Abstract
Importance: At present, the diagnoses of many psychiatric and neurological disorders are based on clinical symptoms and behaviors, neglecting the laboratory test indicators. This narrative review assessed the existing published literature on the role of microRNAs (miRNAs) in psychiatric and neurological disorders.
Observations: The miRNAs are involved in numerous biological processes, and their dysregulation can lead to neuropsychiatric disorders. They also play a prominent role in neurotransmitter signaling pathways, and their impairment can lead to psychiatric disorders. The miRNAs act as key regulators of neuronal functions such as neuroplasticity, differentiation, and protection of neurons and also play a role in establishing memory and cognition. Neurodegenerative processes therefore involve miRNAs as potential targets for treatment and early detection. The functional regulation of miRNAs of several genes has made miRNAs potent targets for therapeutic development.
Conclusions and Relevance: miRNAs have the potential to be used as biomarkers for diagnosis and treatment response in psychiatric and neurological disorders.
Prim Care Companion CNS Disord 2026;28(2):25nr04069
Author affiliations are listed at the end of this article.
The microRNAs (miRNAs) are small, noncoding RNA molecules that predominantly function as posttranscriptional regulators of gene expression. By binding to specific sequences on messenger RNA (mRNA), they can prompt mRNA degradation or inhibit its translation, thereby reducing the expression of the target gene. This mechanism effectively serves as a “molecular dimmer switch,” finely tuning protein levels within the cell.1 Furthermore, miRNAs potentially act as a bridge between environmental factors and alterations in gene expression. These molecules may be implicated in the development of psychiatric and neurological disorders, and there is genetic evidence that miRNA genes, as well as their targets, are associated with such disorders. In individuals with psychiatric and neurological disorders, miRNA expression levels are found to be altered in brain tissues, cerebrospinal fluid (CSF), and circulating blood. Hence, miRNAs are emerging as potential biomarkers for diagnosing and predicting psychiatric disorders, as well as evaluating treatment responses.2 Some miRNAs show consistent dysregulation across brain tissue, CSF, and peripheral blood, reinforcing their reliability as biomarkers that reflect central nervous system pathology.
This review explores the complex roles of miRNAs in a range of neuropsychiatric disorders including Alzheimer disease (AD), Parkinson disease (PD), frontotemporal dementia (FTD), brain injuries, amyotrophic lateral sclerosis (ALS), multiple sclerosis (MS), major depressive disorder (MDD), posttraumatic stress disorder (PTSD), bipolar disorder (BPD), schizophrenia (SCZ), and anxiety. This narrative review highlights how miRNA dysregulation can shed light on the molecular mechanisms underlying these conditions and serves as a potential biomarker for early diagnosis and prognosis. Finally, it emphasizes the promise of miRNAs in advancing precision neuropsychiatry by enabling prediction of treatment response and improving disease management.
miRNAs AS CLINICAL BIOMARKERS IN NEUROPSYCHIATRIC ILLNESSES
Role of miRNAs in Neurological Disorders
The miRNAs participate in a variety of biological processes, including neurogenesis, neuronal differentiation, synaptic plasticity, apoptosis, and neuronal metabolism. Their dysregulation can contribute to neurological disorders.3 They have been linked to both neurodegenerative and non-neurodegenerative neurological conditions.3,4
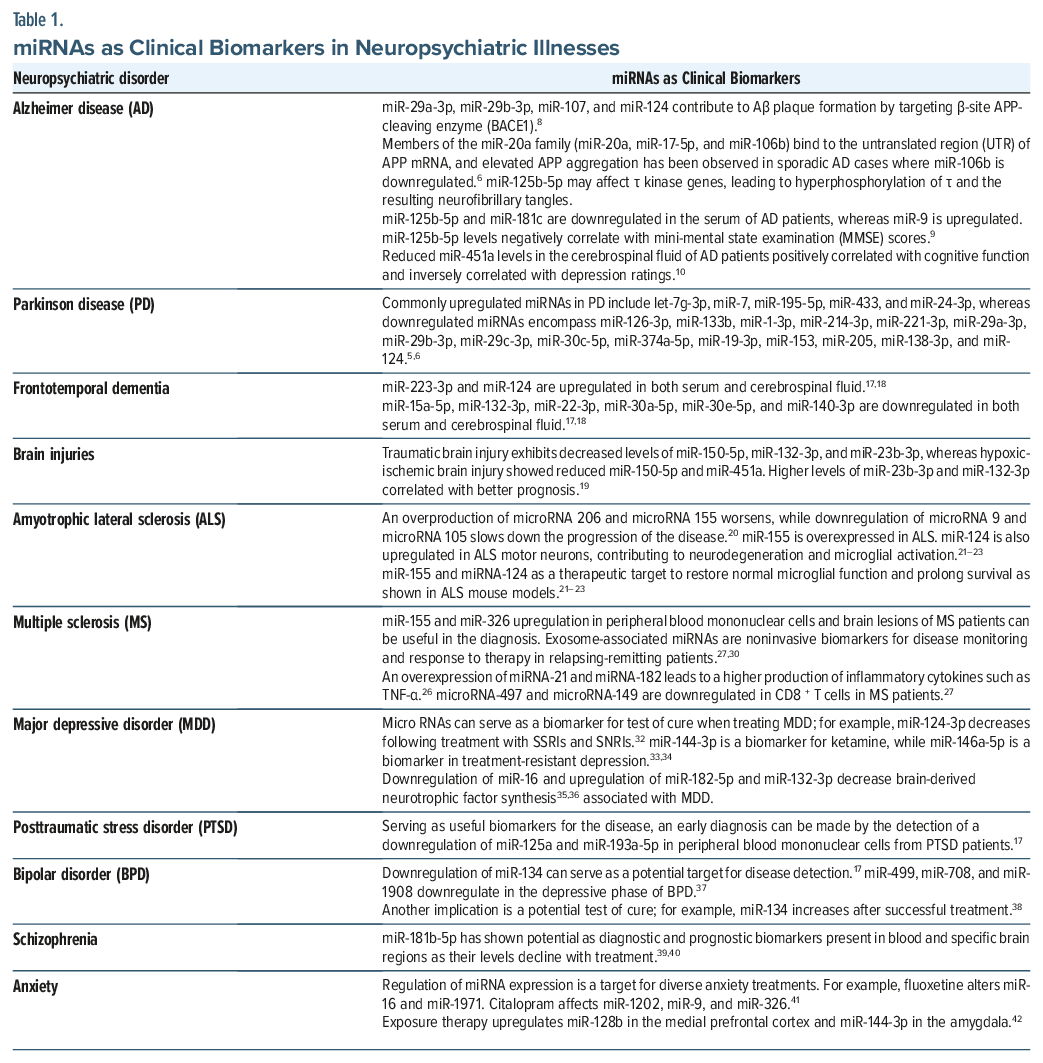
Alzheimer disease. AD is one of the most frequently occurring neurodegenerative disorders, typified by extracellular amyloid-β (Aβ) plaques and neuronal hyperphosphorylated τ neurofibrillary tangles. While these protein aggregates are late-stage markers, with limited sensitivity and specificity compared to blood-based markers, they are considered highly accurate when assessed via PET imaging or CSF analysis.5 Notably, miRNAs have demonstrated promise as potential early-stage indicators of AD, given their involvement in neuroinflammatory, cognitive, and protein aggregation pathways, among others, that underlie AD-related neurodegeneration.6,7
Multiple circulating miRNAs are reported to be either upregulated or downregulated in relation to AD, including let-7f-5p, let-7g-5p, miR-103a-3p, miR-107, miR-106b, miR-125b-5p, miR-127-3p, miR-135a-5p, miR-136-3p, miR-137, miR-138-5p, miR-142-3p, miR-143-3p, miR-146a-5p, miR-151a-3p, miR-15a-5p, miR-15b-5p, miR-181c-5p, miR-191-5p, miR-26b-5p, miR-29a-3p, miR-29b-3p, miR-342-3p, miR-34a-5p, miR-451a, and miR-9-5p.5,6
