Abstract
The Psychiatric Consultation Service at Massachusetts General Hospital sees medical and surgical inpatients with comorbid psychiatric symptoms and conditions. During their twice-weekly rounds, Dr Stern and other members of the Consultation Service discuss diagnosis and management of hospitalized patients with complex medical or surgical problems who also demonstrate psychiatric symptoms or conditions. These discussions have given rise to rounds reports that will prove useful for clinicians practicing at the interface of medicine and psychiatry.
Prim Care Companion CNS Disord 2026;28(3):25f04157
Author affiliations are listed at the end of this article.
Have you ever wondered why so many psychiatric and neuropsychiatric conditions share clinical manifestations? Have you puzzled over how to distinguish conditions with shared features, such as bipolar spectrum disorders (BSDs) and major depressive disorder (MDD)? Have you been uncertain about whether and how to manage similar symptoms found in different conditions? If you have, the following case vignette and discussion should prove useful.
CASE VIGNETTE
Mr B, a 21-year-old college senior, was brought to the college’s counseling center because of concerns about his well-being. Over the past week, he had been sleeping only 3 to 4 hours a night without feeling tired, talking rapidly in class, and dominating group projects with unrealistic business ideas; moreover, he had reached his credit card limit on purchases for these businesses. Faculty members were struck by his irritability.
When interviewed at the counseling center, Mr B was restless, his speech was pressured, and he had flight of ideas. He acknowledged feeling extraordinarily productive, and he talked with passion about his business ideas. He described an episode of depression during his freshman year that was marked by poor concentration, social withdrawal, and failing grades. These were initially managed by his primary care provider (PCP) with antidepressants, but the treatment response was suboptimal, and he had become agitated on 2 occasions.
One of Mr B’s paternal aunts had been hospitalized for “manic depression,” and his mother had been diagnosed as having MDD. He denied using drugs or alcohol. When Mr B screened positive on the Mood Disorder Questionnaire (MDQ), he was referred for a comprehensive diagnostic evaluation at the local psychiatric emergency department.
DISCUSSION
What Are the Signs and Symptoms of BSDs and MDD?
On average, 10 years pass before a correct diagnosis of a BSD is made,1 and one-third of patients remain misdiagnosed for 10 years2; these statistics underscore how difficult it can be to diagnose bipolar disorder. Making an accurate diagnosis is complicated given that individuals with BSDs often present for care during a depressive episode, but they have many more depressive episodes than hypomanic or manic episodes, and they spend much more time in a depressed state than in a hypomanic or manic episode.3,4 Therefore, there is a tendency to diagnose depressive illnesses. Moreover, the symptoms present during a depressive phase of a BSD are virtually indistinguishable from those seen during the depressive phase of MDD, thus having a history of a hypomanic or manic episode is critical for making the diagnosis.5 According to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, Text Revision (DSM-5-TR),6 hypomania is defined by having at least 4 consecutive days of abnormally and persistently elevated, expansive, or irritable mood as well as abnormally and persistently increased activity or energy, which is present most of the day, on nearly every day. Mania is defined by having at least 1 week of consecutive days (or less, if hospitalization has been needed) that is marked by abnormally and persistently elevated, expansive, or irritable mood as well as abnormally and persistently goal-directed behavior or energy that is present most of the day, on nearly every day. During the phase with a mood disturbance and increased energy or activity in hypomania/mania, 3 (or more) of the following symptoms must be present to a significant degree (with 4 required if the mood is only irritable) with a noticeable change in the individual’s usual behavior: having an inflated self-esteem or grandiosity, a decreased need for sleep, being more talkative than usual or having pressured speech, flight of ideas or the subjective experience of racing thoughts, distractibility, an increase in goal-directed activity or psychomotor agitation, and being intensely involved in activities that have a high potential for painful consequences. In children and adolescents, BSD is notable for chronic irritability, mixed episodes, and rapid cycling as opposed to the clear episodic mood changes experienced by adults; this also has a very high rate of co-occurrence with attention-deficit/hyperactivity disorder (ADHD).7 In geriatric patients (those aged ≥ 65 years), bipolar disorder presents with predominantly depressive polarity, melancholic features, and pronounced cognitive impairments in verbal learning and memory, along with higher rates of medical comorbidities compared to younger adults.8
Who Develops BSDs, How Common Are They, and How Often Are They Seen in Primary Care Practices?
Within primary care practices, the prevalence rates of BSD are between 0.5% and 9.8%,9 depending on whether a structured psychiatric diagnostic interview or a screening questionnaire was used. Among those with depression in primary care settings, the prevalence of BSD is closer to 17%, and the use of structured clinical interviews has resulted in fewer missed cases than when screening questionnaires have been employed.10 In terms of risk factors for BSD, several studies have suggested that an earlier age at onset and a family history of BSD are associated with the diagnosis of BSD; however, there is no clear age cutoff, and having a family history is not as definitive.5 However, in those who present during a depressive episode and have a family history of BSD, this family history is a risk factor for BSD and suggests a need for psychoeducation about symptoms of hypomania/mania so that patients and family members can be on the lookout for the emergence of hypomanic/manic symptoms and provide closer follow-up after the initiation of antidepressants (to monitor for a switch in mood to hypomania/mania).
How Can You Screen For and Diagnose BSDs?
