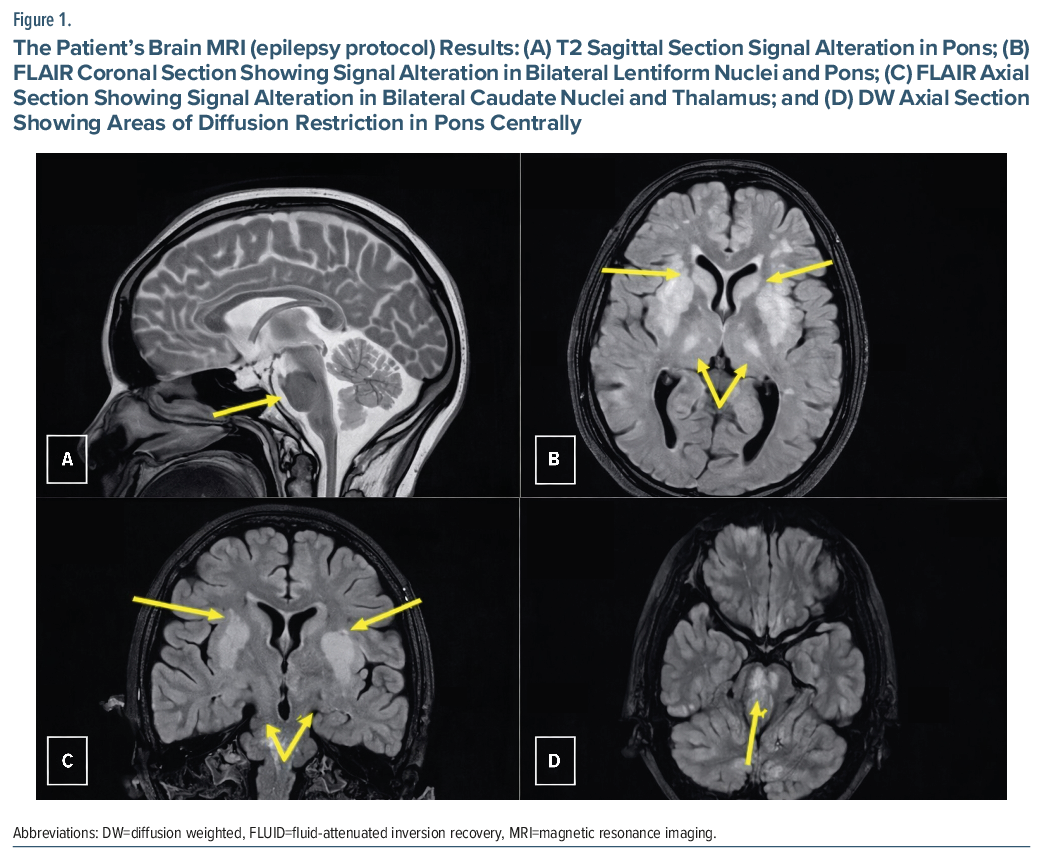
In psychiatric practice, extrapyramidal symptoms (EPS) are commonly encountered as side effects of dopamine antagonists, such as antipsychotics. However, these symptoms can also arise from underlying neurological insults, including osmotic demyelination syndrome (ODS), which is precipitated by rapid correction of chronic hyponatremia.1 ODS may involve extrapontine structures like the basal ganglia, leading to parkinsonian features and increased sensitivity to dopamine antagonists.2 This is particularly relevant in patients with epilepsy on medications like carbamazepine, which can induce hyponatremia via syndrome of inappropriate antidiuretic hormone secretion (SIADH).3 We present a case in which initial psychiatric referral for presumed domperidone-induced EPS uncovered a complex interplay of hyponatremia, demyelination, and neuroleptic hypersensitivity, underscoring the importance of multidisciplinary evaluation in atypical presentations.
Case Report
Ms A, a 44-year-old woman, was referred to the psychiatry outpatient department (OPD) for evaluation of acute behavioral changes and motor symptoms. Her medical history included focal epilepsy since the age of 16 years (last seizure in 2024), managed with carbamazepine controlled release 300 mg twice daily, alongside hypertension for 20 years (treated with cilnidipine 10 mg and telmisartan 40 mg once daily). Family history was notable for hypertension and diabetes in her parents. There was no prior psychiatric history, substance use, or known allergies.