Research indicates that miRNAs influence AD-related protein aggregation by modulating specific cleavage enzymes. For example, miR-29a-3p, miR-29b-3p, miR-107, and miR-124 contribute to Aβ plaque formation by targeting β-site amyloid precursor protein–cleaving enzyme.8 Likewise, members of the miR-20a family (miR-20a, miR-17-5p, and miR-106b) bind to the untranslated region of APP mRNA, and elevated APP aggregation has been observed in sporadic AD cases where miR-106b is downregulated.6 Another miRNA, miR-125b-5p, may affect τ kinase genes, leading to hyperphosphorylation of τ and the resulting neurofibrillary tangles. Specifically, miR-125b-5p and miR-181c are downregulated in the serum of AD patients, whereas miR-9 is upregulated; miR-125b-5p levels negatively correlate with Mini-Mental State Examination scores.9 A recent study by Feng and colleagues10 found that reduced miR-451a levels in the CSF of AD patients positively correlated with cognitive function and inversely correlated with depression ratings.
Parkinson disease. PD is the second most common progressive neurodegenerative disorder, characterized by striatal dopamine deficiency, dopaminergic neuronal loss in the substantia nigra pars compacta, gliosis, and the accumulation of synuclein-alpha (SNCA) and ubiquitin-positive Lewy bodies.11 Key genes implicated in these pathological processes include SNCA, leucine-rich repeat kinase 2 (LRRK2), fibroblast growth factor 20 (FGF20), PTEN-induced serine-threonine kinase 1 (PINK1), Parkin, and disease J protein 1 (DJ-1), most of which are regulated by miRNAs.6
Commonly upregulated miRNAs in PD include let-7g-3p, miR-7, miR-195-5p, miR-433, and miR-24-3p, whereas downregulated miRNAs encompass miR-126-3p, miR-133b, miR-1-3p, miR-214-3p, miR-221-3p, miR-29a-3p, miR-29b-3p, miR-29c-3p, miR-30c-5p, miR-374a-5p, miR-19-3p, miR-153, miR-205, and miR-124.5 Reduced expression of miR-124 correlates with dopaminergic neuronal loss and neuroinflammation in PD.12 miR-30c-5p, which is abundant in neurons and glial cells, influences the ubiquitination pathway of SNCA to confer neuroprotection, but this effect is diminished when miR-30c-5p levels are decreased.13 Both miR-7 and miR-153 regulate SNCA, thereby affecting SNCA expression and cellular processes linked to oxidative stress and neuroinflammation.14 In addition, miR-205 and miR-138-3p interact with the LRRK2 gene, and their downregulation elevates LRRK2 activity and protein aggregates.6 LRRK2 mutations are also associated with decreased miR-19b-3p, which influences the extracellular matrix and adipocytokine pathways in PD.15 Disruption of a binding site for miR-433 has been linked to increased expression of FGF20 and higher SNCA levels.16 Additional miRNAs such as miR-27a/b and miR-140 for PINK1; miR-218, miR-103a-3p, and miR-181a-5p for Parkin; and miR-494, miR-874, miR-4639-5p, and miR-145-3p for DJ-1 are also associated with PD pathogenesis.6
Frontotemporal dementia. FTD is a neurodegenerative condition marked by atrophy and other pathological changes in the frontal and temporal lobes, leading to disruptions in behavior, language, and executive function.17 It is primarily categorized into behavioral-variant FTD (bvFTD), primary progressive aphasia, and movement disorders.17 FTD pathology may involve genetic alterations in fused in sarcoma (FUS) and TAR DNA-binding protein 43 (TDP-43), which can disrupt miRNA regulation and expression. In bvFTD, miR-223-3p is upregulated while miR-15a-5p is downregulated in both serum and CSF. Additionally, miR-132-3p is consistently reduced in frontotemporal degeneration and dementia relative to healthy controls. In one CSF-based comparison between bvFTD and AD, miR-22-3p, miR-30a-5p, miR-30e-5p, and miR-140-3p were all significantly decreased in bvFTD.18 Moreover, bvFTD patients exhibit lower serum and CSF levels of miR-22-3p compared to AD subjects and healthy individuals. Conversely, miR-124 is linked to social behavior in bvFTD and is elevated in the CSF of bvFTD patients relative to AD and healthy controls.17
Brain injuries. Several miRNAs have been studied in the context of traumatic brain injury (TBI) and hypoxic-ischemic brain injury (HIBI), notably miR-150-5p, miR-132-3p, miR-23b-3p, miR-451a, and miR-16-5p. miR-132-3p, miR-23b-3p, and miR-451a are implicated in inflammatory and apoptotic pathways associated with ischemic and perfusion insults. At baseline, TBI patients exhibit decreased levels of miR-150-5p, miR-132-3p, and miR-23b-3p, whereas in HIBI, reduced miR-150-5p and miR-451a are observed. Higher levels of miR-23b-3p and miR-132-3p appear to correlate with better prognosis.19
