Abstract
Background: To compare the severity of alcohol craving between euthymic patients with unipolar and bipolar disorders and comorbid alcohol use disorder (AUD) and to explore associations between craving severity and clinical and biological markers, including carbohydrate-deficient transferrin (CDT) and homocysteine, to better characterize diagnostic-specific patterns of dual diagnosis.
Methods: This cross-sectional study assessed 50 euthymic outpatients with comorbid AUD and either major depressive disorder (MDD) or bipolar disorder. Participants were evaluated using the Alcohol Use Disorder Identification Test (AUDIT), Visual Analog Scale (VAS) for craving, and blood biomarkers including homocysteine and CDT. Group differences and correlations between craving severity and clinical/biological variables were analyzed. Participants were consecutively recruited over a 12-month period, from June 2015 to May 2016.
Results: Patients with AUD and MDD reported significantly higher alcohol craving (VAS), AUDIT scores, and CDT levels than those with AUD and bipolar disorder. In both groups, alcohol craving positively correlated with AUDIT scores, CDT levels, and plasma homocysteine concentrations. No significant associations were found with sociodemographic variables.
Conclusions: Patients with AUD and MDD may represent a more severe clinical subtype of dual diagnosis. Elevated homocysteine may serve as a potential biomarker for craving intensity and mood instability. These findings support the need for integrated, personalized treatment approaches in dual-diagnosis populations.
Prim Care Companion CNS Disord 2026;28(2):25m04123
Author affiliations are listed at the end of this article.
A growing body of evidence indicates a strong and bidirectional association between mood disorders and alcohol use disorder (AUD), with individuals affected by one condition being at substantially increased risk of developing the other.1 This comorbidity, commonly referred to as dual diagnosis, represents a major public health concern, as both mood disorders and AUD are among the most prevalent and disabling psychiatric conditions worldwide. Epidemiologic studies consistently report high rates of co-occurrence, particularly in major depressive disorder (MDD) and bipolar disorder (BD), where AUD is associated with greater clinical severity, poorer outcomes, and increased relapse risk.2,3 Despite its clinical relevance, dual diagnosis remains challenging to manage.4 Patients with comorbid AUD and mood disorders tend to present with complex clinical profiles, a higher prevalence of additional psychiatric comorbidities, reduced treatment response, more frequent hospitalizations, and an increased risk of suicidal behavior.3 These difficulties are further compounded by fragmented treatment systems and the limited availability of objective markers to support diagnosis, risk stratification, and personalized interventions.5–8 As a result, many patients with dual diagnoses are either misdiagnosed or diagnosed too late, which inevitably leads to worsening of symptoms, reduced treatment efficacy, and serious repercussions for overall health, functioning, and quality of life.3,9,10 Emerging evidence suggests that the clinical expression of AUD may differ between MDD and BD. Patients with BD and comorbid AUD often show earlier onset of alcohol use and more severe longitudinal courses, whereas depressive symptomatology appears to be particularly closely linked to alcohol craving, a key predictor of relapse.11–17 Importantly, craving may persist even during euthymic phases, suggesting an underlying vulnerability that is not fully captured by mood symptom severity alone.18 In this context, the identification of biological correlates of alcohol use and craving is of particular interest. Carbohydrate-deficient transferrin (CDT) is a well-established biomarker of chronic alcohol consumption,19 while homocysteine has been implicated in both mood disorders and alcohol-related neurobiological dysfunction.20 However, few studies have examined these biomarkers in relation to alcohol craving across different mood disorder diagnoses, and direct comparisons between euthymic patients with MDD and BD are lacking. The present cross-sectional study aimed primarily to compare the severity of alcohol craving between euthymic patients with unipolar and bipolar disorders and comorbid AUD. A secondary aim was to explore associations between craving severity and clinical and biological markers, including CDT and homocysteine, to better characterize diagnostic-specific patterns of dual diagnosis.
METHODS
Study Design and Overview
This was a prospective, observational, cross-sectional study conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. All participants provided written informed consent prior to inclusion in the study. The study protocol was approved by the Local Ethics Committee (approval code: ASLCN2/SerD1).
Participants were recruited at the Addiction Treatment Unit of Alba, Italy, a public outpatient service providing specialized assessment and treatment for substance use disorders and psychiatric comorbidities.