The patient presented with a 4-day history of mutism, dysphagia, child- like behavior progressing to abnormal mannerisms, reduced sleep, and tremors. These symptoms emerged shortly after receiving domperidone for recurrent vomiting and dizziness during a recent hospitalization. On psychiatric evaluation, she exhibited mask-like facies, bradykinesia, rigidity, and hypertonia in all limbs, suggestive of severe EPS with high sensitivity to domperidone, a peripheral dopamine antagonist. Sensory functions were preserved, but she displayed emotional lability. The ear, nose, and throat assessment confirmed upper motor neuron palsy of the vocal cords. No psychotic features, mood disturbances, or cognitive deficits were evident, raising suspicion for an organic etiology beyond simple medication side effects.
The clinical timeline was as follows: July 21, 2025: Hospitalized for recurrent vomiting and dizziness; laboratory findings revealed severe hyponatremia (Na+ 112 mmol/L) and hypokalemia, attributed to chronic carbamazepine use. Domperidone was initiated for symptom control, alongside conservative management. July 31, 2025: Discharged after sodium correction. August 2, 2025: Developed abnormal behavior and child-like speech, progressing to mutism, dysphagia, and tremors. August 4, 2025: Readmitted and referred to psychiatry OPD for suspected domperidone-induced EPS. Further evaluation identified ODS secondary to rapid hyponatremia correction, explaining the neuroleptic hypersensitivity.
Initial psychiatric workup focused on ruling out catatonia, conversion disorder, or antipsychotic-induced EPS, but the temporal association with hyponatremia correction prompted neurological consultation. Laboratory results included hemoglobin of 10.7 g/dL, total leukocyte count of 4,900/mm3, C-reactive protein of 113 mg/L, sodium of 134 mmol/L, and potassium of 3.0 mmol/L. Brain magnetic resonance imaging (MRI) demonstrated central pontine myelinolysis and extrapontine myelinolysis (Figure 1), which confirmed ODS as the underlying cause, with basal ganglia involvement accounting for the exaggerated EPS and sensitivity to domperidone’s dopaminergic blockade.
Given the psychiatric presentation, initial management addressed potential neuroleptic effects with discontinuation of domperidone and supportive measures. Multidisciplinary input led to the continuation of anticonvulsants (adjusted to lacosamide 100 mg intravenous twice daily and levetiracetam 750 mg once daily to minimize hyponatremia risk) and dopaminergic therapy (amantadine 100 mg at bedtime, escalated to twice daily), alongside antihypertensives (telmisartan 40 mg twice daily), melatonin (3 mg/day) for sleep, and nutritional support via Ryle tube feeding, which was later transitioned to oral intake as swallowing improved.
Over the following weeks, the patient showed progressive resolution of mutism, EPS, and behavioral abnormalities. She regained verbal communication, resumed oral feeding, and demonstrated improved emotional regulation. Repeat assessments confirmed no emergent psychiatric disorder, emphasizing the iatrogenic neurological basis of her symptoms.
Discussion
This case illustrates a rare psychiatric presentation of ODS in a patient with focal epilepsy, precipitated by carbamazepine- induced hyponatremia and subsequent rapid sodium correction, leading to demyelination particularly in the basal ganglia. Carbamazepine, commonly used for seizure control and mood stabilization, is a well-known cause of hyponatremia through the SIADH, which can set the stage for ODS if sodium levels are corrected too aggressively.3 The resulting neuroleptic hypersensitivity amplified the extrapyramidal effects of domperidone, a peripheral dopamine antagonist administered for gastrointestinal symptoms, mimicking a primary medication-induced EPS or even a functional neurological disorder. In psychiatric settings, such atypical motor and behavioral presentations, including mutism, rigidity, and emotional lability, can be diagnostically challenging, often leading to initial misattribution to psychotropic side effects or psychiatric conditions like catatonia.2 Recent literature highlights similar psychiatric manifestations of ODS, such as catatonia following hyponatremia correction, emphasizing the overlap between neurological insults and neuropsychiatric symptoms.4
This case underscores the importance of vigilance for organic etiologies underlying apparent EPS, especially in those on hyponatremia- inducing agents like carbamazepine. Electrolyte monitoring is crucial in epilepsy patients on such medications, as chronic hyponatremia increases vulnerability to ODS upon rapid correction (recommended limit ≤8 mmol/L/day).1 Early MRI is essential for confirming basal ganglia involvement, which can explain heightened sensitivity to dopaminergic blockade and guide management. In this patient, switching from carbamazepine to lacosamide, a sodium channel blocker with lower hyponatremia risk, helped mitigate recurrence, aligning with reports of improved electrolyte balance postsubstitution.5
Therapeutically, dopaminergic agents like amantadine were pivotal in reversing rigidity and motor symptoms, consistent with basal ganglia dysfunction in extrapontine myelinolysis. While supportive care remains the cornerstone, emerging evidence supports adjunctive plasmapheresis for refractory or chronic ODS, even when initiated weeks after onset, leading to complete clinical and radiological recovery in select cases.6 Multidisciplinary collaboration prevented diagnostic delays, highlighting the need for integrated psychiatric-neurological approaches.