Amyotrophic lateral sclerosis. ALS is a relatively common neurodegenerative disease characterized by a progressive loss of motor neurons leading to muscular atrophy and weakness,20 and recent studies have discovered an expression of more than 30 miRNAs in the muscles of patients who develop ALS.20 There is an overproduction of miRNA 206 and miRNA 155, whereas a downregulation of miRNA 9 and miRNA 105 has been shown to slow down the progression of the disease.20 These discoveries have led to the impression that miRNAs can be a potential diagnostic tool and a therapeutic target for the disease.
miRNAs are involved in the regulation of gene expression and RNA metabolism, which are critical in ALS. Dysregulation of miRNAs such as miR-129-5p, miR-155, and miR-124 has been implicated in ALS. For instance, miR-129-5p is upregulated in SOD1-linked ALS models and sporadic ALS patients, leading to the downregulation of the RNA-binding protein ELAVL4/HuD, which is essential for neuronal function. Similarly, miR-155 is overexpressed in ALS, and targeting it can restore normal microglial function and prolong survival in ALS mouse models. miR-124 is also upregulated in ALS motor neurons, contributing to neurodegeneration and microglial activation.21–23
miRNAs hold promise as biomarkers for ALS. For example, miR-206 and miR-338-3p have been identified as potential plasma-based biomarkers for sporadic ALS, with differential expression patterns that can aid in diagnosis. Additionally, miR-129-5p and miR-23a-3p have been found to be upregulated in peripheral blood cells and plasma exosomes of ALS patients, respectively, suggesting their utility in noninvasive diagnostic tests.24,25
miRNA-based therapies are being explored, although they are still in the experimental stage. Antisense oligonucleotides (ASOs) targeting specific miRNAs, such as miR-129-5p and miR-155, have shown promising results in preclinical models. Inhibition of miR-129-5p with ASOs improved survival and neuromuscular function in ALS mouse models. Similarly, targeting miR-155 with anti-miR-155 inhibitors prolonged survival and ameliorated disease in SOD1 mice.21,22 In summary, miRNAs are crucial in the pathogenesis of ALS, serve as potential diagnostic biomarkers, and are being investigated as therapeutic targets.
Multiple sclerosis. MS is a chronic demyelinating disease that results in the degeneration of neurons with a higher proportion of the patient population being young females.26 The disease is characterized by optic neuritis, sensory and cognitive deficits, and neuronal dysfunction leading to progressive disability.26 An overexpression of miRNAs, such as miRNA-21 and miRNA-182, has been discovered that may lead to a higher production of inflammatory cytokines such as TNF alpha.26 A study by Ma et al27 found that miRNA-497 and miRNA-149 are downregulated in CD8+ T cells in patients with MS.
miRNAs are involved in regulating immune responses, which are central to MS pathogenesis. Dysregulated miRNAs such as miR-155, miR-326, and miR-21 have been implicated in the disease. For instance, miR-155 is upregulated in MS and contributes to the activation of pathogenic immune cells and the permeability of the blood-brain barrier. miR-326 is involved in the differentiation of Th17 cells, which are crucial in MS pathogenesis. Additionally, miR-142-3p has been linked to synaptopathy and disease progression in MS.28,29
miRNAs have potential as biomarkers for MS. miR-155 and miR-326 are upregulated in peripheral blood mononuclear cells and brain lesions of MS patients, suggesting their utility in diagnosis. Exosome-associated miRNAs, such as those identified in serum and CSF, can also serve as non-invasive biomarkers for disease monitoring and therapy response. For example, a panel of exosome-associated miRNAs has been proposed to monitor response to interferon-β therapy in relapsing-remitting MS patients.27,30
miRNA-based therapies are being explored for MS. Strategies include miRNA inhibition therapy using ASOs, locked nucleic acids (LNAs), and antagomirs to target dysregulated miRNAs like miR-155 and miR-326. Additionally, miRNA restoration therapy using miRNA mimics is under investigation. Current disease-modifying treatments, such as interferon-β and glatiramer acetate, may exert their effects partly by modulating miRNA expression. For instance, dimethyl fumarate has been shown to ameliorate disease progression by targeting miR-142-3p.31 In summary, miRNAs are crucial in the pathogenesis of MS, serve as potential diagnostic biomarkers, and are being investigated as therapeutic targets (Table 1).
