Prim Care Companion CNS Disord 2021;23(1):20l02618
To cite: Manche Gowda S, Madhupreetha S, KG VK. Quetiapine for the management of tardive dyskinesia in schizoaffective disorder comorbid with diabetes mellitus and chronic kidney disease. Prim Care Companion CNS Disord. 2021;23(1):20l02618.
To share: https://doi.org/10.4088/PCC.20l02618
© Copyright 2021 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, National Institute of Mental Health and Neuro Sciences, Bengaluru, India
*Corresponding author: Shayanth Manche Gowda, MD, DNB, Department of Psychiatry, National Institute of Mental Health and Neuro Sciences, Lakkasandra, Hosur Rd, Bengaluru, Karnataka 560029, India ([email protected]).
The prevalence of tardive dyskinesia (TD) is reported to be around 30% in those taking antipsychotic medication.1 Risk factors include elderly age, prior history of acute extrapyramidal symptoms, diabetes mellitus, structural brain damage, smoking, and use of typical antipsychotics.2 Of the atypical antipsychotic medications, clozapine and olanzapine are known to have the least risk of causing TD.3 These 2 antipsychotic medications have been utilized in the management of patients with psychosis who have developed TD.4,5 However, these medications are known to worsen glycemic status,6 which limits their use in patients with concomitant diabetes. Hence, there is a need for exploration of safer options while managing TD in patients with psychosis and comorbid diabetes and renal complications. In contrast, quetiapine has less potential to worsen glycemic status. Quetiapine has also been effectively utilized in the management of tardive phenomenon in patients with psychosis.7–14 However, its safety for long-term use situations of compromised renal function is scarcely reported.
In this report, we describe the successful management of TD in a middle-aged man diagnosed with schizoaffective disorder with comorbid diabetes mellitus with renal and neurologic complications as well as primary hypothyroidism and essential hypertension.
Case Report
Mr A, a 55-year-old married man with an illness duration of 12 years and on treatment for the last 11½ years, presented with exacerbation of depressive and psychotic symptoms for 2 months in January 2019. The precipitating factor was noncompliance with his prescription of tablet risperidone 6 mg/d for 2 months prior to the date of presentation due to nonavailability in his hometown. However, he was compliant with other medications since 2016: tablet sodium valproate 1,200 mg/d, escitalopram 10 mg/d, tenepride 10 mg/d, thyroxine 50 mcg/d, aspirin 75 mg/d+atorvastatin 10 mg/d, and injection human mixtard insulin 20 units/d. The patient had been prescribed risperidone 6 mg/d for 18 months prior to presentation with which initially he had experienced extrapyramidal symptoms in the form of bilateral cogwheel rigidity and bilateral postural tremors. Thus, he had been taking tablet trihexyphenidyl 4 mg/d since then.
Detailed physical examination revealed involuntary, repetitive, irregular hyperkinetic movements of the jaw and tongue and choreoathetoid movements of the right upper limb, which was reported to be present for nearly a year. A trial of tablet diazepam 10 mg/d had been tried for a period of 3 months with no improvement in TD.
Mr A had been using nicotine in a dependence pattern for the past 20 years by smoking around 20 cigarettes per day. He was diagnosed with type 2 diabetes mellitus 10 years earlier and essential hypertension 6 years ago and had suffered a cerebrovascular accident 6 years earlier. He also had been diagnosed with chronic kidney disease and hypothyroidism 4 years earlier. Following the cerebrovascular accident, he had left-sided hemiparesis. Considering his clinical condition, Mr A was managed as an inpatient.