Once an individual or their family, friends, or teachers raise concerns about a person’s behavior, further assessment is prudent. Screening often aims to identify disturbances in affect, behavior, and cognition that can be investigated by a comprehensive clinical evaluation that includes creation of a broad differential diagnosis (eg, a mood disorder such as MDD, bipolar disorder, substance-induced mood disorder). Making a diagnosis of bipolar disorder can be challenging in primary care settings, especially when a person presents with a depressive episode. People rarely seek help from physicians when their mood is elevated; however, they frequently present for care when they are depressed, have low energy levels, or are impulsive. Clinicians should ask about lifetime mood episodes (eg, by asking about cycles of low and high energy, changes in sleep, and episodes of irritability or expansive mood). In addition, details of the family’s psychiatric history, especially bipolar disorder in first-degree relatives, are essential, as bipolar disorder is highly heritable. Equally important is the documentation of a prior antidepressant response, as unexpected, abrupt “activation,” worsening, or mixed features following antidepressant use can indicate an underlying BSD. Tracking functional fluctuations (eg, academic disruption, inconsistent work performance, or unstable relationships) can reveal patterns that point toward a BSD.
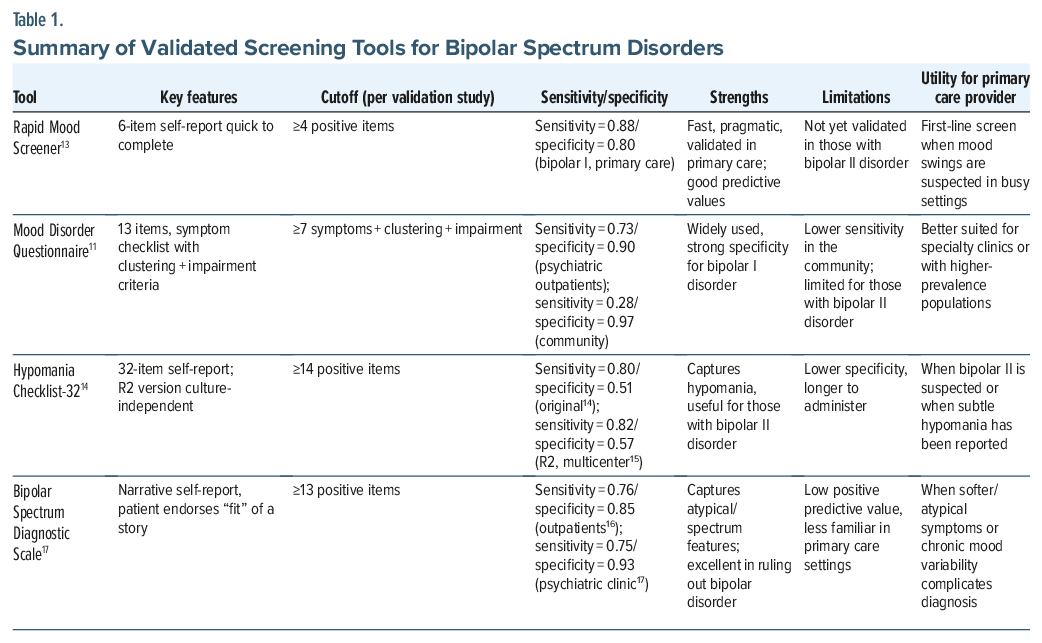
Several screening tools have been developed to aid in the detection of BSDs. Of these, the MDQ is among the most referenced. When it was originally validated in psychiatric outpatients, a threshold of 7 of 13 symptoms yielded a sensitivity of ∼0.73 and a specificity of ∼0.90 for BSDs.11 However, when it was applied to those in the general population, the MDQ’s sensitivity dropped (to ∼0.28), albeit with very high specificity (∼0.97).12 The limited sensitivity in community samples is partly due to the MDQ being better for the detection of bipolar I disorder and manic symptoms, but it is less sensitive for detecting those with bipolar II disorder and those with subthreshold bipolarity. This underscores the importance of interpreting negative or borderline MDQ results.
The Rapid Mood Screener (RMS) is a concise tool designed for use in primary care settings.13 It was developed to address the limitations (particularly the length and scoring complexity) of the MDQ. Endorsing 4 or more items on the RMS yielded a sensitivity of 0.88 and a specificity of 0.80, with a positive predictive value of 0.80 and a negative predictive value of 0.88.13 This demonstrates favorable validity compared to the MDQ. However, the RMS has not been validated in those with bipolar II disorder, which limits its generalizability.
Another widely used instrument, the Hypomania Checklist-32 (HCL-32), provides an alternative screening option with a somewhat higher sensitivity for bipolar II disorder; however, the trade-off is a lower specificity.14 A larger, multinational study used the HCL-32 revised version 2, and the results showed that it had a high sensitivity (∼82%) and moderate specificity (∼57%) when distinguishing those with unipolar depression from those with BSD, using standard DSM-IV criteria.15
Less well known, but still useful in certain settings, is the Bipolar Spectrum Diagnostic Scale (BSDS). It captures “softer” or atypical presentations of BSD, including subthreshold hypomania.16 While data from the BSDS differ by study, sensitivity is generally in the 0.70–0.80 range with specificity that ranges from ∼0.65 to 0.90, depending on the cutoff and the population studied.17 Overall, this scale is effective at ruling out BSD at the expense of a low positive predictive value (0.16). Table 1 summarizes details of the screening tools, as well as their specificity and sensitivity.