Participants were consecutively recruited over a 12-month period, from June 2015 to May 2016. During this period, all patients presenting to the clinic who met the eligibility criteria were invited to participate. A total of 57 individuals were approached, of whom 7 (5 males and 2 females) declined participation. No additional limitations related to staffing or service availability influenced enrollment. Inclusion criteria were (1) diagnosis of AUD comorbid with MDD or BD type I or II (BD I/BD II), according to Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition [DSM-IV] criteria; (2) clinical condition of mood euthymia at the time of enrollment; (3) absence of additional substance use disorders, with the exception of nicotine, according to DSM-IV criteria; (4) absence of other severe psychiatric, neurological, or relevant medical conditions, including renal, cardiac, or hepatic insufficiency, decompensated hypo-or hyperthyroidism, or chronic cerebrovascular disease; and (5) age 18 years or older. Psychiatric diagnoses were confirmed using the Mini-International Neuropsychiatric Interview Plus,21 a structured diagnostic interview widely used in clinical research. Mood euthymia was determined at the time of enrollment using 2 clinician-administered rating scales: the Montgomery-Asberg Depression Rating Scale,22 with a cutoff score of ≤4 to exclude residual depressive symptoms, and the Young Mania Rating Scale,23 with a score of <12 to exclude subthreshold manic symptoms. Given the exploratory nature of the study and the limited availability of eligible patients within the recruitment period, no a priori sample size calculation was performed.
Data Collection and Instruments
Hazardous and harmful alcohol use was assessed using the Alcohol Use Disorder Identification Test (AUDIT).24 Alcohol craving severity was evaluated using the Visual Analog Scale (VAS),25 a self-report measure assessing the intensity of craving at the time of evaluation. Venous blood samples were collected during the initial clinical visit to measure plasma homocysteine and CDT concentrations. Homocysteine was included due to its reported association with mood disorders and alcohol-related neurobiological alterations,26,27 while CDT was used as an objective biomarker of chronic alcohol consumption.28
Statistical Analysis
All statistical analyses were performed using the online platform Social Science Statistics (http://www.socscistatistics.com/). Continuous variables are reported as mean±SD, and categorical variables as percentage. Group differences between unipolar and bipolar patients were analyzed using independent-samples t-tests for normally distributed variables and Mann-Whitney U tests for non-normally distributed variables. Normality was assessed using the Kolmogorov-Smirnov test. Associations between alcohol craving severity and clinical or biological variables (AUDIT scores, plasma homocysteine levels, CDT values, and sociodemographic characteristics) were examined using Pearson correlation coefficient or point-biserial correlation for normally distributed data and Kendall τ-b for non-normally distributed data. Statistical significance was set at P<.05 for all analyses.
RESULTS
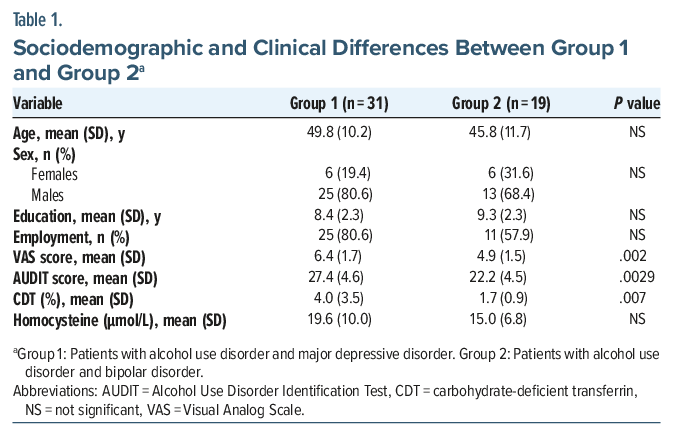
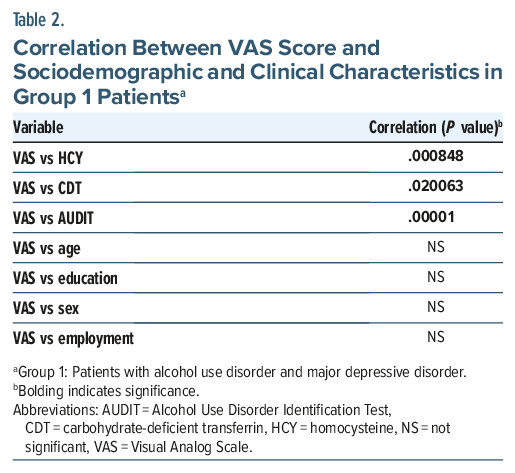
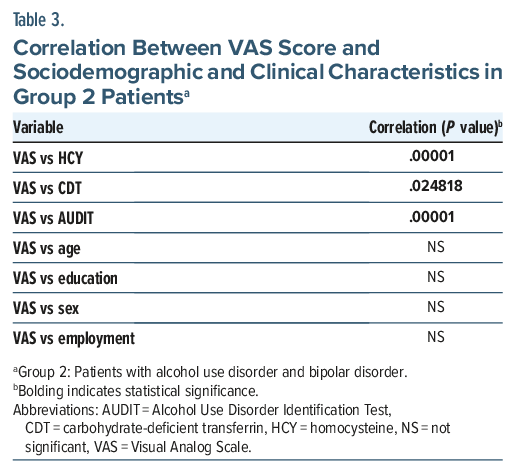