Role of miRNAs in Psychiatric Disorders
Psychiatric disorders are shaped by various forms of neural dysregulation, including inflammation, oxidative stress, and abnormalities in neurotrophic factors, neurotransmitters, neuroplasticity, and synaptic function. Epigenetic changes affecting DNA, histones, RNA, and miRNAs further contribute to these conditions.43 Among the miRNAs most commonly implicated in disorders such as MDD, SCZ, and BPD are miR-34a, miR-106, miR-132, and miR-134. These specific miRNAs play significant roles in neurotransmitter signaling pathways, which are critically disrupted in these psychiatric conditions.44
Major depressive disorder. The miRNAs are central to modulating many of the neuronal dysregulatory processes in depression. This includes the regulation of serotonin and brain-derived neurotrophic factor (BDNF), both of which are pivotal in depression pathophysiology. By influencing BDNF levels, miRNAs indirectly affect neurogenesis, synaptic plasticity, and mood-related behaviors. In particular, miR-206, known to be involved in various neuropsychiatric disorders (including depression), has been shown to significantly impact BDNF expression.45 The miRNAs also appear to regulate the brain-gut-microbiome axis, another important factor in depression.43 Frequently altered miRNAs in MDD include miR-24-3p, miR-124-3p, miR-132-3p, miR-182-5p, miR-345-5p, miR-425-3p, miR-636, and several let-7 family members.2,5
Because BDNF is reduced in depression, any miRNA that affects its expression is especially relevant. Downregulation of miR-16 and upregulation of miR-182-5p and miR-132-3p have each been linked to decreased BDNF synthesis.35,36 Elevated levels of miR-132-3p have also been positively correlated with Self-Rating Depression Scale (SDS) scores, Hamilton Anxiety Rating Scale scores, and impaired visual memory function in MDD.35,46
Increased miR-124-3p levels have been documented in both serum and postmortem brain tissue of MDD patients. This miRNA is associated with neurogenesis, neuronal growth, and synaptic plasticity, which are key processes disrupted in MDD. Notably, miR-124-3p decreases following treatment with antidepressants such as selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs) like venlafaxine and duloxetine, and mirtazapine.32 In addition, miR-144-3p has emerged as a potential therapeutic biomarker for ketamine and miR-146a-5p for repetitive transcranial magnetic stimulation (rTMS) in treatment-resistant depression.33,34
Posttraumatic stress disorder. Alterations in epigenetic regulation, including changes in miRNA expression, may underlie the immunological dysregulation observed in PTSD. Common blood-based miRNA biomarkers in PTSD include whole-blood miR-3130-5p, plasma-derived miR-18a-3p and miR-7-1-5p, peripheral blood mononuclear cell (PBMC)-associated miR-125a, miR-181c, miR-193a-5p, miR-671-5p, and miR-7113-5p, as well as exosomal miR-4488, miR-5100, miR-7641, miR-203a-3p, miR-10b-5p, miR-502-3p, and miR-874-3p.47
Variants in miRNAs that target the FK506-binding protein 5 gene have been linked to PTSD and other stressor-induced psychiatric disorders.48 In particular, the downregulation of miR-125a and miR-193a-5p in PBMCs from PTSD patients is associated with increased levels of proinflammatory cytokines.47
Bipolar disorder. Extensive research underscores the importance of miRNAs in the neurophysiology, diagnosis, and treatment of BPD. Specifically, miR-34a, miR-378a-5p, miR-30d-5p, miR-30e, miR-21-3p, miR-140-3p, miR-330-5p, miR-134, and miR-137 have been frequently implicated.2,6,47,49 miRNA expression alterations in BPD have been investigated in multiple sample types, including whole blood, plasma, exosomes, monocytes, and lymphoblastoid cell lines as well as across different brain regions such as the prefrontal cortex, hippocampus, anterior cingulate, cerebellum, and amygdala.49
BPD is particularly associated with disturbances in synaptic formation, growth, and plasticity. One notable example is miR-134, which helps regulate dendritic spine growth and synapse formation; downregulation of miR-134 has been reported in manic patients relative to healthy controls.17 In a study by Rong and colleagues,38 baseline levels of miR-134 increased following combined therapy with an antipsychotic and a mood stabilizer, suggesting that miR-134 could serve as both a diagnostic and prognostic biomarker in bipolar mania. Similarly, miR-499, miR-708, and miR-1908 were found to be downregulated in the depressive phase of BPD.37 Conversely, miR-29a-3p, miR-106a-5p, miR-125a-3p, miR-106b-5p, miR-125b-5p, and miR-9-5p exhibit upregulation during manic or euthymic phases; miR-125a-3p and miR-107 are notably increased in the manic phase compared to the euthymic phase.50 Beyond their diagnostic and prognostic value, certain miRNAs such as miR-34a, miR-34b, miR-34c, miR-378a-5p, miR-19b-3p, miR-30d-5p, miR-21-3p, miR-140-3p, and miR-330-5p may distinguish BPD from MDD and SCZ.47
Schizophrenia. In SCZ, miRNAs have been implicated in modulating synaptic plasticity, neuroinflammation, neuronal connectivity, and both neurotrophic and neurotransmitter signaling pathways. Several promising candidate miRNAs for SCZ include miR-7-5p, miR-181-5p, miR-193-3p, miR-218-5p, miR-675-3p, miR-1262, miR-34a-5p, miR-130b, and miR-30e-5p.33,51 In particular, miR-181b, miR-30e, miR-34a, and miR-7 are all found to be upregulated in SCZ. Of these, miR-181b-5p has shown potential as a diagnostic biomarker present in both blood and specific brain regions (eg, the dorsolateral prefrontal cortex and superior temporal gyrus); it also appears to serve as a prognostic marker, declining with antipsychotic treatment and correlating positively with improvements in negative symptoms.39,40
Rather than focusing on a single miRNA, recent studies suggest that specific miRNA combinations may provide better diagnostic precision. Examples include miR-22-3p + miR-92a-3p + miR-137, miR-432-5p + miR-449a, and miR-34a-5p + miR-432-5p + miR-449a.52,53