Which Screening Test Should Be Used When Evaluating Mood Symptoms?
When a PCP encounters a patient with depression and “mood swings,” the RMS offers a sensible starting point. Its 6 items can be completed within minutes, and studies have demonstrated a high sensitivity and specificity for bipolar I disorder, making it well suited for busy primary care practices. The HCL-32 is useful when attempting to capture more subtle features of hypomania. Patients may describe stretches of increased energy, sociability, or productivity that fall short of full mania, and they are often framed as periods when they “felt like themselves again” or “finally got things done.” These episodes may not cause obvious disruptions, which explains why they frequently escape recognition when targeted questions are omitted. In the same vein, the BSDS helps when a person’s story reflects softer or atypical symptoms (eg, chronic irritability, variable energy levels, or shifts in motivation) that do not meet DSM criteria. A PCP might notice inconsistent academic or occupational performance, cycles of interpersonal conflict, or narratives of “being up and down” without having clear episodes of mania. In such cases, the BSDS can identify patterns that other tools miss. The MDQ is familiar and widely used, particularly in psychiatric outpatient settings, but in community or primary care settings, its use can be time-consuming, and it is less sensitive than the RMS.
How Can BSDs Be Distinguished From Other Conditions?
Differential diagnosis. Before settling on a psychiatric diagnosis, medical and neurologic conditions that may mimic the mood swings of BSDs must be considered. The differential diagnosis is shaped by the patient’s age, comorbidities, and context of the presentation. In younger individuals, epilepsy, particularly temporal lobe epilepsy, can produce episodic mood changes, behavioral lability, and confusion that is reminiscent of a manic or hypomanic state. Traumatic brain injuries, including even mild repeated concussions, are another notable mimic, which sometimes leads to late-onset mood instability or personality changes. Multiple sclerosis and other neuroinflammatory disorders occasionally produce affective lability, especially when lesions involve limbic structures. In older adults or in those with risk factors of vascular disease, sudden behavioral or mood changes may reflect cerebrovascular events (eg, stroke) rather than a primary psychiatric disorder. Endocrine disturbances, most notably thyroid dysfunction, can cause reversible presentations of mania throughout the lifespan. Infectious etiologies of mood liability, like HIV infection and syphilis, need to be considered in those with acute or atypical features.
Once secondary causes of mood disorders have been excluded, physicians should disentangle BSD from other psychiatric syndromes with overlapping symptoms, particularly in primary care settings where overlapping symptoms and comorbidities (eg, borderline personality disorder (BPD), ADHD, and trauma-related states) may each echo the mood lability or functional shifts seen in BSDs.
BSD or MDD?
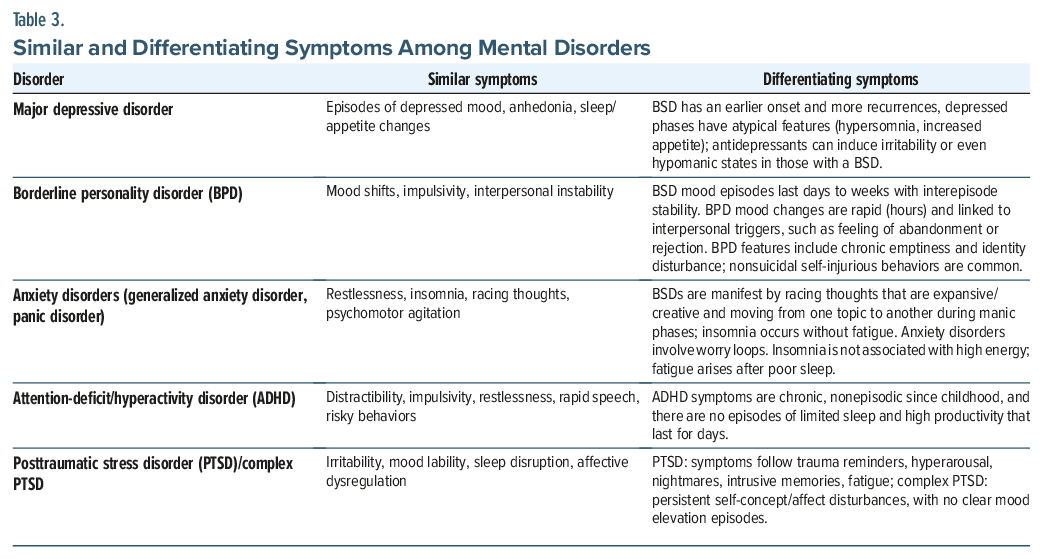
Most patients with BSD present during a depressive episode, making unipolar depression the most common misdiagnosis. Studies suggest that more than half of patients with BSD initially receive a diagnosis of unipolar depression.18 However, bipolar depression tends to have an earlier onset, a greater rate of recurrence, and more atypical features (eg, hypersomnia, increased appetite, mood lability, and psychomotor retardation) compared to unipolar depression. A history of antidepressant-induced activation, in which an individual becomes agitated or switches rapidly after the initiation of a selective serotonin reuptake inhibitor, strongly suggests the presence of a BSD. A family history of BSD further increases the probability of a BSD diagnosis.
BSD or BPD?