A total of 50 patients—12 females and 38 males—with a mean age of 48.28 years (SD=10.86) were enrolled in the study. Participants were divided into 2 groups. Group 1 included 31 patients (6 females and 25 males; mean age: 49.8 years, SD=10.2) diagnosed with AUD comorbid with MDD. Group 2 included 19 patients (6 females and 13 males; mean age: 45.8 years, SD =11.7) diagnosed with AUD comorbid with BD (5 BD I and 14 BD II). Sociodemographic and clinical characteristics are reported in Table 1. Analysis of group differences in sociodemographic and clinical characteristics revealed that patients in group 1 had significantly higher VAS and AUDIT scores, as well as a greater percentage of plasma CDT, compared to group 2. No significant group differences were found in sociodemographic characteristics or in plasma homocysteine concentrations (Table 1). Finally, we analyzed the correlations between VAS scores and sociodemographic and clinical characteristics in both groups. In group 1 (Table 2), VAS scores were positively correlated with AUDIT scores (P<.001), plasma homocysteine concentrations (P=.000848), and CDT percentage (P =.020063). No significant correlations were found between VAS scores and age, gender, education level, or employment status. Similarly, in group 2 (Table 3), VAS scores were positively correlated with AUDIT scores (P<.001), plasma homocysteine concentrations (P< .001), and CDT percentage (P =.024818). No significant correlations were found between VAS scores and age, gender, education level, or employment status.
DISCUSSION
This study provides evidence that euthymic patients with comorbid MDD and AUD exhibit significantly higher alcohol craving severity compared with euthymic patients with comorbid BD and AUD. Moreover, alcohol craving showed a strong association with the alcohol consumption markers examined in the present study—specifically AUDIT scores and CDT levels—as well as with plasma homocysteine concentrations, consistently across diagnostic groups. The finding of greater craving severity in patients with MDD, despite clinical euthymia, extends previous literature linking depressive symptomatology to alcohol craving and relapse risk.11–17,29,30 Importantly, our results suggest that vulnerability to craving in MDD may persist beyond acute mood episodes, pointing to a more enduring psychopathological or neurobiological link between MDD and AUD compared with BD. This diagnostic-specific difference has rarely been explored in clinically stable populations and adds novel insight to the field. Consistent with prior studies, CDT showed a robust association with both alcohol consumption and craving severity,31,32 supporting its utility as an objective marker of clinically relevant alcohol use in patients with dual diagnosis. The integration of CDT with subjective craving measures may improve the identification of individuals at higher risk for continued drinking and relapse, even during euthymic phases. In addition, elevated homocysteine levels were significantly associated with craving severity. Homocysteine has been implicated in affective disorders and alcohol dependence through mechanisms involving oxidative stress, glutamatergic excitotoxicity, monoaminergic dysfunction, and epigenetics.20,26,27,33–42 Its association with craving in the present study suggests that homocysteine may reflect a shared biological vulnerability underlying both mood dysregulation and addictive behaviors. From a clinical perspective, these findings highlight the value of combining clinical and biological measures to better characterize heterogeneity within dual diagnosis. Patients with elevated craving, CDT, and homocysteine levels may represent a subgroup with increased neurobiological vulnerability and potentially poorer outcomes, warranting closer monitoring and more intensive, integrated treatment strategies. Overall, this study underscores the importance of considering diagnostic-specific features and biological correlates in patients with comorbid AUD and mood disorders. Future studies with larger, multicenter, and longitudinal designs are needed to confirm these findings and to further clarify their implications for personalized treatment approaches.