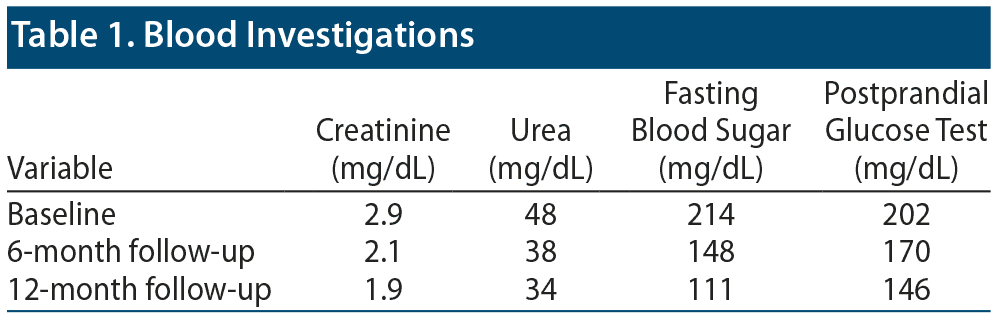
Mr A scored 22 on the Abnormal Involuntary Movement Scale (AIMS)15 at admission. His relevant blood investigations were fasting blood glucose: 214 mg/dL, postprandial blood glucose: 202 mg/dL, HbA1C: 10.8%, serum creatinine: 2.9 mg/dL, urea: 48 mg/dL, and serum valproate: 69 mcg/mL. Ultrasound of the abdomen revealed bilateral renal parenchymal disease. Routine blood investigations, electrocardiogram (EKG), and computed tomography of the brain revealed no abnormality. Endocrinology comanagement was sought for diabetes mellitus, and doses of hypoglycemic agents were optimized (tenepride 20 mg/d and injection human mixtard 10-0-20 units/d [10 units in the morning and 20 minutes at nighttime]). Repeat glycemic parameters after 5 days showed a fasting blood glucose level of 122 mg/dL and a postprandial blood glucose level of 156 mg/dL.
As Mr A had multiple medical comorbidities, the rule of starting slow and going slow was adapted. He was initiated on tablet quetiapine 50 mg uptitrated to 150 mg over a period of 6 days, with which Mr A reported complete improvement in psychotic as well as affective symptoms by day 16. He began showing improvement in TD movements by day 10 after initiation of tablet quetiapine, and on day 45, his AIMS score was 3. Follow-up at 75 days of quetiapine therapy revealed an AIMS score of zero—tardive movements had ceased. At 135-day follow-up, he had stopped taking psychotropic medications, which resulted in worsening of his psychotic and TD symptoms, with an AIMS score of 21. Thus, he was reinitiated on quetiapine, and the dose was titrated up to 150 mg/d over 1 week. He was then followed up once every 2 months for the next 12 months with the latest follow-up appointment being in February 2020, at which he reported maintained improvement in psychotic and affective symptoms and TD movements (AIMS score of zero). Mr A tolerated tablet quetiapine well during the entire period of 1-year period with no adverse effects, including a repeat EKG that was within normal limits.
Discussion
The mechanisms of action of antipsychotic medications for the treatment of TD are still unclear. Quetiapine’s kiss and run mechanism—fast dissociation due to low affinity for dopamine receptors, which is responsible for lesser propensity to cause TD, may also explain its role in improvement of TD movements.7–9 As quetiapine is metabolized mostly in the liver, it is considered relatively safe, and dose adjustment in renal impairments is not routinely recommended. However, Yamada et al16 reported acute kidney injury in a 73-year-old man with congestive heart failure and pulmonary complication. He was prescribed quetiapine up to 150 mg for managing acute delirium post valvular repair. Establishing causality is difficult, as this individual had complex multisystem dysfunction with several other concurrent prescriptions.16 He et al17 reported interstitial nephritis developing during quetiapine use (up to 200 mg) for psychotic depression. This 27-year-old man redeveloped features of nephritis after switching over to olanzapine at a dose of 20 mg/d, implicating dibenzodiazepine derivatives quetiapine and olanzapine in the development of chronic interstitial nephritis.17 In the present case, the patient had multiple risk factors that might have contributed to the development of TD such as nicotine dependence syndrome, structural brain damage, diabetes mellitus, and initial extrapyramidal symptoms with risperidone, and, interestingly, the patient’s affective and especially psychotic symptoms resolved with subtherapeutic doses of quetiapine. We were successful in managing TD with quetiapine with no worsening of renal function. A strong temporal correlation was observed, indicating the role of quetiapine in the management of TD. When Mr A was taking quetiapine, TD improved significantly, whereas during the period of nonadherence to psychotropics including quetiapine due to poor insight, he experienced worsening of TD. However, reintroduction of quetiapine during follow-up resulted in improvement of TD. We are able to report that 12 months of quetiapine use has shown sustained improvement in TD without worsening pre quetiapine renal functioning as shown in Table 1. Our experience in this case leads us to believe that quetiapine is safe in patients with diabetes mellitus and renal disease, while being effective in managing TD. We conclude that quetiapine could be a safer alternative to clozapine and olanzapine for the management of TD. We hope that larger studies or studies with larger samples will clarify these findings.
Published online: February 18, 2021.
Potential conflicts of interest: None.
Funding/support: None.