Distinguishing BSD, especially bipolar II disorder, from BPD remains one of the most frequent diagnostic dilemmas in primary care settings. Both conditions involve mood shifts, impulsivity, and interpersonal instability. In addition, co-occurrence is common, and nearly 20% of those with BSD also meet criteria for BPD, which further complicates the diagnosis.19 Yet, the time course provides a critical clue; mood changes associated with bipolar II disorder occur in episodes that last days to weeks, with interepisode stability, whereas mood fluctuations in BPD are rapid, often lasting only hours, and are tightly linked to interpersonal stressors.20 Other clinical clues help differentiate the 2 conditions. BPD is characterized by chronic feelings of emptiness, identity disturbance, and an intense fear of abandonment, which are not diagnostic features of BSD. In contrast, grandiosity, a decreased need for sleep, and elevated goal-directed activity strongly support a diagnosis of BSD.
BSD or Anxiety Disorder?
Symptoms of anxiety frequently complicate the diagnosis of BSD and, in some patients, dominate the initial presentation due to high comorbidity.21 Generalized anxiety disorder and panic disorder, in particular, may mimic hypomanic states. Patients may describe restlessness, an inability to relax, difficulty concentrating, or racing thoughts. They often report lying awake at night replaying worries, which can superficially resemble the insomnia and accelerated thought processes of hypomania. Increased psychomotor activity (including pacing, fidgeting, and agitation) can be mistaken for heightened energy. The distinction lies in the quality and temporal pattern of symptoms. In those with BSD, racing thoughts are often expansive, creative, or goal-directed, with the patient describing new plans, projects, or ideas. By contrast, in anxiety disorders, cognitive acceleration reflects worry loops (ie, ruminative, repetitive concerns about potential threats or catastrophes). Similarly, insomnia in BSD often occurs without fatigue, as patients feel energized despite minimal sleep, whereas insomnia in anxiety stems from physiological hyperarousal and usually results in exhaustion.
BSD or ADHD?
Distinguishing BSD from ADHD can be challenging, particularly in adolescents and young adults. A developmental history remains informative but not definitive. While the DSM criteria emphasize that ADHD’s onset arises before age 12 years, many individuals with ADHD go unrecognized until later in life. Both conditions share distractibility, impulsivity, restlessness, rapid speech, and even risky behaviors (eg, substance use or poor decision-making). Emotional lability also appears in both, creating further overlap.
Several features help distinguish them. In those with a BSD, symptoms cluster into episodic shifts in mood and behavior with intervening periods of relative stability, whereas symptoms of ADHD are more continuous across time, even if they vary in intensity.22 Sleep patterns also diverge; individuals in a hypomanic episode often report a markedly reduced need for sleep without fatigue, while those with ADHD may experience insomnia but usually feel tired. Bipolar episodes frequently involve expansive or elevated mood, increased goal-directed activity, and grandiosity, phenomena rarely seen in ADHD. Although ADHD carries risks of impulsive or dangerous behaviors, these do not typically occur in the context of discrete mood episodes.
BSD, PTSD, or Complex PTSD?
Trauma-related disorders, including posttraumatic stress disorder (PTSD) and complex PTSD, can mimic BSD, especially when they present with irritability, mood lability, disrupted sleep, and affective dysregulation.23 In those with PTSD, these symptoms often follow trauma reminders and are accompanied by hyperarousal, nightmares, and intrusive memories, whereas in BSD, symptoms occur in discrete episodes that are not tethered to external triggers. Complex PTSD adds persistent disturbances in self-concept, affect regulation, and relationships, which may superficially resemble bipolar II disorder but lack clear periods of elevated or expansive mood. Studies confirm that there are high rates of comorbidity, with trauma exposure worsening the course of BSD and complicating its recognition.
Key distinctions rest on the temporal pattern and the quality of symptoms. Those with BSD present with episodic mood elevation, a decreased need for sleep without fatigue, grandiosity, and an increase in goal-directed activity. PTSD and complex PTSD produce chronic or trigger-based affective shifts, nightmares, hypervigilance, and fatigue after poor sleep. A trauma history, especially of repeated early adversity, strongly suggests a diagnosis of complex PTSD, while spontaneous episodes of elevated mood and energy support a diagnosis of a BSD. In primary care settings, probing for episodicity, triggers, and expansive mood remains central to differentiation from other conditions.
How Can BSDs Be Managed Effectively?
