (reference range, <500 μg/mL). Her Wells score was 3, and the revised-Geneva score was 0, both indicating low clinical probability.5,6 Despite this, because of persistent symptoms, computed tomography pulmonary angiography was performed and demonstrated pulmonary embolism (Figure 2).
She was transferred to the intensive care unit for thrombolytic therapy. Clozapine was discontinued due to suspected adverse drug reaction. The Naranjo score was calculated as 6, suggesting a “probable” relationship between clozapine and the pulmonary embolism.7
After stabilization, she returned to the psychiatry ward on rivaroxaban. A new antipsychotic regimen consisting of haloperidol 20 mg/day, valproate 1,000 mg/day, quetiapine 100 mg/day, and aripiprazole long-acting injectable was initiated. Her delusion gradually improved, and she was discharged.
Discussion
Pulmonary embolism is an uncommon but serious complication associated with clozapine therapy, with several epidemiologic and pharmacovigilance studies identifying clozapine among the antipsychotics most strongly linked to venous thromboembolism.1˒2 Although D-dimer testing generally has a strong negative predictive value, false-negative results can occur, particularly in patients with small or subsegmental emboli.3 A study reported that 9 of 1,270 patients with negative D-dimer levels nonetheless had imaging-confirmed pulmonary embolism, supporting the importance of clinical vigilance when respiratory symptoms persist.4
Several mechanisms may contribute to clozapine-associated pulmonary embolism, including sedation-related immobility, metabolic disturbances, platelet activation, and procoagulant effects.2,8 In the present case, the temporal association between clozapine dose escalation and symptom onset, together with a Naranjo score of 6, supports a probable drug-related causality.9 Similar reports have emphasized the importance of early recognition of severe clozapine-related adverse reactions, including cardiovascular and hematologic complications.
Diagnostic evaluation in psychiatric settings can be challenging, as symptoms such as dyspnea, chest discomfort, and anxiety may be misattributed to psychiatric or medication-related causes. Despite the low Wells and revised-Geneva scores in this patient, pulmonary embolism was radiologically confirmed, demonstrating that clinical prediction tools may underestimate risk during clozapine therapy. This reinforces the need for a low threshold for imaging when cardiopulmonary symptoms arise in clozapine-treated individuals.
Management following clozapine-associated pulmonary embolism is complex. Large registry data suggest that aripiprazole long-acting injectable and oral olanzapine may have lower risks of treatment failure after clozapine discontinuation.9 Conversely, rechallenging clozapine is generally discouraged following life-threatening adverse reactions such as myocarditis or thromboembolism.10 In this case, continuing aripiprazole long-acting injectable and adding haloperidol provided stable symptom control without reexposure to clozapine.
This case highlights that pulmonary embolism can occur during clozapine treatment even in the absence of conventional risk factors or abnormal laboratory indicators. Clinicians should remain attentive to new or unexplained cardiopulmonary symptoms and consider prompt imaging when necessary.
Article Information
Published Online: May 5, 2026. https://doi.org/10.4088/PCC.25cr04155
© 2026 Physicians Postgraduate Press, Inc.
Prim Care Companion CNS Disord 2026;28(3):25cr04155
Submitted: November 28, 2025; accepted January 27, 2026.
To Cite: Karatas KS, Yilmaz C. Acute pulmonary embolism with negative D-dimer level in a schizoaffective patient during the course of clozapine use. Prim Care Companion CNS Disord 2026;28(3):25cr04155.
Author Affiliations: Department of Psychiatry, School of Medicine, Kutahya Health Sciences University, Kutahya, Turkey (Karatas); Sligo Leitrim Mental Health Service, Sligo, Ireland (Yilmaz).
Corresponding Author: Kader Semra Karatas, MD, Department of Psychiatry, School of Medicine, Kutahya Health Sciences University, Kutahya, Turkey ([email protected]).
Financial Disclosure: None.
Funding/Support: None.
Patient Consent: Consent was received from the patient to publish the case report and images. Information has been de-identified to protect anonymity.
ORCID: Kader Semra Karatas: https://orcid.org/0000-0003-3595-8019; Ceyhun Yilmaz: https://orcid.org/0000-0003-2184-5881
References (10)

- Bachmann CJ, Aagaard L, Bernardo M, et al. International trends in clozapine use: a study in 17 countries. Acta Psychiatr Scand. 2017;136(1):37–51. PubMed CrossRef
- Hägg S, Jönsson AK, Spigset O. Antipsychotic-induced venous thromboembolism: a review of the evidence. CNS Drugs. 2018;32(1):47–64. PubMed
- Glober N, Nguyen M, Nguyen A, et al. False-negative D-dimer results in patients with pulmonary embolism. Am J Emerg Med. 2018;36(9):1746–1751.
- Luykx JJ, Stam AH, Tanskanen A, et al. Antipsychotic effectiveness and safety after clozapine discontinuation: a real-world study. Schizophr Bull. 2020;46(4):1008–1017.
- Wells PS, Anderson DR, Rodger M, et al. Derivation of a simple clinical model to categorize patients probability of pulmonary embolism: increasing the models utility with the SimpliRED D-dimer. Thromb Haemost. 2000;83(3):416–420. PubMed
- Le Gal G, Righini M, Roy PM, et al. Prediction of pulmonary embolism in the emergency department: the revised Geneva score. Ann Intern Med. 2006;144(3):165–171. PubMed CrossRef
- Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30(2):239–245. PubMed CrossRef
- De Leon J, Ruan C-J, Schoretsanitis G, et al. A rational use of clozapine based on adverse drug reactions, pharmacokinetics, and clinical pharmacopsychology. Psychother Psychosom. 2020;89(4):200–214. PubMed CrossRef
- Ronaldson KJ, Fitzgerald PB, Taylor AJ, et al. Rapid clozapine titration and lung function changes associated with myocarditis. J Clin Psychiatry. 2012;73(6):880–886.
- Masood B, Hasan A, Wadoo O. Clozapine-induced pulmonary embolism: case report and literature review. Case Rep Psychiatry. 2017;2017:1–4.
Enjoy this premium PDF as part of your membership benefits!