Limitations
This study has several important limitations that should be taken into account when interpreting the findings. First, the overall sample size was relatively small, which may limit the statistical power and reduce the ability to detect more subtle associations or group differences. A limited sample also increases the risk of type II errors and may compromise the robustness and reproducibility of the results. In addition, the distribution of patients was unequal between group 1 (patients with MDD) and group 2 (patients with BD), which should be considered when interpreting between-group comparisons. Second, the number of female participants in the sample was notably low, which constrains the possibility of conducting meaningful gender-stratified analyses. Given the evidence suggesting that biological sex and gender-related psychosocial factors can influence the clinical presentation, neurobiology, and treatment outcomes of both mood disorders and substance use disorders, this is a significant limitation that warrants further investigation in more balanced samples. Third, the study was conducted at a single outpatient addiction treatment center in Alba, Italy, which may restrict the generalizability of the findings to broader clinical populations. Patients referred to this specific center may differ from those in other geographical regions or health care systems in terms of sociodemographic characteristics, clinical severity, comorbidities, and access to care. Consequently, the external validity of the study may be limited. Fourth, the group of patients with BD was heterogeneous, comprising both bipolar I and bipolar II disorder. These 2 variants may exhibit distinct characteristics in relation to the study objectives; however, the number of patients with each subtype was too small to permit a 3-arm study design. In addition, the cross-sectional design precludes any conclusions about causality or the temporal direction of the observed associations. Future research should aim to address these limitations by including larger, more diverse, and multicenter samples, with longitudinal designs that allow for more comprehensive assessment of causal relationships and trajectories over time.
CONCLUSION
This study supports the close association between mood disorders and AUD, showing that alcohol craving and intoxication severity are correlated with plasma homocysteine levels in patients with dual diagnosis. Patients with comorbid MDD exhibited higher craving intensity and alcohol-related severity compared with those with BD, despite being in a euthymic phase, suggesting a persistent vulnerability to alcohol-related behaviors beyond mood episodes. These findings highlight the clinical relevance of accurately characterizing mood disorder subtypes in patients with AUD and suggest that plasma homocysteine may represent a promising peripheral biomarker for identifying more severe dual-diagnosis profiles. Future studies with larger, multicenter, and longitudinal designs are warranted to confirm these results, clarify underlying mechanisms, and evaluate the potential utility of biomarker-informed and personalized treatment strategies in this population.
Article Information
Published Online: April 21, 2026. https://doi.org/10.4088/PCC.25m04123
© 2026 Physicians Postgraduate Press, Inc.
Submitted: October 18, 2025; accepted January 9, 2026.
To Cite: Coppola M, Mondola R. Differences in the severity of alcohol craving between euthymic unipolar and bipolar patients. Prim Care Companion CNS Disord 2026;28(2):25m04123.
Author Affiliations: Department of Addiction ASLCN1, Cuneo, Italy (Coppola); Department of Mental Health ASLCN1, Saluzzo (CN), Italy (Mondola).
Corresponding Author: Maurizio Coppola, MD, Department of Addiction ASLCN1, Cuneo, Italy ([email protected]).
Financial Disclosure: None.
Funding/Support: None.
Clinical Points
- Assess mood disorder subtype: Craving and alcohol-related severity differ between major depressive disorder and bipolar disorder even during euthymia, so accurate diagnosis is crucial.
- Use biomarkers to guide care: Plasma homocysteine and carbohydrate-deficient transferrin can help identify high-risk patients and support personalized, integrated treatment strategies.