Epidemiologically, ODS remains rare, occurring in approximately 0.05% of hyponatremic hospitalizations, with risk factors including severe initial hyponatremia (<120 mmol/L), hypokalemia, and hypovolemia, though not all cases involve rapid correction.7 This report advocates for heightened awareness in psychiatric OPDs to recognize ODS as an organic mimic of EPS or catatonia, potentially averting misdiagnosis and improving outcomes through timely intervention and preventive strategies.
Article Information
Published Online: April 21, 2026. https://doi.org/10.4088/PCC.25cr04093
© 2026 Physicians Postgraduate Press, Inc.
Prim Care Companion CNS Disord 2026;28(2):25cr04093
Submitted: September 24, 2025; accepted December 12, 2025.
To Cite: Pant V, Jain A, Garg S, et al. Osmotic demyelination syndrome presenting as extrapyramidal symptoms and neuroleptic sensitivity in a patient with focal epilepsy: a psychiatric perspective. Prim Care Companion CNS Disord 2026;28(2):25cr04093.
Author Affiliations: Department of Psychiatry, Shri Guru Ram Rai Institute of Medical and Health Sciences, Uttarakhand, India (all authors).
Corresponding Author: Shobit Garg, MD, DPM, Department of Psychiatry, Shri Guru Ram Rai Institute of Medical and Health Sciences, Shri Guru Ram Rai University, Dehradun, Uttarakhand, 248001 India ([email protected]).
Patient Consent: Written informed consent was obtained from the patient to publish the case report and accompanying images, and information, including dates, has been de-identified to protect patient anonymity.
References (7)

- Suppadungsuk S, Krisanapan P, Kazeminia S, et al. Hyponatremia correction and osmotic demyelination syndrome risk: a systematic review and meta-analysis. Kidney Med. 2024;7(3):100953. PubMed CrossRef
- Jha S. Catatonia in extrapontine myelinolysis: a case report and review of the literature. Ann Mov Disord. 2024;7(1):45–47. PubMed CrossRef
- Pham VM, White JM, Gunther SR, et al. Lithium for carbamazepine-induced syndrome of inappropriate antidiuretic hormone. Clin Neuropharmacol. 2021;44(1):33–34. PubMed CrossRef
- Choi HY, Chiu BMM, Wong YK, et al. Catatonia and osmotic demyelination syndrome in a patient with schizophrenia: a case report. East Asian Arch Psychiatry. 2024;34(4):147–149. PubMed CrossRef
- Morimoto M, Suzaki I, Shimakawa S, et al. Three cases in which drug-induced hyponatremia was improved by replacing carbamazepine with lacosamide. Clin Case Rep. 2020;8(7):1166–1170. PubMed CrossRef
- Lim KY, Chia YK, Khoo CS, et al. Case series of osmotic demyelination syndrome treated with plasmapheresis: experience from two tertiary hospitals. J Clin Neurol. 2022;18(1):117–119. PubMed CrossRef
- MacMillan TE, Shin S, Topf J, et al. Osmotic demyelination syndrome in patients hospitalized with hyponatremia. NEJM Evid. 2023;2(4):EVIDoa2200215. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!