Anxiety. Anxiety disorders arise from changes in gene expression influencing synaptic density, brain plasticity, neurotrophin (BDNF) levels, neuronal signal processing, neurotransmission, hypothalamic-pituitary-adrenal axis function, neurogenesis, myelination, and overall neuronal development.54 Key miRNAs that modulate these functions include miR-15a, miR-17-92, miR-34, miR-101, miR-124, miR-135, and miR-155.41
Regulation of miRNA expression is also observed in response to diverse anxiety treatments. For instance, fluoxetine therapy alters miR-16 levels in the raphe nucleus and locus coeruleus, as well as miR-1971 in the medial prefrontal cortex. Citalopram and escitalopram treatment affects miR-1202 in the medial prefrontal cortex and miR-9 and miR-326 in the nucleus accumbens, respectively.41 Notably, fear extinction or exposure therapy is linked to the upregulation of miR-128b in the medial prefrontal cortex and miR-144-3p in the amygdala, with miR-128b overexpression itself associated with reduced fear.42 Environmental enrichment, another therapeutic approach, has been shown to elevate miR-124 and miR-183 in the amygdala 30 days posttreatment, alongside increased miR-124 and miR-132 in the hippocampus, which corresponds to enhanced neurogenesis.55,56
USE OF miRNAs IN THE TREATMENT OF NEUROPSYCHIATRIC DISORDERS
miRNAs are pivotal regulators of neuronal functions such as neuroplasticity, differentiation, and neuronal protection, and they also contribute to memory and cognition.57 Because of these essential roles, miRNAs have become potential targets for both early detection and treatment of neurodegenerative processes. For example, abnormal let-7 miRNA activity has been linked to cognitive decline in Alzheimer dementia through its impact on neuronal regeneration.57 AD may additionally involve the downregulation of miRNA-132 and miRNA-212.57 In FTD, a mutation in miRNA 34a-5p can lead to behavioral deficits,57 whereas reverse sequence inhibition of miRNA 18a-5p and let-7a-5p has been tied to vascular dementia.
In Huntington disease, downregulated miRNA-128-a normally binds to the mutated huntingtin gene; once reduced, transcriptional errors arise, ultimately causing the disease.57 Meanwhile, in BPD, miRNA-579 is upregulated, and miRNA-34a is downregulated.2 In SCZ, miRNA-132 modulation has been associated with increased expression of several genes.2 miRNAs may also drive depressive pathology by altering neurogenesis in hippocampal neurons,2 and identifying these miRNAs could offer new diagnostic tools and possible therapeutic targets for treating depressive symptoms.2
ALS involves neuronal degeneration of motor neurons leading to progressive muscle weakness. Freischmidt and colleagues58 discovered miRNA sequences being downregulated in ALS. Other studies have found an associated upregulation of miRNAs,59 such as an increase in miRNA 155 leading to the progression of ALS in some patients.20 Therefore, 2 proposed therapeutic interventions for the treatment of ALS are “antisense technology” to block the upregulation and “replacement therapy” to correct the deficiency in gene expression in case there is a downregulation of miRNAs in patients59 with the delivery to host cells achieved via retroviruses, adenoviruses, or a lipid-based system.
Some proposed therapeutic targets for MS patients are miRNA-155, miRNA-326, and miRNA-12.27 The target miRNAs are overexpressed in patients with MS, and suppression may lead to inhibition of T-helper cell production and downregulation of interleukins, thereby decreasing the inflammation associated with disease progression and flare ups.27
The functional regulation of miRNAs across multiple genes makes them compelling targets for therapeutic development.60 This therapeutic strategy may involve oligonucleotides designed to modulate miRNA function, particularly for managing loss-of-function or gain-of-function mutations.60 Potential agents include miRNA mimics, aimed at enhancing miRNA function in conditions where they are downregulated, such as dementia or miRNA antagonists that counteract upregulated miRNAs.60 Nonetheless, numerous challenges remain, especially in central nervous system drug development.60 These obstacles involve the blood-brain barrier, potential off-target effects influencing multiple genes, and subtle shifts in gene expression (eg, overexpression of the target gene).60
Another important class of nucleic acids that may serve as therapeutic interventions targeting miRNAs includes ASOs and small interfering RNAs (siRNAs).61 ASOs induce the degradation of miRNA targets often via cleavage or alternative splicing, while siRNAs guide “RNA-induced silencing complexes” to suppress protein expression.61 Both ASOs and siRNAs can be delivered into cells using viral vectors or lipid-based nanoparticle systems.61
Bioinformatic analyses have identified specific miRNAs and corresponding protein targets implicated in psychiatric disorders.62 For instance, disruptions in the Golgi apparatus may undermine neuronal connectivity, while overexpression of miRNA-185 can potentially restore dendritic formation. Further experimental research is required to elucidate the roles of miRNA-185 in the pathophysiology and treatment of SCZ.62
Some potential drugs can mimic miRNAs that enhance the function, especially in psychiatric illnesses that involve the downregulation of miRNAs such as Alzheimer dementia,60 or those that act as antagonists of miRNAs. However, several challenges would need to be addressed along the drug development process especially for central nervous system formulations. Blood-brain barrier penetration, an effect on several genes leading to an “off-target effect,”60 and subtle changes in the specific target gene, particularly overexpression of target genes, may pose a potential challenge for clinicians when developing interventions regarding this class of drugs.