References (42)

- Boden JM, Fergusson DM. Alcohol and depression. Addiction. 2011;106(5):906–914. PubMed CrossRef
- Castillo-Carniglia A, Keyes KM, Hasin DS, et al. Psychiatric comorbidities in alcohol use disorder. Lancet Psychiatry. 2019;6(12):1068–1080. PubMed CrossRef
- National Institute on Alcohol Abuse and Alcoholism (NIH). Mental Health Issues: Alcohol Use Disorder and Common Co-occurring Conditions. National Institute on Alcohol Abuse and Alcoholism (NIH); 2025. https://www.niaaa.nih.gov/health-professionals-communities/core-resource-on-alcohol/mental-health-issues-alcohol-use-disorder-and-common-co-occurring-conditions
- Woody G. The challenge of dual diagnosis. Alcohol Health Res World. 1996;20(2):76–80. PubMed
- Chetty A, Guse T, Malema M. Integrated vs non-integrated treatment outcomes in dual diagnosis disorders: a systematic review. Health SA. 2023;28:2094. PubMed CrossRef
- Salloum IM, Brown ES. Management of comorbid bipolar disorder and substance use disorders. Am J Drug Alcohol Abuse. 2017;43(4):366–376. PubMed CrossRef
- Scala JJ, Ganz AB, Snyder MP. Precision medicine approaches to Mental health care. Physiol (Bethesda). 2022;38:82–98. CrossRef
- Boness CL, Witkiewitz K. Precision Medicine in Alcohol Use Disorder: Mapping etiologic and maintenance mechanisms to mechanisms of behavior change to improve patients outcome. Exp Clin Psychopharmacol. 2022;31:769–779. PubMed
- Sinclair E, South K, Vencel M. DUAL DIAGNOSIS: Serious Mental Illness And Co-Occurring Substance Use Disorders. Treatment Advocacy Center. Office of Research & Public Affairs. 2021. https://www.tac.org/wp-content/uploads/2024/01/Dual-Diagnosis.pdf
- Bonner-Reid FT, Dar A, Zerin A, et al. Comprehensive review on association of bipolar disorder and substance abuse: dual diagnosis and treatment approaches. Cureus. 2025;19(17):e86363. PubMed CrossRef
- Grunze H, Schaefer M, Scherk h, et al. Comorbid bipolar and alcohol use disorder—a therapeutic challenge. Front Psychiatry. 2021;12:660432. PubMed CrossRef
- Xia Y, Ma D, Perich T, et al. Demographic and clinical differences between bipolar disorder patients with and without alcohol use disorders. Front Psychiatry. 2020;11:570574. PubMed CrossRef
- Martins JS, Fogelman N, Wemm S, et al. Alcohol craving and withdrawal at treatment entry prospectively predict alcohol use outcomes during outpatient treatment. Drug Alcohol Depend. 2022;231:109253. PubMed CrossRef
- Stillman MA, Sutclif J. Predictors of relapse in alcohol use disorder: identifying individuals most vulnerable torelapse. Addict Subst Abuse. 2020;1:3–8.
- Prisciandaro JJ, DeSantis SM, Chiuzan C, et al. Impact of depressive symptoms on future alcohol use in patients with Co-occurring bipolar disorder and alcohol dependence: a prospective analysis in an 8-Week Randomized Controlled Trial of Acamprosate. Alcohol Clin Exp Res. 2012;36:967–969. PubMed CrossRef
- Hong M, Ha TY, Lee S, et al. Clinical correlates of alcohol Use disorder in depressed patients with unipolar and bipolar disorder. Psychiatry Investig. 2019;16(12):926–932. PubMed CrossRef
- Ze-Yu L, Shih-Chun M, Ming-Chyi H. Relapse risk factors associated with cravings in patients with alcohol Use disorder. Taiwan J Psychiatry. 2025;39:111–117.
- Beerten-Duijkers JCLM, Vissers CTWM, Rinck M, et al. Inhibitory Control and craving in dual disorders and Recurrent substance Use. Preliminary findings. Front Psychiatry. 2021;12:569817. PubMed CrossRef
- Bortolotti F, De Paoli G, Tagliaro F. Carbohydrate-deficient transferrin (CDT) as a marker of alcohol abuse: a critical review of the literature 2001-2005. J Chromatogr B Analyt Technol Biomed Life Sci. 2006;841(1−2):96–109. PubMed CrossRef
- Osher Y, Sela BA, Levine J, et al. Elevated homocysteine levels in euthymic bipolar disorder patients showing functional deterioration. Bipolar Disord. 2004;6(1):82–86. PubMed CrossRef
- Sheehan D V, Lecrubier Y, Sheehan KH, et al The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. 1998;20:22–33. PubMed
- Montgomery SA, Asberg M. A New depression scale designed to be sensitive to change. Change. Br J Psychiatry. 1979;134:382–389. PubMed CrossRef
- Young RC, Biggs JT, Ziegler VE, et al. A rating scale for mania: reliability, validity and sensitivity. Br J Psychiatry. 1978;133:429–435. PubMed CrossRef
- Saunders JB, Aasland OG, ìBabor TF, et al. Development of the Alcohol Use Disorders Identification Test (AUDIT): WHO Collaborative Project on Early Detection of Persons with Harmful Alcohol Consumption-II. Addiction. 1993;88(6):791–804. PubMed CrossRef
- Drobes DJ, Thomas SE. Assessing craving for alcohol. Alcohol Res Health. 1999;23(3):179–186. PubMed
- Cavalcante da Silva V, Chiaratti de Oliveira A, D’Almeida V. Homocysteine and psychiatric disorders. J Inborn Errors Metab Screen. 2017;5:1–8.