Principles of management. Because BSDs are chronic and recurrent, long-term, individualized management should be at the foundation of treatment strategies.24 Therefore, the management goals for BSDs include achieving mood stability, preventing relapse, minimizing adverse medication effects, and improving overall function. Treatment should be individualized and based on the patient’s needs and personal preference.23,24
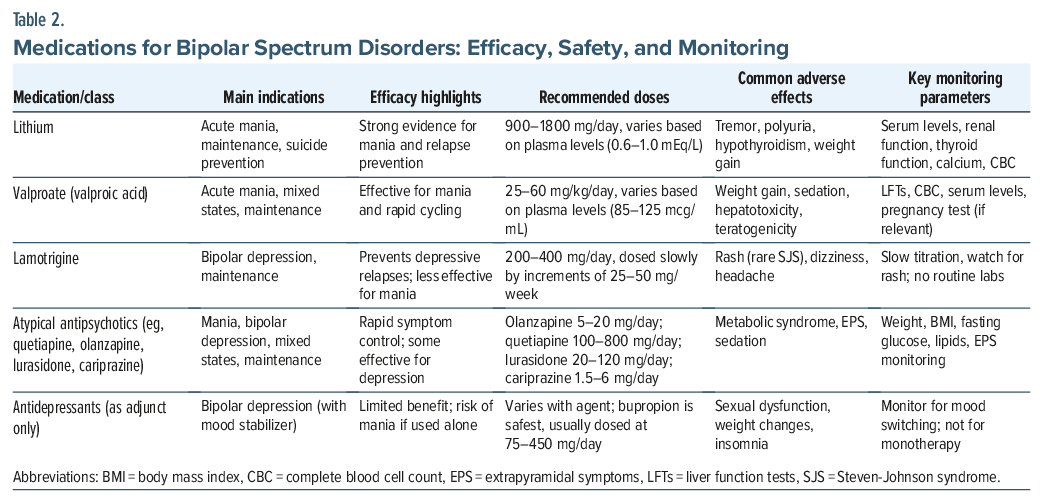
Pharmacologic interventions. Historically, mood stabilizers (eg, lithium, valproate, lamotrigine, and atypical antipsychotics) have been the cornerstones of treatment for BSDs for both acute and maintenance phases of treatment. These agents work differently from one another and have different therapeutic targets. For example, lithium and valproate are used primarily for treating acute mania and for preventing relapses of mania.25,26 Lithium has also demonstrated efficacy as an antidepressant,26 as well as an agent to prevent suicide.27
Lamotrigine has shown efficacy for the depressive phase of bipolar depression but not for the acute treatment of mania, and it has prevented depressive relapses.28 However, dosing of lamotrigine needs to be increased slowly to mitigate adverse reactions, eg, Steven-Johnson syndrome.29
Atypical antipsychotic agents are increasingly used, especially for mixed episodes or when psychotic features arise during a manic episode. Quetiapine and lurasidone are also beneficial in bipolar depression.30,31
Use of antidepressant monotherapy in BSDs is generally discouraged. This caution applies across the bipolar spectrum, as hypomania can be difficult to distinguish from evolving mania, and 5%–15% of individuals initially diagnosed with bipolar II later meet criteria for bipolar I.9 The use of antidepressants without an effective “safety net” of coadministered mood stabilizers carries a risk of mood destabilization that can cause a switch into mania or hypomania, as well as promotion of rapid cycling.32 If antidepressants are used, they should be used adjunctively to mood stabilizers, and even in those cases, they should be dosed as low as possible.33 Bupropion is thought to be the antidepressant that is least likely to induce affective cycling, and it is often a good choice for those who have bipolar depression.34 Individuals who are taking antidepressants should be closely monitored for mood instability.
When selecting a medication, clinicians should balance efficacy with tolerability, safety, and comorbidity. Some medications used for BSDs require periodic blood monitoring. For example, lithium levels should be checked periodically to determine whether they are within the therapeutic window of efficacy and safety. Likewise, use of lithium requires monitoring of kidney and thyroid function, as well as blood counts.35,36
Medications, such as valproate and carbamazepine, also call for blood tests for therapeutic levels, as well as for monitoring of hematologic and hepatic panels.37,38 Atypical antipsychotics require periodic metabolic laboratory testing because of their risk of inducing weight gain, dyslipidemia, and diabetes.39 In women, the risks of treatments recommended during pregnancy should always be considered due to their potential risk to the fetus (eg, teratogenicity from mood stabilizers).37,40 Table 2 summarizes these points and lists target symptoms, patterns of efficacy, doses, key side effects, and recommended monitoring strategies.
Nonpharmacologic interventions. For some individuals, pharmacotherapy alone is not enough to manage BSD. In those instances, adjunctive strategies should be considered. Psychoeducation can increase treatment adherence, facilitate early recognition of emerging mood symptoms, and promote lifestyle regularity (eg, sleep hygiene).41,42 Cognitive-behavioral therapy and family-focused therapy can mitigate residual mood symptoms, particularly depression, reduce chances of recurrence or relapse, and teach patients and family members useful coping skills to manage interpersonal stressors.43,44 Finally, lifestyle measures (eg, exercise, a healthy diet, sleep regulation, and substance use reduction), when needed, can improve long-term outcomes (Table 3).45
Management of comorbidities. Individuals with BSD have higher rates of metabolic syndrome, cardiovascular disease, and substance use disorders. BSD is associated with premature mortality: Life expectancy is shortened by roughly 12–14 years, with elevated cardiovascular mortality (about 1.6-to 2-fold) and occurs, on average, by nearly 2 decades earlier than the general population.24 This medical burden emphasizes collaborative care models that integrate psychiatric and medical treatment goals, particularly in settings where specialty access is constrained. Primary care physicians who treat BSD patients need to monitor weight and check for diabetes, hyperlipidemia, and hypertension on a regular basis, particularly when they are receiving atypical antipsychotics.46,47
When Should Patients Be Referred to a Mental Health Professional?