Despite these promising avenues, challenges persist in applying miRNA-based therapies to neuropsychiatric conditions. Many studies focus on singular brain regions linked to specific disorders, complicating the development of broad-spectrum interventions.62 Additionally, since miRNAs can interact with multiple target genes, understanding these interactions could offer deeper insights into their therapeutic potential.62 Discovering the interaction of miRNAs with many target genes can provide further insight into the role miRNAs may play as a potential target for many treatment modalities in a vast variety of neuropsychiatric disorders62 in the future.
CONCLUSION
In summary, miRNAs are pivotal regulators of gene expression in neurodevelopment, synaptic plasticity, stress responses, and neuronal integrity. Their dysregulation is implicated in a range of neuropsychiatric and neurodegenerative disorders, pointing to the potential of miRNAs both as biomarkers and therapeutic targets. Despite promising discoveries, challenges remain, such as ensuring blood-brain barrier penetration, avoiding off-target effects, and accounting for sex differences in miRNA expression. Ongoing interdisciplinary research and technological advances in delivery methods offer promising avenues for translating miRNA insights into personalized diagnostics and treatments for neuropsychiatric conditions. Large-scale multicenter validation studies are needed before routine clinical implementation.
Article Information
Published Online: April 30, 2026. https://doi.org/10.4088/PCC.25nr04069
© 2026 Physicians Postgraduate Press, Inc.
Submitted: August 27, 2025; accepted December 1, 2025.
To Cite: Khan S, Sayana P, Oluwanifesimi OL, et al. The role of microRNAs as biomarkers in neuropsychiatric disorders. Prim Care Companion CNS Disord 2026;28(2):25nr04069.
Author Affiliations: PPD Thermofisher, Austin, Texas (Khan); University of Alabama, Huntsville, Alabama (Sayana); Department of Psychiatry, University of Chicago, Chicago, Illinois (Oluwanifesimi); Mcmaster University Health Sciences, Hamilton, Ontario, Canada (Waseem); Department of Psychiatry, Wake Forest University, Winston-Salem, North Carolina (Shah); Department of Biomedical Science, University of Houston, Texas (Sheikh); Department of Psychiatry, Yale New Haven School of Medicine, New Haven, Connecticut (Mansuri); Department of Psychiatry, Texas Tech University Health Science Center at Permian Basin, Midland, Texas (Yadav).
Corresponding Author: Sadaf Khan, MBBS, PPD Thermofisher, 7551 Metro Center Dr, Austin, TX 78744 ([email protected]).
Financial Disclosure: None.
Funding/Support: None.
Clinical Points
- MicroRNAs (miRNAs) have emerged as promising clinical biomarkers for neuropsychiatric disorders due to their diagnostic, prognostic, and therapeutic potential; miRNAs are detectable in peripheral biofluids such as serum, plasma, cerebrospinal fluid, and saliva, providing minimally invasive biomarkers for clinical use.
- Specific miRNAs show distinct expression patterns in patients vs. healthy controls, enabling early and differential diagnosis of disorders such as schizophrenia, depression, Alzheimer disease, and autism.
- Certain circulating miRNAs correlate with symptom severity and cognitive dysfunction, allowing them to serve as objective measures of disease progression or clinical status.
- miRNA profiles can predict treatment response to antidepressants, antipsychotics, and other neuropsychiatric therapies.
References (62)

- MacFarlane L, Murphy P. MicroRNA: biogenesis, function and role in cancer. Curr Genomics. 2010;11(7):537–561. PubMed CrossRef
- Roy B, Yoshino Y, Allen L, et al. Exploiting circulating MicroRNAs as biomarkers in psychiatric disorders. Mol diagnosis Ther. 2020;24(3):279–298. PubMed CrossRef
- Juźwik CA, Drake SS, Zhang Y, et al. microRNA dysregulation in neurodegenerative diseases: a systematic review. Prog Neurobiol. 2019;182:101664. PubMed CrossRef
- Vieira AS, Dogini DB, Lopes-Cendes I. Role of non-coding RNAs in non-aging-related neurological disorders. Braz J Med Biol Res = Revista brasileira de pesquisas medicas e Biol. 2018;51(8):e7566. PubMed CrossRef
- Van den Berg MMJ, Krauskopf J, Ramaekers JG, et al. Circulating microRNAs as potential biomarkers for psychiatric and neurodegenerative disorders. Prog Neurobiol. 2020;185:101732. PubMed CrossRef
- Khoodoruth MAS, Khoodoruth WNC, Uroos M, et al. Diagnostic and mechanistic roles of MicroRNAs in neurodevelopmental & neurodegenerative disorders. Neurobiol Dis. 2024;202:106717. PubMed CrossRef
- Liu W, Rao X, Sun W, et al. The neuroinflammatory role of microRNAs in Alzheimer’s disease: pathological insights to therapeutic potential. Mol Cel Biochem. 2024;20(5):2689–2706. PubMed CrossRef
- Fang M, Wang J, Zhang X, et al. The miR-124 regulates the expression of BACE1/β-secretase correlated with cell death in Alzheimer’s disease. Toxicol Lett. 2012;209(1):94–105. PubMed CrossRef