- Bleich S, Degner D, Sperling W, et al Homocysteine as a neurotoxin in chronic alcoholism. Prog Neuropsychopharmacol Biol Psychiatry. 2004;28:453-564. PubMed CrossRef
- Fiorelli D, Romani L, Treglia M, et al. Carbohydrate-deficient transferrin (CDT) as a biomarker of alcohol abuse: a retrospective study of the Italian drinking trend among drivers from 2016 to 2022. Toxics. 2023;11:914. PubMed CrossRef
- Miller ML, Cornelius JR, Kirisci L, et al. Depressive and Anxiety symptoms predict Obsessive and Compulsive cravings among depressed Alcoholics. Addict Disord Their Treat. 2020;19(4):228–233. PubMed CrossRef
- de Timary P, Cordovil de Sousa Uva M, Denoël C, et al. The associations between self-Consciousness, depressive state and craving to Drink among alcohol dependent patients Undergoing Protracted withdrawal. PloS One. 2013;8:e71560. PubMed CrossRef
- Harris JC, Leggio L, Farokhnia M. Blood biomarkers of alcohol use: a scoping review. Curr Addict Rep. 2021;8(4):500–508. PubMed CrossRef
- Saini RS, Pettinati HM, Semwanga AM, et al. Carbohydrate-deficient transferrin: an investigative biochemical marker of heavy alcohol consumption. Psychopharmacol Bull. 1997;33(1):171–175. PubMed
- Coppola M, Mondola R. Correlation between plasma homocysteine levels and craving in alcohol dependent stabilized patients. Clin Nutr. 2018;37(3):1061–1065. PubMed CrossRef
- Oliva F, Coppola M, Mondola R, et al. Blood homocysteine concentration and mood disorders with mixed features among patients with alcohol use disorder. BMC Psychiatry. 2017;17(1):181. PubMed CrossRef
- Kamat PK, Mallonee CJ, George AK, et al. Homocysteine, alcoholism and its potential epigenetic mechanism. Alcohol Clin Exp Res. 2016;40(12):2474–2481. PubMed CrossRef
- Bottiglieri T, Laundy M, Crellin R, et al. Homocysteine, folate, methylation, and monoamine metabolism in depression. J Neurol Neurosurg Psychiatry. 2000;69(2):228–232. PubMed CrossRef
- Kruman II, Culmsee C, Chan SC L, et al. Homocysteine elicits a DNA damage response in neurons that promotes apoptosis and hypersensitivity to excitotoxicity. J Neurosci. 2000;20(18):6920–6926. PubMed CrossRef
- Lipton SA, Kim WK, Choi YB, et al. Neurotoxicity associated with dual actions of homocysteine at the N-methyl-D-aspartate receptor. Proc Natl Acad Sci U S A. 1997;94(11):5923–5928. PubMed CrossRef
- Almohmadi NH, Al-kuraishy HM, Al-Gareeb AI, et al. Glutamatergic dysfunction in neurodegenerative diseases focusing on Parkinson’s disease: role of glutamate modulators. Brain Res Bull. 2025;225:111349. PubMed CrossRef
- Topal G, Brunet A, Millanvoye E, et al. Homocysteine induces oxidative stress by uncoupling of NO synthase activity through reduction of tetrahydrobiopterin. Free Radic Biol Med. 2004;36(12):1532–1541. PubMed CrossRef
- Peng Z, Jia Q, Mao J, et al. Neurotransmitters crosstalk and regulation in the reward circuit of subjects with behavioral addiction. Front Psychiatry. 2025;15:1439727. PubMed CrossRef
- Shao X, Zhu G. Associations among monoamine neurotransmitter pathways, personality traits, and major depressive disorder. Front Psychiatry. 2020;11:381. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!