Considerable research has shown the benefits of using collaborative care models (between PCPs and specialty care providers) for the diagnosis and management of BSD,48,49 suggesting a critical role for consultation with specialty care providers and the need for systems of care to facilitate such consultation, especially where mental health professionals are less available or when waits to see them are especially long. Given the challenge of accurately diagnosing bipolar disorder, especially in the depressive phase, PCPs should maintain a low threshold considering consultation with a mental health professional for diagnostic clarification. When concern for hypomania/mania arises, the visit can be organized into 3 steps: (1) immediate safety screen, (2) same-day disposition plan, and (3) rapid coordination of specialty care. The safety screen could be brief, focusing on psychosis or severe agitation, suicidal or homicidal ideation, markedly impaired judgment, and inability to sleep for several nights without the need for sleep, accompanied by high energy, substance intoxication or withdrawal, and imminent high-risk behaviors (eg, access to weapons). Presence of any of these features should trigger same-day escalation, typically emergency evaluation. If urgent escalation is not indicated, the outpatient plan should prioritize short-term risk reduction and containment rather than diagnostic clarification. Patients should be advised to pause major decisions, limit use of substances (including stimulants), involve a trusted support person with the patient’s consent, and schedule follow-up contact within 24 to 72 hours. This workflow could be strengthened by having a named psychiatry contact for rapid consultation and a standing plan for expedited emergency psychiatric evaluation and inpatient admission when needed, reducing delays at the point of greatest clinical risk.
What Happened to Mr B?
On the inpatient unit, Mr B underwent a formal psychiatric evaluation that confirmed his diagnosis of bipolar I disorder. He was started on lithium as a mood stabilizer, given its evidence-based efficacy in classic euphoric mania. Baseline laboratory tests (including serum creatinine, blood urea nitrogen, thyroid-stimulating hormone, and electrolytes, along with an electrocardiogram) were obtained, consistent with lithium monitoring guidelines. Potential side effects, the importance of medication adherence, hydration, and limiting the use of nonsteroidal anti-inflammatory drugs (as they can raise lithium levels) were discussed with Mr B.
Over the next few weeks, Mr B’s hypomanic symptoms subsided, and his mood stabilized, without the recurrence of depressive symptoms. Regular follow-up was arranged for monitoring his lithium levels, assessing his renal and thyroid function tests (every 3–6 months), and continuing his psychiatric care. Psychosocial interventions (including psychoeducation about early warning signs) and lifestyle regularity were provided to reduce his relapse risk.
CONCLUSION
Among those with depression in primary care settings, the prevalence of BSD is close to 17%, and the use of structured clinical interviews has resulted in fewer missed cases than when screening questionnaires have been employed. Diagnosis of BSD is often complicated by the fact that most individuals with BSD present for care during a depressive episode, which is more frequent than hypomanic or manic episodes; therefore, there is a tendency to diagnose depressive illnesses. While an earlier age at onset and a family history of a BSD are associated with the diagnosis of BSD, there is no clear age cutoff, and having a family history is not as definitive as having family members with MDD.
When distinguishing BSDs from other disorders, it is helpful to know that those with BPD have chronic feelings of emptiness, an identity disturbance, and an intense fear of abandonment; however, those are not characteristics of BSD. In those with BSD, symptoms cluster into episodic shifts in mood and behavior with intervening periods of relative stability, whereas symptoms of ADHD are more continuous.
Since pharmacotherapy alone is not enough to manage BSD, adjunctive strategies should be considered. Moreover, both pharmacology and psychoeducation can increase treatment adherence, facilitate early recognition of emerging mood symptoms, and promote lifestyle regularity that are often disrupted by BSD.
Article Information
Published Online: May 12, 2026. https://doi.org/10.4088/PCC.25f04157
© 2026 Physicians Postgraduate Press, Inc.
Submitted: December 1, 2025; accepted January 28, 2026.
To Cite: Soliman M, Ghaznavi S, Mischoulon D, et al. Differentiating bipolar spectrum disorders from other psychiatric disorders with shared features: strategies and implications. Prim Care Companion CNS Disord. 2026;28(3):25f04157.
Author Affiliations: Department of Psychiatry, Massachusetts General Hospital, Boston, Massachusetts (Soliman, Ghaznavi, Mischoulon, Stern); Harvard Medical School, Boston, Massachusetts (Soliman, Ghaznavi, Mischoulon, Stern).
Soliman, Ghaznavi, and Mischoulon are co-first authors; Stern is the senior author.
Corresponding Author: Mohamed A. Soliman, MBA, PhD, MSc, @ Department of Psychiatry, Massachusetts General Hospital and Harvard Medical School, Boston, Massachusetts ([email protected]).
Financial Disclosure: Dr Stern has received royalties from Elsevier for editing textbooks on psychiatry. Drs Soliman, Ghaznavi, and Mischoulon report no financial relationships.
Funding/Support: None.
Clinical Points
- People rarely seek help from physicians when their mood is elevated; however, they frequently present for care when they are depressed, have low energy levels, or are impulsive.
- Because hypomania and mania are often underreported and present through downstream consequences, emergency department follow-up visits for injury, intoxication, or acute psychosocial crisis are practical opportunities for primary care clinicians to screen for recent sleep reduction and episodic behavioral activation suggestive of bipolar spectrum illness.
- Clinicians should ask about lifetime mood episodes, as well as details of the family’s psychiatric history, especially bipolar disorder in first-degree relatives, and documentation of a prior antidepressant response.
- Primary care physicians should maintain a low threshold for considering consultation with a mental health professional for diagnostic clarification and management of bipolar spectrum disorders.