- Tan L, Yu JT, Liu QY, et al. Circulating miR-125b as a biomarker of Alzheimer’s disease. J Neurol Sci. 2014;336(1–2):52–56. PubMed CrossRef
- Feng H, Hu P, Chen Y, et al. Decreased miR-451a in cerebrospinal fluid, a marker for both cognitive impairment and depressive symptoms in Alzheimer’s disease. Theranostics. 2023;13(9):3021–3040. PubMed CrossRef
- Le W, Sayana P, Jankovic J. Animal models of Parkinson’s disease: a gateway to therapeutics? Neurotherapeutics. 2014;11(1):92–110. PubMed CrossRef
- Yao L, Zhu Z, Wu J, et al. MicroRNA-124 regulates the expression of p62/p38 and promotes autophagy in the inflammatory pathogenesis of Parkinson’s disease. FASEB J official Publ Fed Am Soc Exp Biol. 2019;33(7):8648–8665. PubMed CrossRef
- Martins M, Rosa A, Guedes LC, et al. Convergence of miRNA expression profiling, α-synuclein interacton and GWAS in Parkinson’s disease. PloS one. 2011;6(10):e25443. PubMed CrossRef
- Doxakis E. Post-transcriptional regulation of alpha-synuclein expression by mir-7 and mir-153. J Biol Chem. 2010;285(17):12726–12734. PubMed CrossRef
- Botta-Orfila T, Morató X, Compta Y, et al. Identification of blood serum micro-RNAs associated with idiopathic and LRRK2 Parkinson’s disease. J Neurosci Res. 2014;92(8):1071–1077. PubMed CrossRef
- Wang G, van der Walt JM, Mayhew G, et al. Variation in the miRNA-433 binding site of FGF20 confers risk for Parkinson disease by overexpression of alpha-synuclein. Am J Hum Genet. 2008;82(2):283–289. PubMed CrossRef
- Martinez B, Peplow PV. MicroRNA biomarkers in frontotemporal dementia and to distinguish from Alzheimer’s disease and amyotrophic lateral sclerosis. Neural Regen Res. 2022;17(7):1412–1422. PubMed CrossRef
- Denk J, Oberhauser F, Kornhuber J, et al. Specific serum and CSF microRNA profiles distinguish sporadic behavioural variant of frontotemporal dementia compared with Alzheimer patients and cognitively healthy controls. PloS one. 2018;13(5):e0197329. PubMed CrossRef
- Musso N, Bivona D, Bonomo C, et al. Investigating microRNAs as biomarkers in disorders of consciousness: a longitudinal multicenter study. Sci Rep. 2023;13(1):18415. PubMed CrossRef
- Zhang J, Chen Z, Chen H, et al. Recent advances in the roles of MicroRNA and MicroRNA-based diagnosis in neurodegenerative diseases. Biosensors. 2022;12(12):1074. PubMed CrossRef
- Loffreda A, Nizzardo M, Arosio A, et al. miR-129-5p: a key factor and therapeutic target in amyotrophic lateral sclerosis. Prog Neurobiol. 2020;190:101803. PubMed CrossRef
- Butovsky O, Jedrychowski MP, Cialic R, et al. Targeting miR-155 restores abnormal microglia and attenuates disease in SOD1 mice. Ann Neurology. 2015;77(1):75–99. PubMed CrossRef
- Vaz AR, Vizinha D, Morais H, et al. Overexpression of miR-124 in motor neurons plays a key role in ALS pathological processes. Int J Mol Sci. 2021;22(11):6128. PubMed CrossRef
- Daneshafrooz N, Joghataei MT, Mehdizadeh M, et al. Identification of let-7f and miR-338 as plasma-based biomarkers for sporadic amyotrophic lateral sclerosis using meta-analysis and empirical validation. Sci Rep. 2022;12(1):1373. PubMed CrossRef
- Liu Y, Ding M, Pan S, et al. MicroRNA-23a-3p is upregulated in plasma exosomes of bulbar-onset ALS patients and targets ERBB4. Neuroscience. 2023;524:65–78. PubMed CrossRef
- Cipriano GL, Schepici G, Mazzon E, et al. Multiple sclerosis: roles of miRNA, lcnRNA, and circRNA and their implications in cellular pathways. Int J Mol Sci. 2024;25(4):2255. PubMed CrossRef
- Ma X, Zhou J, Zhong Y, et al. Expression, regulation and function of microRNAs in multiple sclerosis. Int J Med Sci. 2014;11(8):810–818. PubMed CrossRef
- Wang L, Liang Y. MicroRNAs as T Lymphocyte regulators in multiple sclerosis. Front Mol Neurosci. 2022;15:865529. PubMed CrossRef
- De Vito F, Musella A, Fresegna D, et al. MiR-142-3p regulates synaptopathy-driven disease progression in multiple sclerosis. Neuropathology Appl Neurobiol. 2022;48(2):e12765. PubMed CrossRef
- Manna I, Iaccino E, Dattilo V, et al. Exosome-associated miRNA profile as a prognostic tool for therapy response monitoring in multiple sclerosis patients. FASEB J Official Publ Fed Am Soc Exp Biol. 2018;32(8):4241–4246. PubMed CrossRef
- Zabalza A, Pappolla A, Comabella M, et al. MiRNA-based therapeutic potential in multiple sclerosis. Front Immunol. 2024;15:1441733. PubMed CrossRef
- He S, Liu X, Jiang K, et al. Alterations of microRNA-124 expression in peripheral blood mononuclear cells in pre-and post-treatment patients with major depressive disorder. J psychiatric Res. 2016;78:65–71. PubMed CrossRef
- Van der Zee YY, Eijssen LMT, Mews P, et al. Blood miR-144-3p: a novel diagnostic and therapeutic tool for depression. Mol Psychiatry. 2022;27(11):4536–4549. PubMed CrossRef
- Valiuliene G, Valiulis V, Zentelyte A, et al. Anti-neuroinflammatory microRNA-146a-5p as a potential biomarker for neuronavigation-guided rTMS therapy success in medication resistant depression disorder. Biomed Pharmacother = Biomedecine Pharmacother. 2023;166:115313. PubMed CrossRef