Acknowledgments: Sharad Philip, MD, has contributed by proofreading this manuscript. Dr Philip reports no conflicts of interest related to the subject of this report.
Patient consent: Informed consent was obtained from the patient and his family members to publish this case report, and information has been de-identified to protect anonymity.
References (17)

- Llorca P-M, Chereau I, Bayle F-J, et al. Tardive dyskinesias and antipsychotics: a review. Eur Psychiatry. 2002;17(3):129–138. PubMed CrossRef NLM
- Solmi M, Pigato G, Kane JM, et al. Clinical risk factors for the development of tardive dyskinesia. J Neurol Sci. 2018;389:21–27. PubMed CrossRef NLM
- Carbon M, Kane JM, Leucht S, et al. Tardive dyskinesia risk with first- and second-generation antipsychotics in comparative randomized controlled trials: a meta-analysis. World Psychiatry. 2018;17(3):330–340. PubMed CrossRef NLM
- Kinon BJ, Jeste DV, Kollack-Walker S, et al. Olanzapine treatment for tardive dyskinesia in schizophrenia patients: a prospective clinical trial with patients randomized to blinded dose reduction periods. Prog Neuropsychopharmacol Biol Psychiatry. 2004;28(6):985–996. PubMed CrossRef NLM
- Mentzel TQ, van der Snoek R, Lieverse R, et al. Clozapine monotherapy as a treatment for antipsychotic-induced tardive dyskinesia: a meta-analysis. J Clin Psychiatry. 2018;79(6):17r11852. PubMed CrossRef NLM
- Huhn M, Nikolakopoulou A, Schneider-Thoma J, et al. Comparative efficacy and tolerability of 32 oral antipsychotics for the acute treatment of adults with multi-episode schizophrenia: a systematic review and network meta-analysis. Lancet. 2019;394(10202):939–951. PubMed CrossRef NLM
- Alptekin K, Kivircik BB. Quetiapine-induced improvement of tardive dyskinesia in three patients with schizophrenia. Int Clin Psychopharmacol. 2002;17(5):263–264. PubMed CrossRef NLM
- Farah A. Reduction of tardive dyskinesia with quetiapine. Schizophr Res. 2001;47(2–3):309–310. PubMed CrossRef NLM
- Vesely C, Küfferle B, Brücke T, et al. Remission of severe tardive dyskinesia in a schizophrenic patient treated with the atypical antipsychotic substance quetiapine. Int Clin Psychopharmacol. 2000;15(1):57–60. PubMed CrossRef NLM
- Nelson MW, Reynolds RR, Kelly DL, et al. Adjunctive quetiapine decreases symptoms of tardive dyskinesia in a patient taking risperidone. Clin Neuropharmacol. 2003;26(6):297–298. PubMed CrossRef NLM
- Mohapatra S. Successful management of tardive dyskinesia with quetiapine and clonazepam in a patient of schizophrenia with type 2 diabetes mellitus. Clin Psychopharmacol Neurosci. 2016;14(2):218–220. PubMed CrossRef NLM
- Gourzis P, Polychronopoulos P, Papapetropoulos S, et al. Quetiapine in the treatment of focal tardive dystonia induced by other atypical antipsychotics: a report of 2 cases. Clin Neuropharmacol. 2005;28(4):195–196. PubMed CrossRef NLM
- Emsley R, Turner HJ, Schronen J, et al. A single-blind, randomized trial comparing quetiapine and haloperidol in the treatment of tardive dyskinesia. J Clin Psychiatry. 2004;65(5):696–701. PubMed CrossRef NLM
- Bressan RA, Jones HM, Pilowsky LS. Atypical antipsychotic drugs and tardive dyskinesia: relevance of D2 receptor affinity. J Psychopharmacol. 2004;18(1):124–127. PubMed CrossRef NLM
- Abnormal Involuntary Movement Scale (117-AIMS). In: Guy W, ed. ECDEU Assessment Manual for Psychopharmacology. Rockville, MD: National Institute of Mental Health; 1976:534–537.
- Yamada H, Katsumori Y, Kawano M, et al. Quetiapine-related acute kidney injury requiring transient continuous hemodiafiltration. Intern Med. 2018;57(12):1763–1767. PubMed CrossRef NLM
- He L, Peng Y, Fu X, et al. Dibenzodiazepine derivative quetiapine- and olanzapine-induced chronic interstitial nephritis. Ren Fail. 2013;35(5):657–659. PubMed CrossRef NLM
Enjoy this premium PDF as part of your membership benefits!