References (49)

- Drancourt N, Etain B, Lajnef M, et al. Duration of untreated bipolar disorder: missed opportunities on the long road to optimal treatment. Acta Psychiatr Scand. 2013;127(2):136–144. PubMed CrossRef
- Lish JD, Dime-Meenan S, Whybrow PC, et al. The National Depressive and manic-depressive association (DMDA) survey of bipolar members. J Affect Disord. 1994;31(4):281–294. PubMed CrossRef
- Judd LL, Akiskal HS, Schettler PJ, et al. The long-term natural history of the weekly symptomatic status of bipolar I disorder. Arch Gen Psychiatry. 2002;59(6):530–537. PubMed CrossRef
- Judd LL, Akiskal HS, Schettler PJ, et al. A prospective investigation of the natural history of the long-term weekly symptomatic status of bipolar II disorder. Arch Gen Psychiatry. 2003;60(3):261–269. PubMed CrossRef
- Vöhringer PA, Perlis RH. Discriminating between bipolar disorder and major depressive disorder. Psychiatr Clin North Am. 2016;39(1):1–10. PubMed CrossRef
- American Psychiatric Association. (2022). Bipolar and related disorders. In Diagnostic and statistical manual of mental disorders (5th ed., text rev.).
- McClellan J, Kowatch R, Findling RL, et al. Practice parameter for the assessment and treatment of children and adolescents with bipolar disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(1):107–125. PubMed CrossRef
- Nivoli A, Murru A, Valenti P, et al. Bipolar disorder in the elderly: a cohort study comparing older and younger patients. Acta Psychiatr Scand. 2014;130(5):364–373. PubMed
- Cerimele JM, Chwastiak LA, Dodson S, et al. The prevalence of bipolar disorder in general primary care samples: a systematic review. Gen Hosp Psychiatry. 2014;36(1):19–25. PubMed CrossRef
- Daveney J, Panagioti M, Waheed W, et al. Unrecognized bipolar disorder in patients with depression managed in primary care: a systematic review and meta-analysis. Gen Hosp Psychiatry. 2019;58:71–76. PubMed CrossRef
- Hirschfeld RM, Williams JB, Spitzer RL, et al. Development and validation of a screening instrument for bipolar spectrum disorder: the Mood Disorder Questionnaire. Am J Psychiatry. 2000;157(11):1873–1875. PubMed CrossRef
- Hirschfeld RA, Holzer C, Calabrese JR, et al. Validity of the mood disorder questionnaire: a general population study. Am J Psychiatry. 2003;160(1):178–180.
- McIntyre RS, Patel MD, Masand PS, et al. The Rapid Mood Screener (RMS): a novel and pragmatic screener for bipolar I disorder. Curr Med Res Opin. 2021;37(1):135–144. PubMed CrossRef
- Angst J, Adolfsson R, Benazzi F, et al. The Hypomania Checklist (HCL-32): towards a self-assessment tool for hypomanic symptoms in outpatients. J Affect Disord. 2005;88(2):217–233. PubMed CrossRef
- Gamma A, Angst J, Azorin JM, et al. Transcultural validity of the Hypomania Checklist-32 (HCL-32) in patients with major depressive episodes. Bipolar Disord. 2013;15(6):701–712. PubMed CrossRef
- Ghaemi SN, Miller CJ, Berv DA, et al. Sensitivity and specificity of a new bipolar spectrum diagnostic scale. J Affect Disord. 2005;84(2–3):273–277. PubMed
- Zimmerman M, Galione JN, Chelminski I, et al. Performance of the bipolar spectrum diagnostic scale in psychiatric outpatients. Bipolar Disord. 2010;12(5):528–538. PubMed CrossRef
- McIntyre RS, Zimmerman M, Goldberg JF, et al. Differential diagnosis of major depressive disorder versus bipolar disorder: current status and best clinical practices. J Clin Psychiatry. 2019;80(3):ot18043ah2. PubMed CrossRef
- Fornaro M, Orsolini L, Marini S, et al. The prevalence and predictors of bipolar and borderline personality disorders comorbidity: systematic review and meta-analysis. J Affect Disord. 2016;195:105–118. PubMed CrossRef
- Berk M, Corrales A, Trisno R, et al. Bipolar II disorder: a state-of-the-art review. World Psychiatry. 2025;24(2):175–189. PubMed CrossRef
- McIntyre RS, Soczynska JK, Bottas A, et al. Anxiety disorders and bipolar disorder: a review. Bipolar Disord. 2006;8(6):665–676. PubMed CrossRef
- Comparelli A, Polidori L, Sarli G, et al. Differentiation and comorbidity of bipolar disorder and attention deficit and hyperactivity disorder in children, adolescents, and adults: a clinical and nosological perspective. Front Psychiatry. 2022;13:949375. PubMed CrossRef
- Nierenberg AA, Agustini B, Köhler-Forsberg O, et al. Diagnosis and treatment of bipolar disorder: a review. JAMA. 2023;330(14):1370–1380. PubMed CrossRef
- Goes FS. Diagnosis and management of bipolar disorders. BMJ. 2023;381:e073591. PubMed CrossRef
- Mari J, Dieckmann LHJ, Prates-Baldez D, et al. The efficacy of valproate in acute mania, bipolar depression and maintenance therapy for bipolar disorder: an overview of systematic reviews with meta-analyses. BMJ Open. 2024;14(11):e087999. PubMed CrossRef