- Li YJ, Xu M, Gao ZH, et al. Alterations of serum levels of BDNF-related miRNAs in patients with depression. PloS one. 2013;8(5):e63648. PubMed CrossRef
- Ortega MA, Alvarez-Mon MA, García-Montero C, et al. MicroRNAs as critical biomarkers of major depressive disorder: a comprehensive perspective. Biomedicines. 2021;9(11):1659. PubMed CrossRef
- Banach E, Dmitrzak-Weglarz M, Pawlak J, et al. Dysregulation of miR-499, miR-708 and miR-1908 during a depression episode in bipolar disorders. Neurosci Lett. 2017;654:117–119. PubMed CrossRef
- Rong H, Liu TB, Yang KJ, et al. MicroRNA-134 plasma levels before and after treatment for bipolar mania. J Psychiatr Res. 2011;45(1):92–95. PubMed CrossRef
- Beveridge NJ, Tooney PA, Carroll AP, et al. Dysregulation of miRNA 181b in the temporal cortex in schizophrenia. Hum Mol Genet. 2008;17(8):1156–1168. PubMed CrossRef
- Song HT, Sun XY, Zhang L, et al. A preliminary analysis of association between the down-regulation of microRNA-181b expression and symptomatology improvement in schizophrenia patients before and after antipsychotic treatment. J psychiatric Res. 2014;54:134–140. PubMed CrossRef
- Murphy CP, Singewald N. Role of MicroRNAs in anxiety and anxiety-related disorders. Curr Top Behav Neurosci. 2019;42:185–219. PubMed CrossRef
- Lin Q, Wei W, Coelho CM, et al. The brain-specific microRNA miR-128b regulates the formation of fear-extinction memory. Nat Neurosci. 2011;14(9):1115–1117. PubMed CrossRef
- Chen CY, Wang YF, Lei L, et al. MicroRNA-specific targets for neuronal plasticity, neurotransmitters, neurotrophic factors, and gut microbes in the pathogenesis and therapeutics of depression. Prog neuro-psychopharmacology Biol Psychiatry. 2024;136:111186. PubMed CrossRef
- Tsermpini EE, Kalogirou CI, Kyriakopoulos GC, et al. miRNAs as potential diagnostic biomarkers and pharmacogenomic indicators in psychiatric disorders. Pharmacogenomics J. 2022;22(4):211–222. PubMed CrossRef
- Zheng YB, Jin X. Evidence for the contribution of the miR-206/BDNF pathway in the pathophysiology of depression. Int J Neuropsychopharmacol. 2024;27(10):pyae039. PubMed CrossRef
- Liu Y, Yang X, Zhao L, et al. Increased miR-132 level is associated with visual memory dysfunction in patients with depression. Neuropsychiatric Dis Treat. 2016;12:2905–2911. PubMed CrossRef
- Martinez B, Peplow PV. MicroRNAs as potential biomarkers for diagnosis of post-traumatic stress disorder. Neural Regen Res. 2025;20(7):1957–1970. PubMed CrossRef
- Kang HJ, Yoon S, Lee S, et al. FKBP5-associated miRNA signature as a putative biomarker for PTSD in recently traumatized individuals. Sci Rep. 2020;10(1):3353. PubMed CrossRef
- Fries GR, Carvalho AF, Quevedo J. The miRNome of bipolar disorder. J Affect Disord. 2018;233:110–116. PubMed CrossRef
- Camkurt MA, Karababa İF, Erdal ME, et al. MicroRNA dysregulation in manic and euthymic patients with bipolar disorder. J Affect Disord. 2020;261:84–90. PubMed CrossRef
- Li K, Zhu L, Lv H, et al. The role of microRNA in schizophrenia: a scoping review. Int J Mol Sci. 2024;25(14):7673. PubMed CrossRef
- He K, Guo C, Guo M, et al. Identification of serum microRNAs as diagnostic biomarkers for schizophrenia. Hereditas. 2019;156:23. PubMed CrossRef
- Ma J, Shang S, Wang J, et al. Identification of miR-22-3p, miR-92a-3p, and miR-137 in peripheral blood as biomarkers for schizophrenia. Psychiatry Res. 2018;265:70–76. PubMed CrossRef
- Koskinen MK, Hovatta I. Genetic insights into the neurobiology of anxiety. Trends Neurosciences. 2023;46(4):318–331. PubMed CrossRef
- Ragu Varman D, Marimuthu G, Rajan KE. Environmental enrichment upregulates micro-RNA-183 and alters acetylcholinesterase splice variants to reduce anxiety-like behavior in the little Indian field mouse (Mus booduga). J Neurosci Res. 2013;91(3):426–435. PubMed CrossRef
- Brenes JC, Lackinger M, Höglinger GU, et al. Differential effects of social and physical environmental enrichment on brain plasticity, cognition, and ultrasonic communication in rats. J Comp neurology. 2016;524(8):1586–1607. PubMed CrossRef
- Blount GS, Coursey L, Kocerha J. MicroRNA networks in cognition and dementia. Cells. 2022;11(12):1882. PubMed CrossRef
- Freischmidt A, Muller K, Zondler L, et al. Serum microRNAs in sporadic amyotrophic lateral sclerosis. Neurobiol Aging. 2015;36(9):2660.e15–2660.e20. PubMed CrossRef
- Liu J, Zhou F, Guan Y, et al. The biogenesis of miRNAs and their role in the development of amyotrophic lateral sclerosis. Cells. 2022;11(3):572. PubMed CrossRef
- Woldemichael BT, Mansuy IM. Micro-RNAs in cognition and cognitive disorders: potential for novel biomarkers and therapeutics. Biochem Pharmacol. 2016;104:1–7. PubMed CrossRef
- Ilieva MS. Non-Coding RNAs in neurological and neuropsychiatric disorders: unraveling the hidden players in disease pathogenesis. Cells. 2024;13(12):1063. PubMed CrossRef
- Issler O, Chen A. Determining the role of microRNAs in psychiatric disorders. Nat Rev. Neurosci. 2015;16(4):201–212. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!