- Barroilhet SA, Ghaemi SN. When and how to use lithium. Acta Psychiatr Scand. 2020;142(3):161–172. PubMed CrossRef
- Smith KA, Cipriani A. Lithium and suicide in mood disorders: updated meta-review of the scientific literature. Bipolar Disord. 2017;19(7):575–586. PubMed CrossRef
- Haenen N, Kamperman AM, Prodan A, et al. The efficacy of lamotrigine in bipolar disorder: a systematic review and meta-analysis. Bipolar Disord. 2024;26(5):431–441. PubMed CrossRef
- Goldsmith DR, Wagstaff AJ, Ibbotson T, et al. Lamotrigine: a review of its use in bipolar disorder. Drugs. 2003;63(19):2029–2056. PubMed
- Kadakia A, Dembek C, Heller V, et al. Efficacy and tolerability of atypical antipsychotics for acute bipolar depression: a network meta-analysis. BMC Psychiatry. 2021;21(1):249. PubMed CrossRef
- Li S, Xu C, Hu S, et al. Efficacy and tolerability of FDA-approved atypical antipsychotics for the treatment of bipolar depression: a systematic review and network meta-analysis. Eur Psychiatry. 2024;67(1):e29. PubMed CrossRef
- Viktorin A, Lichtenstein P, Thase ME, et al. The risk of switch to mania in patients with bipolar disorder during treatment with an antidepressant alone and in combination with a mood stabilizer. Am J Psychiatry. 2014;171(10):1067–1073. PubMed CrossRef
- Pacchiarotti I, Bond DJ, Baldessarini RJ, et al. The International Society for Bipolar Disorders (ISBD) task force report on antidepressant use in bipolar disorders. Am J Psychiatry. 2013;170(11):1249–1262. PubMed CrossRef
- Leverich GS, Altshuler LL, Frye MA, et al. Risk of switch in mood polarity to hypomania or mania in patients with bipolar depression during acute and continuation trials of venlafaxine, sertraline, and bupropion as adjuncts to mood stabilizers. Am J Psychiatry. 2006;163(2):232–239. PubMed CrossRef
- Nikolova VL, Pattanaseri K, Hidalgo-Mazzei D, et al. Is lithium monitoring NICE? Lithium monitoring in a UK secondary care setting. J Psychopharmacol. 2018;32(4):408–415. PubMed CrossRef
- Shine B, McKnight RF, Leaver L, et al. Long-term effects of lithium on renal, thyroid, and parathyroid function: a retrospective analysis of laboratory data. Lancet. 2015;386(9992):461–468. PubMed CrossRef
- Carvalho AF, Firth J, Vieta E. Bipolar disorder. N Engl J Med. 2020;383(1):58–66. PubMed CrossRef
- Johannessen Landmark C, Wolden M, et al. A precision approach to therapeutic drug monitoring of valproate to evaluate markers of toxicity. Epilepsia. 2025;66(10):3741–3756. PubMed CrossRef
- Marder SR, Cannon TD. Schizophrenia. N Engl J Med. 2019;381(18):1753–1761. PubMed CrossRef
- Galbally M, Roberts M, Buist A, et al. Mood stabilizers in pregnancy: a systematic review. Aust New Zeal J Psychiatry. 2010;44(11):967–977. PubMed CrossRef
- Miklowitz DJ, Efthimiou O, Furukawa TA, et al. Adjunctive psychotherapy for bipolar disorder: a systematic review and component network meta-analysis. JAMA Psychiatry. 2021;78(2):141–150. PubMed CrossRef
- Soo SA, Zhang ZW, Khong SJ, et al. Randomized controlled trials of psychoeducation modalities in the management of bipolar disorder: a systematic review. J Clin Psychiatry. 2018;79(3):17r11750. PubMed CrossRef
- Chiang KJ, Tsai JC, Liu D, et al. Efficacy of cognitive-behavioral therapy in patients with bipolar disorder: a meta-analysis of randomized controlled trials. PLoS ONE. 2017;12(5):e0176840. PubMed
- Firth J, Solmi M, Wootton RE, et al. A meta-review of “lifestyle psychiatry”: the role of exercise, smoking, diet and sleep in the prevention and treatment of mental disorders. World Psychiatry. 2020;19(3):360–380. PubMed CrossRef
- Simjanoski M, Patel S, Boni RD, et al. Lifestyle interventions for bipolar disorders: a systematic review and meta-analysis. Neurosci Biobehav Rev. 2023;152:105257. PubMed CrossRef
- McElroy SL, Keck PE. Metabolic syndrome in bipolar disorder: a review with a focus on bipolar depression. J Clin Psychiatry. 2014;75(1):46–61. PubMed CrossRef
- Simoons M, Mulder H, Doornbos B, et al. Metabolic syndrome at an outpatient clinic for bipolar disorders: a case for systematic somatic monitoring. Psychiatr Serv. 2019;70(2):143–146. PubMed CrossRef
- Cerimele JM, Fortney JC, Pyne JM, et al. Bipolar disorder in primary care: a qualitative study of clinician and patient experiences with diagnosis and treatment. Fam Pract. 2018;36(1):32–37. CrossRef
- Hoppe I, Watson S, Ahuja N, et al. Reducing the delay in the diagnosis of bipolar disorder: a qualitative study. Health Expect. 2025;28(4):e70398. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!